All published articles of this journal are available on ScienceDirect.
Dentinal Tubule Disinfection with Propolis & Two Extracts of Azadirachta indica Against Candida albicans Biofilm Formed on Tooth Substrate
Abstract
Aim:
This study evaluates the disinfection of dentinal tubules using Propolis, Azadirachta indica (alcoholic and aqueous extracts), 2% chlorhexidine gel and calcium hydroxide against Candida albicans biofilm formed on tooth substrate.
Materials & Method:
One hundred and five human teeth were infected with Candida albicans for 2 days. Samples were divided into 7 groups. Group I- Propolis, Group II- Alcoholic extract of Azadirachta indica, Group III- Aqueous extract of Azadirachta indica, Group IV- 2% Chlorhexidine, Group V- Calcium hydroxide, Group VI- Ethanol and Group VII- Saline (negative control). At the end of 1,3 and 5 days, the antimicrobial efficacy of medicaments against Candida albicans was assessed at the depths of 200 µm and 400 µm.
Results:
The overall percentage inhibition of fungal growth (at 200 µm and 400 µm depth) was 99.2% with 2% chlorhexidine gel. There was no statistical difference between propolis, alcoholic extract of Azadirachta indica (neem) and 2% chlorhexidine.
Conclusion:
Propolis and alcoholic extract of Azadirachta indica performed equally well as that of 2% Chlorhexidine.
INTRODUCTION
Dentinal tubules are exposed to microbial invasion when a breach in the integrity of the overlying enamel or cementum occurs, following a cariogenic or a periodontal insult. If this invasion cannot be overcome by the host defences, it results in pulpal necrosis, infection of the root canal system and periapical disease [1]. One of the important objectives of root canal treatment is the elimination of the microorganisms from the root canal system [2]. Several hundred microbial species are known to inhabit the oral cavity but relatively small and selected group of microbes is involved in the invasion of dentinal tubules and subsequent infection of the root canal space. Gram-positive organisms dominate the tubule microflora in both carious and non-carious dentin. It is difficult to deliver intracanal medicaments in the depth of dentinal tubules, leading to survival of the microbes. If opportunistic conditions occur, they may become a focal source of reinfection [3].
Fungi are chemoorganotroph eukaryotic microorganisms that may be found in 2 basic forms: molds and yeasts. Candida species have surface molecules that mediate adherence to host tissues. Candida species are also able to bind to collagen types I and IV. C.albicans produces hydrolytic enzymes that may be involved in the damage to the periradicular tissues [4]. Enzymes include secreted aspartyl proteinase, collagenase, aminopeptidases, glucosaminidases, acid and alkaline phosphatases, hyaluronidase, and chondroitin sulfatase, all of which have some effect in the degradation of extracellular matrix proteins [5]. It has been shown that a collagenolytic enzyme produced by this fungal species may degrade the human dentin collagen [6]. C. albicans is often described as a dimorphic fungus that exists in blastospore and hyphal forms. It has been consistently suggested that the transition from the blastospore to the hyphal form represents a change from a commensal to a pathogenic state [7, 8]. but it has not been found to be true [9]. The properties of a growing hypha may confer the ability to invade host tissues and escape phagocytosis by macrophages [7]. However, although transformation from yeast to hyphal form may be important, it is not always a prerequisite for infection to occur [10]. Indeed, most infections caused by C.albicans are populated by both yeast and filamentous morphologic forms, suggesting that both have a role in the development and progression of the disease [11].
Two percent chlorhexidine is one of the most versatile irrigants and an intracanal medicaments in vital and non-vital teeth. Its efficacy is based on the interaction between the positive charge of the molecule and the negatively charged phosphate groups on the microbial cell wall, which allows the CHX molecule to penetrate into the microbes with toxic effects [12]. It also retains its antimicrobial activity in the presence of blood and other organic matter [13].
Propolis is a brownish resinous substance collected mainly from plants, by the bees. It is a potent antibacterial, antiviral, antifungal and antiprotozoan activities [14]. The main chemical elements present in propolis are flavonoids, phenolics, and various aromatic compounds [15].
Azadirachta indica (Neem) is a tree with medicinal value. Each part of the tree has been explored in phytotherapy. The leaves contain alkaloids, glycosides, saponins, flavonoids, steroids, anthraquinone and tannic acid, which are of medicinal value [16].
Ca(OH)2 is believed to have many of the properties of an ideal root canal dressing, mainly due to its alkaline pH [17, 18]. It is microbicidal [19] and neutralizes the remaining tissue debris in the root canal system. [20] Ca(OH)2 also promotes an alkalinizing osteogenic environment on the surrounding tissues through the continuous release of OH- ions. [17, 21] Furthermore, Ca(OH)2 mediates the neutralization of lipopolysaccharides [22] and thus helps in cleansing the root canal [20].
The aim of this study is to evaluate the dentinal tubule disinfection with Propolis, Alcoholic & aqueous extract of Azadirachta indica, 2% Chlorhexidine and calcium hydroxide against Candida albicans biofim formed on tooth substrate. There has been no study evaluating the dentinal tubule disinfection with Azadirachta indica till date.
MATERIALS & METHOD
Preparation of Dentine Specimens
The model proposed by Haapasalo & Ørstavik (1987) [23] was modified. One hundred and five single-rooted human mandibular premolar teeth freshly extracted for orthodontic reasons were selected for the study.
A rotary diamond disc was used to decoronate the teeth below the cementoenamel junction and the apical part of the root to obtain 6 mm of the middle third of the root (Fig. 1). Cementum was removed from the root surface. Gates Glidden drills no. 3 in a slow-speed handpiece was used to standardize the internal diameter of the root canals. The specimens were placed in an ultrasonic bath of 17% ethylenediaminetetraacetic acid for 5 min followed by 3% NaOCl for 5 min to remove organic and inorganic debris. The traces of chemicals used were removed by immersing the dentine specimens in an ultrasonic bath containing distilled water for 5 min. All the specimens were then sterilized in an autoclave for two cycles. The first cycle at 121oC and the second with the specimens immersed in 1 mL of tryptone soya (TS) broth in individual microcentrifuge tubes [24].

Prepared specimens.
Contamination of the Specimens
C. albicansATCC 10231 (Himedia, Mumbai L. No. 443-207) was grown in saboraud dextrose agar (SDA) (Himedia, Mumbai) for 24h (Fig. 2). The culture was suspended in 5 mL of SDA broth and incubated for 4 h at 37oC and its turbidity adjusted to 0.5 McFarland standard with sterile SDA broth [25]. Each dentine block was placed in pre-sterilized microcentrifuge tubes containing 1 mL of the TS broth. 50 µL of the inoculums containing the C. albicans was transferred into each of the microcentrifuge tubes. At the end of 24 h, the dentine specimens were transferred into fresh broth containing C. albicans. All procedures were carried out under laminar flow. Purity of the culture was checked by subculturing 5 µL of the broth from the incubated dentine specimens in SDA broth on SDA plates [26]. Contamination of the dentine specimens were carried out for a period of 2 days.
Candida albicans cultured on Sabouraud dextrose Agar.
Antimicrobial Assessment
At the end of 2 days, the specimens were irrigated with 5 mL of sterile saline to remove the incubation broth. Then, the specimens were assigned into 7 groups (n = 15 dentine blocks each).
Group 1- Propolis (Herbal Biosolutions, Delhi) was prepared by diluting a 33% commercially available alcoholic extract using warm saline in a ratio 2:1, to form an 11% alcoholic extract [27]. Then, it was made into gel using Hydroxypropyl methylcellulose (Himedia).
Group 2- Alcoholic extract of Azadirachta indica was prepared using 25 g of fresh neem leaves powder of 99% purity (The Indian Neem Tree Company, Mumbai) was added to 50 ml of absolute ethanol (Sterling Chemicals and Alcohols Pvt. Ltd., Mumbai). Mixture was macerated for 1-2 min, then extract was filtered through muslin cloth for coarse residue and then through filter paper for finer residue [28]. It was made into a gel by using HPMC.
Group 3- Aqueous extract of Azadirachta indica (Neem) was prepared by mixing 25 g of dry powder of neem leaves (The Indian Neem Tree Company, Mumbai) with 100 ml of sterile distilled water in a round bottom flask with occasional shaking. The extracts were then filtered through a muslin cloth for coarse residue and then through a Whatman no. 1 filter paper and kept in an airtight amber‑colored container [29] It was made into a gel by using HPMC.
Group 4- 2% Chlorhexidine diacetate powder (2% solution was made and then gel was prepared by mixing it with HPMC as a thickening agent and distilled water) [30].
Group 5- Calcium Hydroxide + HPMC (150 mg of calcium hydroxide powder was mixed with 0.15 ml of the vehicle, i.e., distilled water) [31].
Group 6- Absolute Ethanol
Group 7- Sterile Saline (Negative Control)
Hydoxypropyl methyl cellulose (HPMC) was used as a thickening agent for Groups 1,2,3,4 and 5 to standardize all the test solutions, which is an inert polymer [32].
The medicaments were placed inside the canals and sealed at both ends with paraffin wax. They were then incubated in an aerobic environment for 37oC. At the end of 1, 3, and 5 days (n=5 each), harvesting of dentine was carried out at two depths (200 and 400 µm) with Gates Glidden drills no 4 and 5, respectively [24]. The collected dentine shavings were transferred into 1 mL of sterile SDA broth and incubated in an aerobic environment at 37oC for 24 h. After 24 h, the contents of each tube were serially diluted, 100 µL of the broth in 100 µL of sterile saline five times. 50 ìL of the dilution was then plated on SDA plates and incubated for 24 h. Colonies were counted and readings were tabulated. The results were statistically analysed by ANOVA and post hoc tukey test.
RESULTS
The current study showed that 5 medicaments studied exerted antifungal activity. Contamination of the dentin blocks was confirmed when debris samples harvested from the saline group (negative control) yielded positive growth. Table 1 shows the antifungal activity, measured at 2 depths (200 and 400 μm) and at 3 time intervals (1, 3, and 5 days).
Mean & Standard deviation of colony counts for different intracanal medicaments at 200 and 400 µm depths at different time intervals.
| Group | Day 1 | Day 3 | Day 5 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 200 µm | 400 µm | 200 µm | 400 µm | 200 µm | 400 µm | |||||||
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | |
| Propolis | 0.20 | 0.42 | 0.40 | 0.52 | 0.50 | 0.53 | 0.60 | 0.52 | 3.50 | 0.71 | 4.40 | 0.52 |
| Neem (Alcoholic extract) | 1.60 | 0.52 | 3.20 | 0.42 | 5.50 | 0.53 | 9.40 | 0.52 | 12.60 | 0.52 | 17.40 | 0.52 |
| Neem (Aqueous extract) | 8.40 | 0.52 | 12.80 | 0.42 | 15.50 | 0.53 | 19.40 | 0.52 | 22.60 | 0.52 | 27.40 | 0.52 |
| 2% Chlorhexidine | 0.00 | 0.00 | 0.00 | 0.00 | 0.30 | 0.48 | 0.50 | 0.53 | 1.40 | 0.52 | 2.50 | 0.53 |
| Calcium hydroxide | 28.40 | 0.52 | 42.80 | 0.42 | 55.50 | 0.53 | 69.40 | 0.52 | 82.60 | 0.52 | 87.40 | 0.52 |
| Ethanol | 68.40 | 0.52 | 72.80 | 0.42 | 95.50 | 0.53 | 109.40 | 0.52 | 122.60 | 0.52 | 137.40 | 0.52 |
| Saline | 69.60 | 0.52 | 74.70 | 0.48 | 96.50 | 0.53 | 110.60 | 0.52 | 124.70 | 0.48 | 139.60 | 0.52 |
There was no significant difference between Propolis and Neem (Alc. extract); Propolis and 2% Chlorhexidine; Neem (Alc.) and 2% Chlorhexidine and Ethanol and Saline, whereas all the other groups had significant differences. On the basis of above analysis, the following order of antimicrobial efficacy of different medicaments was observed (Fig. 3):
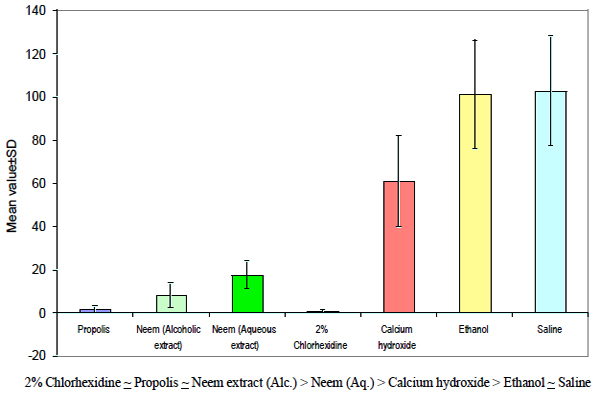
Graph indicating the antimicrobial efficacy of different groups.
DISCUSSION
The in vitro model developed by Haapasalo & Ørstavik (1987) [23] was modified and used to assess the efficacy of endodontic medicaments in the disinfection of dentinal tubules. Human permanent teeth were used instead of the bovine teeth as suggested by Basrani et al. [33], as human dentin blocks would simulate the clinical scenario better. Lynne et al. [34] further modified this model to include quantitative analysis of microbes in the dentinal tubules to define a percentage of reduction in CFU in infected dentine before and after the application of intracanal medicaments. Since apical dentine is mostly sclerotic and physically impedes microbial invasion, mid-root sections were taken. Invasion of coronal and mid-root dentin occurs more readily whereas the extent and depth of invasion are significantly less in apical dentin, because of less patent dentinal tubules [35] The samples were tested at two depths, 200µm and 400µm, because calcium hydroxide is known to penetrate only upto 200 to 300 µm [36]. Thus, disinfection can be assessed better at these two depths. Three time periods have been taken to assess the disinfection because the effective antimicrobial action of the tested intracanal medicaments decreased after 48 hours [37], therefore, time periods chosen to evaluate the antimicrobial assessment were first and third day. Panagiotis et al. [38] have shown that a 5-day period is adequate for Ca(OH)2 pastes to release the OH- ions, so an additional time period of 5 days was also taken.
The microorganism used in the study was Candida albicans (ATCC 10231) since it is the most commonly encountered fungus in the root canals, 21% in primary infections [39] and 18% in cases of retreatments [40]. Dimorphic switching is observed in this species, which is a pivotal factor for biofilm formation and the pathogenic potential of C. albicans. There is an initial period of adherence (0-2 h) followed by microcolony formation (2-4 h). Dimorphic switching occurs thereafter with a transition from budding-yeast forms to filamentous pseudo- and true hyphal forms (4-6 h). The complexity of the biofilm increases with time, taking on three dimensional architecture with spatial heterogeneity as it matures (8-48 h) [25], which is why the 48 h biofilm model was used. Sen et al. [41] investigated the growth patterns of C.albicans in relation to human radicular dentin and observed blastospores and hyphal structures on the root canal walls of all specimens. Dentin colonization may be an important step during infection of the root canal system. The invasion of dentinal tubules can protect microbial cells from the effects of intracanal procedures and may play an important role in the establishment of persistent root canal infections. Yeasts are on average 1 to 6 µm in diameter, whereas hyphae are usually 1.9 to 2.6 µm in diameter [42]. It has also been proposed that the contact-sensing (thigmotropism) ability of hyphal structures of Candida makes dentinal invasion inevitable. Therefore, on the basis of this invasive affinity to dentin, C.albicans is considered a dentinophilic microorganism [43]. Thus, the means to eradicate C.albicans from root canals becomes important for complete disinfection and to avoid reinfection.
Intracanal medicament possessing antimicrobial properties between appointments may reduce or eliminate microbes in the root canal system, thereby increasing the predictability and success of root canal treatment [43]. In our study, 2% CHX has shown the 99.2% microbial inhibition at depths of 200 and 400 µm from day 1 to 5, which is not in accordance with the other studies in which 100% inhibition has been shown. The possible reasons for this variance might be dentine, dentine components (hydroxyapatite & collagen), heat-killed microbial cells and inflammatory exudates in the root canal system, which are inhibitors of CHX [44]. Yet, it has given maximum inhibition in our study. The explanation for this might be its high microbicidal dosage of 2% and increased diffusion of the medicament into the dentinal tubules [45]. Since, it is a positively charged hydrophobic and lipophilic molecule, it interacts with negatively charged phospholipids and lipopolysaccharides on the cell membrane of fungus and enters the cell through some type of active or passive transport mechanism, which alters the osmotic equilibrium of the cells. This increases the permeability of the cell wall, allowing the CHX molecule to penetrate into the micro-organism, followed by leakage of intracellular constituents, particularly phosphate entities such as adenosine triphosphate and nucleic acids. As a consequence, the cytoplasm becomes congealed, with resultant reduction in leakage; thus, there is a biphasic effect on membrane permeability [15]. It also binds to hydroxyapatite and soft tissues, changing their electrical field to compete with microbial binding, thus decreasing microbial adherence. The result of the present study was similar to that of Krithikadatta et al. [30], Gomes et al., [46] and Siqueria and Uzeda, [47] and Bhardwaj et al. [45] Basrani et al. [33] showed that 2% chlorhexidine gel produced a better antimicrobial action as compared to 0.2% chlorhexidine gel or calcium hydroxide mixed with 0.2% chlorhexidine.
In our study, Propolis showed 98.5% microbial inhibition irrespective of depth and time, with no significant difference in comparison to the chlorhexidine group. Propolis exhibits antimicrobial, anti-inflammatory, healing, anesthetic and cariostatic properties [48]. Some components present in propolis extract, like flavonoids (quercetin, galangin, pinocembrin), caffeic acid, benzoic acid and cinnamic acid, probably act on the microbial membrane or cell wall site, causing functional and structural damages [25]. Takaisi- Kikuni and Schilcher [49] have reported that it prevents fungal cell division and also breaks down fungal cell wall and cytoplasm similar to the action of some antibiotics. Kujumgiev et al. [50] have reported that the antimicrobial action of propolis can be due to flavonoids and esters of phenolic acids. Koo et al. [51] tested the antimicrobial activity of propolis using the agar diffusion method and observed that the propolis extract significantly inhibited all of the microorganisms tested, including facultative and strict anaerobic species. Oncag et al. [52] observed that propolis had good in vitro antimicrobial activity against E. faecalis in the root canals of extracted teeth, suggesting that it could be used as an alternative intracanal medicament.
Azadirachta indica (Neem) has also been used in the study. There has been no study so far of dentinal tubule disinfection with Azadirachta indica (AI), this probably is the first study of its kind. The presence of active constituents such as nimbidin, nimbin, nimbolide, gedunin, azadirachtin, mahmoodin, margolone, and cyclictrisulphide contributes to the antifungal activity of AI [53]. These active constituents uncouple mitochondrial oxidative phosphorylation; thus, inhibiting the respiratory chain [54]. This resulted in its anti-adherence activity by altering the microbial adhesion and the ability of the microorganism to colonize thereby causing maximum reduction in adherence to dentin. Rajashekharan et al. [55] have proposed that the inhibitory activities of the leaf extracts of AI were both organism and solvent dependent. Thus, to evaluate the effect of solvent on Candida albicans, both aqueous and alcoholic extracts were taken. Nayak et al. [29] conducted a study in which the minimum inhibitory concentration of aqueous extract of AI was 7.5% as compared to the alcoholic extract of AI which was 3.75%, thus showing alcoholic extract to be better and more efficacious, which is in support of our study.
Calcium hydroxide, the gold standard of intracanal medicament was also taken as a test group. Literature indicates that Candida species are resistant to calcium hydroxide, [26] which has been again verified in our study. Waltimo et al. [56] evaluated the susceptibility of 7 strains of C. albicans to 4 disinfectants: iodine potassium iodide, chlorhexidine acetate, sodium hypochlorite, and calcium hydroxide. C. albicans cells were highly resistant to calcium hydroxide. It survives in a wide range of pH values, the alkalinity of saturated calcium hydroxide solution may not have any effect on C. albicans. The alkalinity of calcium hydroxide also reduces due to the buffering capacity of dentin. In addition, calcium hydroxide solution may readily display the Ca++ ions necessary for the growth and morphogenesis of Candida. These mechanisms may explain why calcium hydroxide has been found to be ineffective against C. albicans [57].
Ethanol was also taken as a test group to check if it influenced the activity of propolis or AI. Ethanol showed slight inhibition of microorganism but there was no significant difference between ethanol and saline, which was taken as a negative control. Previously, a study conducted by Ferreira et al., [14] had concluded that Ethanol did not influence the antimicrobial effect of ethanolic extract of propolis.
CONCLUSION
Under the limitations of the study, it can be concluded that Propolis and alcoholic extract of Azadirachta indica performed equally well as that of 2% Chlorhexidine and thus can be probable alternatives to chlorhexidine, but further in vivo and long term studies are warranted.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
Declared none.


