All published articles of this journal are available on ScienceDirect.
Arch Measurement Changes upon Biomimetic Oral Appliance Therapy for Adults with Obstructive Sleep Apnea
Abstract
Background:
Obstructive Sleep Apnea (OSA) is the most common form of sleep disordered breathing. Patients who arrive at the dental office with a diagnosis of OSA are often treated with a mandibular advancement device (MAD). A biomimetic oral appliance therapy (BOAT) offers an alternative nonsurgical method, which can putatively resolve OSA by combining maxilla-mandibular correction and addressing craniofacial deficiencies.
Aim:
To determine whether maxilla-mandibular correction changes induced by BOAT produce a more favorable upper airway, which might result in a reduction in the severity of OSA.
Methods:
Patients who were diagnosed with mild to moderate obstructive sleep apnea (OSA, 9 males, 8 females; age, mean (SD): 45.76(10.31), BMI mean (SD): 33.5(13.43), underwent BOAT therapy. Subjects had 2 months of follow-up visits, including examinations for progress and adjustment of the appliances. The mean apnea-hypopnea index (AHI) with no appliance in the mouth prior to BOAT and after treatment was recorded. The mid-palatal screw mechanism of the appliance was advanced once per week. The subjects were asked to wear the appliance for 10-12 hours/day and night. Paired T-Test was used to analyze the results.
Results:
The BOAT treatment enhanced upper airway function as the total AHI was significantly lower after treatment (P=0.019). Parameters that were significantly improved by the end of the treatment period included total AHI/Per hour of sleep (p=0.019), NREM-AHI (p=0.019), desaturation index (p=0.041), average SpO2 (p=0.088), and average O2 while in non-REM (p=0.043). Measurements of jaw changes were all statistically significant except lower 6-6 and lower 7-7. Additionally, a strong negative correlation between AHI and jaw changes was shown for upper 6-6 (p=-0.52), upper 7-7 (p=-0.48), and lower 3-3 (p=-0.42).
Conclusion:
The BOAT provides a useful form of therapy for the resolve of OSA. This study suggests that BOATS may be able to reduce the AHI to within normal limits. Still, long-term follow-up is needed to determine whether these subjects need a maintenance program to retain their initial upper airway improvement.
1. INTRODUCTION
Obstructive Sleep Apnea (OSA) is life-threatening condition [1]. It is characterized by repetitive collapse of the pharyngeal walls during sleep, causing a disturbance in gas exchange, which leads to oxygen desaturation, hypercapnia, and sleep fragmentation [2]. The spectrum of the collapse of the pharyngeal walls ranges from partial airway collapse manifested as loud snoring and episodes of hypopnea, to complete blockage of the airway lasting up to sixty seconds [3]. It is thought that the pathophysiology of OSA involves factors that relate to the anatomical dimensions of the upper airway, upper airway resistance, and upper airway muscle activity during sleep [4].
OSA is a condition that can easily remain undiagnosed. Patients with OSA report symptoms such as snoring, witnessing apnea, waking up with a choking sensation, excessive tiredness, and sleepiness [2]. Morning headaches, sexual dysfunction, hallucinations, and short-term memory loss are other problems related to OSA [1, 5, 6]. The rate of automobile accidents and cardiovascular morbidity and mortality are all increased in patients with OSA [3].
Patients with a diagnosis of OSA are often treated with a mandibular advancement device (MAD) using orthodontic functional appliances [7]. Previous studies have established that determining the craniofacial and dentofacial skeletal characteristics is essential before mandibular retrusion is instituted [8, 9]. Biomimetic oral appliance therapy (BOAT) offers an alternative nonsurgical method, which can putatively resolve OSA. In fact, oral appliances are considered an alternative treatment for mild to moderate OSA and for patients who failed to adhere to or refused Continuous Positive Airway Pressure (CPAP) treatment [10]. Oral appliances reduced the Apnea-Hypopnea Index (AHI) [11].
BOAT has been suggested as a novel approach to mild to moderate OSA in multiple studies [12, 13]. BOAT appliance is designed to correct maxillo-mandibular underdevelopment in both children and adults [14]. Typically, the BOAT appliance consists of 6 patented, anterior 3-D Axial Springs™, a midline actuator (such as omega loops or screws), posterior occlusal rests, and a round labial bow (Fig. 1). It combines maxilla-mandibular correction and addressing craniofacial deficiencies [12, 13]. This study aims to investigate the effect of a novel protocol of BOAT using Daytime-Nighttime Appliance (DNA appliance® system) on the maxilla-mandible jaws changes, as a method to treat patients with OSA.

2. MATERIALS AND METHODS
2.1. Participants
Patient recruitment was based on referrals from the sleep medicine clinic at the King Abdullah University Hospital (KAUH) in Jordan. All patients undertook a sleep test and were diagnosed with OSA prior to participation in the study. Patients who were diagnosed by the sleep physician were recruited after signing informed consent to a protocol that had been reviewed and approved by the institutional review board (IRB) at Jordan University of Science and Technology (JUST) (IRB number is 15/108/2017, ClinicalTrials.gov Identifier: NCT05087316). The subjects were protected by following the Declaration of Helsinki of 1975, as revised in 1983. Additionally, the research team had undertaken courses at JUST- and South Korea on the diagnosis of OSA patients and treating them using the BOAT device.
| Upper3-3 | Upper right canine cusp tip to left canine cusp tip |
|---|---|
| Upper4-4 | Upper right buccal cusp tip of 1st premolar to left buccal cusp tip of 1st premolar |
| Upper6-6 | Upper right mesiobuccal cusp tip of 1st molar to left mesiobuccal cusp tip of 1st molar |
| Upper7-7 | Upper right mesiobuccal cusp tip of 2nd molar to left mesiobuccal cusp tip of 2nd molar |
| Lower3-3 | Lower right canine cusp tip to left canine cusp tip |
| Lower4-4 | Lower right buccal cusp tip of 1st premolar to left buccal cusp tip of 1st premolar |
| Lower6-6 | Lower right mesiobuccal cusp tip of 1st molar to left mesiobuccal cusp tip of 1st molar |
| Lower7-7 | Lower right mesiobuccal cusp tip of 2nd molar to left mesiobuccal cusp tip of 2nd molar |
Included in the study were patients (18 years or older) with a confirmed diagnosis of mild to moderate OSA with good oral hygiene, patients with severe OSA but are not complaint to CPAP, and patients having enough number of maxillary and mandibular teeth to retain the removable device. Excluded were patients who are unable to attend regular appointments and unable to consent or patients diagnosed with severe OSA who are continuously using CPAP devices. A total of 150 patients with OSA were screened for suitability between the period of March 2017 - March 2019. One third (50 patients) had mild to moderate OSA and met the inclusion criteria of the study (Table 1). The 50 eligible patients were screened by a trained clinical investigator for suitability for treatment with BOAT devices. Only 27 patients were suitable for treatment. 25 of the 27 patients consented to participate in the study. Of them, 23 attended their appointments regularly, and 17 out of the 23 patients completed the study and underwent post-overnight sleep study. The eligible patients were given BOAT to use. Changes in sleep parameters such as AHI and saturation of peripheral oxygen (SPO2) before and after treatment were evaluated using polysomnography at KAUH, Irbid, Jordan. Polysomnography studies were conducted with attended in-laboratory PSG (PSG single) at the sleep laboratory using Alice (Alice 6; Philips Respironics, Murrysville, USA). A standard PSG montage was used for each attended in-laboratory evaluation and included electroencephalogram (EEG, F4, C4, O2), electrooculogram (EOG), chin electromyogram (EMG), leg EMG, electrocardiogram (ECG), breathing effort parameters, airflow parameters, oxygen saturation (SpO2, finger probe), and body position, according to AASM recommendations. The data were manually scored using Philips G3 software. Sleep stages, arousals, and abnormal respiratory events were quantified according to AASM 2007 criteria.
The Arc measurements were evaluated by measuring the distances between cusp tips of certain teeth. Cusp tips from right canine to left canine, cusp tips from right first premolar to left premolar, mesiolingual cusp of right first molar to left first molar. Measurement changes were determined by quantifying the changes in maxillary and mandibular arch dimensions before and after the treatment.
2.2. Setting of Participants for BOAT
After careful history taking and craniofacial examination, a bite registration was obtained in the upright sitting position with corrected jaw posture in the vertical dimension specific for each participant. Upper and lower impressions were taken for each patient. For that purpose, upper and lower models were mounted on articulator in the dental lab by dental technician. Using the bite registration, the BOATS were custom fabricated for each participant in the dental laboratory and delivered by the trained clinical investigator. Participants were instructed to wear the device for a period of 12-16 hours/day in the evening and at night. Patients were informed not to wear the device while eating [12]. Patients were trained on inserting and removing the appliance. Written and verbal instructions were given to all participants.
2.3. Follow Visits
All participants were recalled for a monthly review. The devices were professionally adjusted every 4 weeks to optimize their efficacy. At each follow-up, observation of the progress of midfacial development was observed and any needed adjustment was accomplished. Impressions were taken of the maxilla and mandible to produce study models during each follow-up visit. Study models that have orthodontic bases were scanned using Sirona dental scanner (Germany). Changes in the width were measured using computer software called 3Shape (Copenhagen, Denmark), and measurements were recorded. The width of jaws on the scanned models was measured and recorded prior to and at the end of the treatment according to the identified points (Table 1).
On insertion, the devices were checked for accuracy, ensured fit, and expansion of the palatal screw. Only gentle pressures were transmitted to the teeth and surrounding tissues. The functionality of the device was checked with the patient activating a mild force on biting.
At the last follow-up visit, post-treatment sleep tests were accomplished with no appliance in the mouth and monitored by a sleep physician. The mean AHI and other parameters including sleep latency (min), waking after sleep onset (WASO, min), total sleep time (min), sleep efficiency (%), total AHI/hour of sleep (count/hour), NREM-AHI (count/hour), desaturation index (count/hour), average SpO2 (%), approximate minimum O2 Value (%), average O2 while awake (%), and average O2 while in non-REM (%). Those parameters were calculated and compared to their values at the diagnosis stage, and at the end of the treatment period.
2.4. Data Analysis
The results were subjected to statistical analysis using the Statistical Package for Social Sciences (SPSS software, Version 23, IBM, Chicago, IL, USA). Paired t-tests to compare the sleep study measures and the changes in the maxilla-mandible jaws before and after the BOAT treatment. P< 0.05 was considered significant. Data were presented as mean±SD.
3. RESULTS
From the consecutive series of patients, 17 patients (9 males, 8 females); age, mean (SD): 45.76(10.31), were set for the post-treatment measurements and committed to the end of the treatment process. Change in body mass index (BMI) was calculated and recorded from the patient’s height and weight in standard units (kg/m2). The pretreatment BMI mean (SD) 32.85 (12.44), and posttreatment 33.5 (13.43).
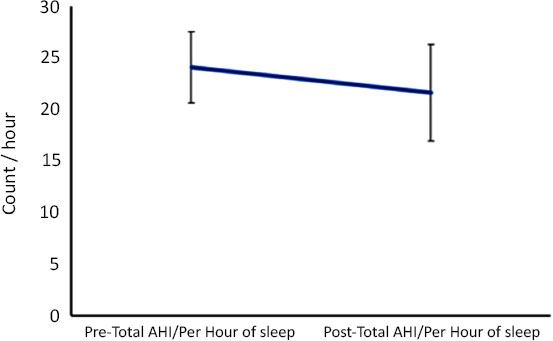
Prior to treatment, at the diagnosis stage, the mean AHI of the sample (n= 17) was 24.0. The mean AHI fell by 5% to 22.8 (p = 0.019) after treatment, indicating enhanced upper airway functions. These results are summarized in Fig. (2). Measurement of sleep test was analyzed using t-test (Table 2). Parameters that were significantly improved by the end of the treatment period included total AHI/Per hour of sleep (p=0.01), NREM-AHI (p=0.02), desaturation index (p=0.04), average SpO2 (p=0.09), and average O2 while in non-REM (p=0.04).
Measurements of jaw changes were all statistically significant except lower 6-6 (p=0.076537) and lower 7-7 (p= 0.06), (Table 3). Additionally, a strong negative correlation between AHI and jaw changes was shown for upper 6-6 (p=-0.52), upper 7-7 (p=-0.48), and lower 3-3 (p=-0.42). On the other hand, no significant correlation was found between SPO2 with jaw changes (Table 3).
| - | Pre-treatment | Post-treatment | P- value |
|---|---|---|---|
| Total recording time (min) | 366.6±61.2 | 402.4±46.2 | 0.13 |
| Sleep latency (min) | 19.59±14.6 | 23.55±12.7 | 0.51 |
| Walking After sleep (min) | 69.99±54.88 | 83.81±41.83 | 0.53 |
| Total sleep time (min) | 276.1±91.72 | 305.0±64.12 | 0.40 |
| Sleep Efficiency (%) | 74.58±17.56 | 74.77±11.72 | 0.98 |
| Total AHI/Per hour of sleep (count/hr) | 26.35±10.7 | 11.81±10.1 | 0.01 |
| NREM_AHI (count/hr) | 25.26±11.47 | 11.10±10.59 | 0.02 |
| Desaturation index (count/hr) | 23.94±10.60 | 12.70±7.50 | 0.04 |
| Average SpO2 (%) | 91.86±2.85 | 93.80±1.55 | 0.09 |
| Approximate Min O2 (%) | 80.13±21.21 | 84.70±3.96 | 0.29 |
| Average O2 while awake (%) | 94.50±1.58 | 94.55±1.44 | 0.95 |
| Average O2 while in non-REM (%) | 93.44±1.83 | 94.88±0.78 | 0.04 |
| - | P-value | t | AHI r* | SpO2 r* |
|---|---|---|---|---|
| Upper 3-3 | 0.015246 | 2.373358 | -0.1817 | -0.18659 |
| Upper 4-4 | 0.018506 | 2.275088 | -0.31876 | -0.22847 |
| Upper 6-6 | 0.019697 | 2.243193 | -0.52222 | -0.19502 |
| Upper 7-7 | 0.020931 | 2.211985 | -0.48097 | -0.25545 |
| Lower 3-3 | 0.046895 | 1.781677 | -0.42445 | -0.03318 |
| Lower 4-4 | 0.036846 | 1.913921 | -0.29145 | -0.19981 |
| Lower 6-6 | 0.076537 | 1.500051 | -0.2813 | -0.22221 |
| Lower7-7 | 0.055784 | 1.684092 | -0.15704 | -0.23344 |
4. DISCUSSION
The current study identified that the maxillomandibular correction changes induced by BOAT produce a more favorable upper airway, which might result in a reduction in the severity of OSA. The BOAT treatment enhanced upper airway function as the total AHI/hr was significantly lower after treatment. Other parameters that were significantly improved by the end of the treatment period included NREMAHI, desaturation index, average SpO2, and average O2 while in non-REM. Measurements of jaw changes were all statistically significant except lower 6-6 and lower 7-7.
Oral appliances work by repositioning the tongue and the mandible forward and downward to reduce airway collapse, widening the lateral aspects of the pharyngeal walls, thus improving the airway patency [15]. Thus, protrusion of the mandible is required for the effectiveness of MADs. It has been reported that the effective degrees of advancement range from 6 to 10 mm or from 65% to 70% of maximum protrusion [16].
The rationale for using biomimetic oral devices is based on the idea that the upper airway is a complex adaptive system, which can undergo remodelling in pathologic conditions. A study showed that nonsurgical maxillary expansion is possible in adults, and possibly curative of OSA. It is conceptually based on the epigenetic premise that the potential for craniofacial growth and development remains intact within and individual and that it can be accomplished with expansion oral appliances [17].
A recent systematic review of epidemiological studies across different geographic locations revealed that the prevalence of OSA ranged from 9% up to 38% and was consistently higher in men [18]. Females are also thought to respond better to OSA devices [19]. Overall, the effectiveness of treatment depends on regular and prolonged use of the device. However, self-reported compliance data from available studies suggest that, on average, 77% of patients consistently used their device for one year [20]. The most frequent reasons why patients discontinued MAD use were discomfort, or the MAD had no effect and the subject used CPAP instead. Other side effects of customized MAD use included dry mouth, tooth pain, jaw discomfort, and TMJ symptoms. Thus, subjects who were compliant with customized MAD for long periods of use had adequate improvements if they continued wearing the MAD.
A case report for 15-year-old female with DNA appliances [14] showed at baseline AHI of 21.4 and after treatment AHI of 18.2. This means an AHI change of -15%. Another study demonstrated that in 10 consecutive OSA adult patients that the use of DNA gradually improved the AHI scores in these subjects over 8.7 months by 65.9%. The improvement was attributed to the remodeling of the midfacial bones and the upper airway in these subjects, which was seen in CBCT [12, 13].
Nevertheless, long-term follow-up is needed to confirm these initial findings, as well as assessment of craniofacial and upper airway modifications to determine the stability of the changes achieved. Although there are no studies in the literature comparing patients with OSA on CPAP therapy with untreated controls, the lack of a control group could be another limitation of the current study.
The American Academy of Sleep Medicine classification of OSAS severity considers mild and moderate AHI as: mild, defined as an AHI of 5– 15; moderate as 15–30; and severe as >30). Originally oral appliances, such as BOAT, are indicated and best suited to patients with mild- moderate OSA, however, it can be used for patients with severe OSA who do not complain or refuse to use CPAP. In patients with malocclusions, OSA syndrome, and chewing patterns can be severe. Treatment with mandibular advancement devices has several skeletal and dental side effects, such as reduction in overjet and overbite, and the upper incisal inclination, and an increase in the lower incisal inclination. These changes are found to be dependent on the duration of use of mandibular advancement devices and are considered to be clinically relevant [21].
CONCLUSION
The current study shows that BOAT provides a useful form of therapy to the resolve of OSA via inducing maxillomandibular correction changes. This study suggests that BOATS may be able to reduce the AHI to within normal limits. Still, long-term follow-up is needed to determine whether these subjects need a maintenance program to retain their initial upper airway improvement.
LIST OF ABBREVIATIONS
| OSA | = Obstructive Sleep Apnea |
| MAD | = Mandibular Advancement Device |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The protocol had been reviewed and approved by an appropriate institutional review board at Jordan University of Science and Technology (JUST) (IRB number is 15/108/2017, ClinicalTrials.gov Identifier: NCT05087316).
HUMAN AND ANIMAL RIGHTS
No animals were used that are the basis of this study. The human subjects were protected by following the Declaration of Helsinki of 1975, as revised in 1983.
CONSENT FOR PUBLICATION
The participants were recruited after signing informed consent.
STANDARDS OF REPORTING
STROBE guidelines have been followed
AVAILABILITY OF DATA AND MATERIALS
Data will be available upon request via e-mailing the corresponding author.
FUNDING
This work was supported by the Scientific Research Funds/Ministry of Higher Education and Scientific Research, Amman, Jordan (grant number MPH/2/6/2016). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


