Toll-Like Receptor 2 Gene Polymorphisms Associated with Aggressive Periodontitis in Japanese
Abstract
Background and Objective:
Aggressive periodontitis is a rare and very severe periodontal disease of early onset, which is closely associated with Porphyromonas.gingivalis (P.g.) infection in the Japanese population. TLR2 encodes Toll-like receptor 2, which plays an important role in the protective response to P.g. infection. We investigated a possible association between TLR2 and aggressive periodontitis.
Material and Methods:
Of 2,460 Japanese patients with periodontitis, 38 patients with aggressive periodontitis were enrolled in this study. These 38 aggressive periodontitis patients and 190 Japanese healthy controls were examined for an insertion/deletion (Ins/Del) polymorphism in exon 1, a polymorphism in intron 1 (rs7696323), and a synonymous polymorphism in exon 3 (rs3804100) in TLR2.
Results:
We found significant associations of resistance to aggressive periodontitis with the Ins allele (allele frequency in the patients versus controls, 0.540 vs. 0.676, OR=0.56, 95% confidence interval (CI); 0.34-0.92, p=0.022) and the T allele of rs3804100 (0.579 vs. 0.716, OR=0.55, 95% CI; 0.33-0.91, p=0.018), although the C allele of rs7696323 showed no significant association (0.733 vs. 0.829, OR=0.58). A permutation test of Ins/Del-rs7696323-rs3804100 haplotype revealed a significant association between Ins-C-T haplotype (0.252 vs. 0.479, p=0.0003) and resistance to aggressive periodontitis.
Conclusions:
The TLR2 polymorphisms were suggested to confer protection against aggressive periodontitis in a Japanese population. The association should be replicated in other cohorts to further identify the responsible TLR polymorphism(s) involved in the pathogenesis of aggressive periodontitis.
INTRODUCTION
Aggressive periodontitis (AgP) is a rare and severe phenotype of periodontitis characterized by rapid bone destruction and attachment loss in the absence of any systemic disease, often with familial aggregation, according to the 1999 International Classification of Periodontal Diseases criteria. The rate of AgP crisis is almost 0.03% in Japanese. Periodontitis is caused by infection with gram-negative bacteria and the subsequent inflammatory response induces destruction of periodontal tissue. Progression of AgP is so rapid that, if not treated, supporting tissues around teeth may be lost early in the life. If predisposing factors to AgP would be defined, patients carrying the predisposing factors could be treated in an early stage to prevent disease progression and to save their teeth. It is well known that susceptibility to periodontitis is influenced by both environmental (such as older age, smoking status, plaque control) and genetic factors. To reveal the genetic predisposing factors to periodontal diseases, many association studies have targeted functional polymorphisms in genes associated with inflammatory cytokines for chronic periodontitis. For example, Kornman firstly reported in 1997 that chronic periodontitis was related to interleukin (IL)-1 (IL-1) polymorphism [1] . Following this report, many other studies attempted to elucidate the associations of chronic periodontitis (CP) with polymorphisms in other genes, including IL-6 and Vitamin D receptor (VDR) [2, 3]. These results suggested that unregulated production of inflammatory cytokines would be a risk factor for chronic periodontitis.
Association studies for AgP have also been performed for inflammatory cytokine genes, reporting that the susceptibility to AgP was associated with a IL-13 polymorphism at -1112 in Taiwanese [4], and IL-1α (IL-1A) polymorphism at +4845 and IL-1β (IL-1B) polymorphism at -511 were associated in Chinese males [5]. In addition, the association of AgP with the IL-1 gene cluster region was investigated in an Italian population, which demonstrated a significant association with polymorphisms in the genes for IL-1 receptor antagonist (IL1-RN) and in IL-1B [6]. Furthermore, other gene polymorphisms in genes for oestrogen receptor-α (ERA), VDR, N-formylpeptide receptor (FPR), CC chemokine receptor 2 (CCR2), and monocyte chemoattractant protein I (MCP-1) were reported to be associated with AgP in some ethnic groups [7].
Porphyromonas gingivalis (P.g.) is a predominant gram-negative periodontopathic bacterium which is strongly associated with AgP in Japanese populations [8, 9], although Aggregatibacter actinomycetemcomitans (A.a.) is the strongest candidate of AgP causative bacterium in other ethnic populations [10, 11]. Component of P.g. is reported to be a ligand for TLR2, although A.a. is recognized by TLR4 [12], suggesting that TLR2 plays a key role in protective response against P.g. in Japanese AgP. However, there have been few studies on the relationship between AgP and polymorphisms, in the TLR genes. Although one study demonstrated a significant association between CP and a polymorphism in TLR4 [13], another study could not replicate the significant association [3]. On the other hand, no association was observed with a TLR2 polymorphism, Agg753Gln, in a German population [14], and a study in Japanese subjects also showed no association of CP with the TLR2 polymorphisms, Arg677Trp and Arg753Gln [13]. TLRs are type I integral membrane glycoproteins, which play crucial roles in the innate immune system via recognizing molecules derived from pathogens. There are different TLR types, and different TLRs recognize a variety of pathogen-associated molecules, including lipids and nucleic acids [15]. It was reported that TLR2 is involved in the recognition of pathogenic bacteria in periodontal disease [16-18]. Although the association of AgP with TLR2 polymorphisms accompanied by amino-acid replacement was not significant, another functional polymorphism, an Ins/Del in exon 1, which is related to the expressivity of TLR2 [19], was not tested for the association. In this study, we investigated three tag polymorphisms of TLR2 including the Ins/Del polymorphism in association with AgP in Japanese. We report here the association of TLR2 with protection against AgP.
MATERIALS AND METHODS
Subjects
Two thousands and four hundreds sixty patients with periodontitis who visited Tokyo Medical and Dental University Dental Hospital from June 2009 to July 2010, were examined. All AgP cases, a total of 38 unrelated Japanese patients (21 males and 17 females; diagnosed with AgP based on the 1999 International Workshop for Classification of Periodontal Diseases and Conditions), were recruited for the study. Age at diagnosis of the patients was 34.4±6.1 years. Written informed consent was given by each patient before blood sampling. Genomic DNAs from blood were prepared by a standard method. DNA samples from 190 Japanese individuals with no systemic disease were prepared from healthy-donor derived B cell lines deposited to the Health Science Research Resource Bank, Japan Health Sciences Foundation (http://www.jhsf.or.jp/English/index_e.html). Periodontal conditions in these 190 healthy controls were unknown, although inclusion of possible AgP patients in the control would decrease the power to detect the association. The research was conducted in full accordance with ethical principles, including the World Medical Association Declaration of Helsinki (version 2002), and the study protocol was approved by the Ethics Review Committee of the Faculty of Dentistry, Tokyo Medical and Dental University, and that of the Medical Research Institute, Tokyo Medical and Dental University.
Clinical Information
Periodontal parameters and radiographs of all AgP patients were assessed at their first visit. Probing pocket depth (PPD) was measured at 6 sites per tooth. Bleeding on probing (BOP) was also recorded at 6 sites per tooth. Alveolar bone loss (BL) was investigated at two sites per tooth on the radiographs. The data for frequency of BOP, rate of PPD over 4 mm, rate of PPD over 7 mm, and frequency of alveolar bone loss are summarized in Table 1.
Clinical Characteristics of AgP Patients
| Clinical Parameters#1 | |
|---|---|
| Smokers (%) | 58.1 |
| Number of teeth present (including wisdom teeth) | 26.5±3.5 |
| Mean probing pocket depth (mm) | 3.8±1.0 |
| Frequency of bleeding on probing (%) | 57.9±28.7 |
| Frequency of alveolar bone loss (%) | 41.0±23.0 |
| Rate of probing pocket depth >4 mm (%) | 76.0±22.6 |
| Rate of probing pocket depth >7 mm (%) | 37.0±22.3 |
#1 Value was represented as mean ± standard deviation, when it was appropriate.
Genotyping of TLR2
Human TLR2 is located on chromosome 4q32 and is composed of three exons. Two tag single nucleotide polymorphism (SNP), rs7696323 in intron 1 and rs3804100 in exon 3, were selected from the International HapMap project database (HapMap Data Phase III/Rel#2, Feb09, on NCBI B36 assembly, dbSNP b126) by cut-off criteria of minor allele frequency >0.1 and r square <0.8 in the Japanese population. In addition, an insertion/deletion (Ins/Del) polymorphism at -196 to -174 was investigated. The restriction fragment length polymorphism method was used for the genotyping of rs7696323 and rs3804100. Briefly, polymerase chain reaction (PCR) was performed with primer pairs, 5’-ggcccctagctcctgtcc-3’and 5’-gtgtagcccctgcttctcc-3’ for rs7696323 typing and 5’-tcatttggcatcattggaaa-3’ and 5’-gagttgcggcaaattcaaag-3’ for rs3804100 typing. The PCR condition was 95°C 5 min followed by 35 cycles of 95°C 30 sec, 58°C or 53°C 30 sec for rs7696323 or rs3804100, respectively, and 72°C 30 sec, and a final extension step of 72°C 10 min. The PCR products were digested by BsrI and MwoI for rs7696323 and rs3804100 typing, respectively, followed by electrophoresis in 3% agarose gel. The Ins/Del typing was done by PCR with a primer pair, 5’-gcggactttcccttttgct-3’ and 5’-gtgcagagagaccacacgag-3’ under the PCR condition described above, except that the annealing temperature was 57°C and electrophoresis was done in 2% agarose gel.
Statistical Analysis
Allele and genotype frequencies of the polymorphisms in the patients and controls were obtained by direct counting. Departure from Hardy-Weinberg equilibrium (HWE) was calculated by chi-square test. Statistical significance of differences between the AgP patients and control subjects were assessed by Mantel-Haenzel chi-square analysis, which was combined with the 2 x 2 contingency tables using the GraphPad in Stat software (GraphPad Software Inc., La Jolla, CA, USA). Fisher’s exact test was used if the number of any cells of the 2 x 2 contingency table was <5. Relative risk with 95% confidence interval (CI) was calculated as the odds ratio. Linkage disequilibrium (LD) among the alleles of tested markers and their haplotype frequencies was estimated by using the Haploview program (Haploview 4.2, http://www.broad.mit.edu/mpg/haploview/index.php). Permutation test (n=5,000) was also performed by using Haploview. P values <0.05 were considered to be statistically significant.
RESULTS
Clinical parameters of enrolled AgP patients were shown in Table 1. Every clinical data showed severe progression of periodontal disease at their ages. A typical AgP case is shown in Fig. (1). The patient was 23 years old female, who showed gum of swelling and redness, and her upper right incisor tooth almost came out. She had serious problems in her mouth, but had no systemic disease other than the severe periodontitis. The allele and genotype frequencies of tested markers are listed in Table 2. It was found that the allele frequencies of Ins/Del alleles and T/C alleles of rs3804100 showed statistically significant associations with AgP, with p values 0.0022 and 0.0018, respectively, although these associations were not statistically significant after correction for multiple testing (Bonferroni’s correction). Because there were only two alleles at each locus, it was difficult to determine which was responsible for the association (for example, whether the Ins allele was protective or the Del allele was susceptible). On the other hand, significant deviations in the genotype distribution were found for Del/Del (OR=2.64, p=0.026), C/C at rs7696323 (OR=0.48, p=0.038), and T/T at rs3804100 (OR=0.44, p=0.028). These data implied that both resistance and susceptibility to AgP might be controlled by TLR2 locus.
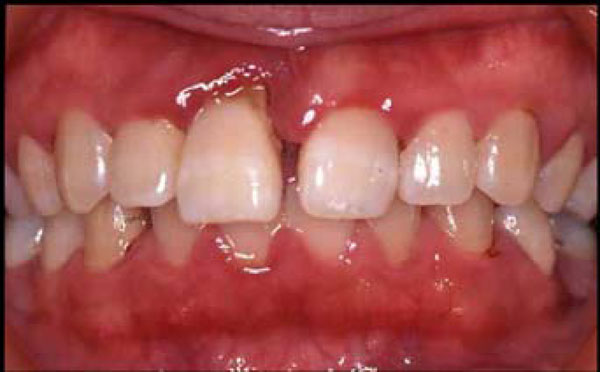
A photograph of teeth from a typical AgP case. The patient was 23 years old. She was reverely affected with swollen gum and almost losing the upper right incisor tooth.
Case-Control Association Study of TLR2 Polymorphisms in Japanese AgP
| Genetic Marker | Patient (n = 38) | Control (n = 190) | OR#4 | 95% CI#5 | p | |
|---|---|---|---|---|---|---|
| -196 to -174 Ins>Del | ||||||
| genotype frequency#1 | Ins/Ins | 12 (31.6%) | 87 (45.8%) | |||
| Ins/Del | 17 (44.7%) | 83 (43.7%) | ||||
| Del/Del | 9 (23.7%) | 20 (10.5%) | 2.64 | 1.09–6.36 | 0.026 | |
| HWE#3 | p = 0.83 | p = 1.00 | ||||
| Allele frequency#2 | Ins | 41 (53.9%) | 257 (67.6%) | 0.56 | 0.34–0.92 | 0.022 |
| Del | 35 (46.1%) | 123 (32.4%) | 1.78 | 1.08–2.94 | ||
| rs7696323 C>T | ||||||
| genotype frequency | C/C | 20 (52.6%) | 133 (70.0%) | 0.48 | 0.23–0.97 | 0.038 |
| C/T | 16 (42.1%) | 49 (25.8%) | ||||
| T/T | 2 (5.3%) | 8 (4.2%) | ||||
| HWE | p = 0.87 | p = 0.46 | ||||
| Allele frequency | C | 56 (73.7%) | 315 (82.9%) | 0.58 | ||
| T | 20 (26.3%) | 65 (17.1%) | 1.73 | |||
| rs3804100 T>C | ||||||
| genotype frequency | T/T | 12 (31.6%) | 97 (51.1%) | 0.44 | 0.21–0.93 | 0.028 |
| T/C | 20 (52.6%) | 78 (41.1%) | ||||
| C/C | 6 (15.8%) | 15 (7.9%) | ||||
| HWE | p = 0.89 | p = 0.99 | ||||
| Allele frequency | T | 44 (57.9%) | 272 (71.6%) | 0.55 | 0.33–0.91 | 0.018 |
| C | 32 (42.1%) | 108 (28.4%) | 1.83 | 1.10–3.04 |
#1, #2 Summation of genotype or allele frequencies at each locus may not be 1.00 because each frequency was rounded.
#3 HWE; Hardy–Weinberg equilibrium.
#4 Odds ratio
#5 95% confidence interval was calculated when p value was less than 0.05.
Because these three polymorphic markers were selected on the basis that they were not in strong LD, we estimated the frequencies of Ins/Del- rs7696323-rs3804100 haplotypes in the patients and controls using Haploview and investigated the association with AgP by the permutation test. The results showed that Ins-C-T haplotype and Del-C-C haplotype were significantly associated with the protection against (p=0.0003) and susceptibility to (p=0.013) AgP, respectively. When we assume multiple testing for the permutation test, p values should be multiplied by the number of presumed haplotypes (n=5). As a result, the association with Del-C-C haplotype did not show statistical significance (Table 3). Therefore, the comprehensive association studies at the allele and genotype level and at the haplotype level suggested a significant association of TLR2 locus with protection against AgP in Japanese.
Permutation Test for Difference in TLR2 Haplotype Frequencies in Ag Patients and Controls
#1 Alleles at Ins/Del-rs7696323-rs3804100 composing of the TLR2 haplotype.
#2 Odds ratio.
#3 Not significant.
#4 P value was corrected for multiple comparison in haplotype frequency (n=5).
DISCUSSION
AgP is a rare serious disease causing tooth loss at a young age. A characteristic of AgP is that it is not secondary to systemic disease accompanied by gum disease. In this study, all 38 AgP patients have no systemic disease, and healthy-donor was selected as the control group. Another characteristic of AgP is the rapid progression of periodontal disease. It has been considered that AgP is caused by the abnormal immune response to periodontopathic bacteria, resulting in the failure to eliminate P.g. bacteria, in the Japanese cases. As P.g. is recognized by TLR2, TLR2 might play an important role in protective response against P.g. infection in AgP. Because TLR2 is polymorphic, some investigators have investigated TLR2 polymorphisms in association with CP and/or AgP, but they could not identify any significant associations [13, 14]. However, previous studies had focused on SNPs with amino acid replacement and several other TLR2 polymorphisms were not tested in the previous studies. These observations prompted us to investigate the association of AgP with TLR2 polymorphisms so far not tested, including the Ins/Del polymorphism in exon 1 which regulates the gene expression [20].
In this study, we revealed for the first time two TLR2 polymorphisms, Ins/Del and rs3804100, which were significantly associated with AgP. A cross-sectional case-control study of polymorphic markers cannot distinguish whether one allele of each polymorphic locus is associated with the susceptibility or whether the other allele of the same locus is associated with the protection or resistance to the disease. To distinguish these possibilities, a prospective cohort study is required. However, a haplotype association study of several polymorphic alleles which were not in strong LD may be useful to evaluate the contribution of markers or their combination in association with the disease. In our study, from the investigation of each polymorphic marker, we can only conclude that TLR2 polymorphism was associated with the susceptibility or resistance to AgP. However, the permutation test for the association with presumed haplotypes revealed that the protection against AgP was associated with a specific TLR2 haplotype, Ins-C-T. The analysis also suggested that none of the associated marker alleles were responsible for the protection, because the frequency of Ins-T-T haplotype was increased in the patients and the frequency of Ins-C-C haplotype was virtually identical in the patients and controls. Therefore, the protection against AgP was assumed to be controlled by an unidentified polymorphism in close LD to the Ins-C-T haplotype. Genetic information on the TLR2 locus might be useful in diagnosis, prevention and treatment of AgP. If the genetic information would be obtained in young age, it might allow preventive strategies and therapeutic intervention for high-susceptibility patients.
In conclusion, we found a significant association of TLR2 polymorphisms and AgP in Japanese. The haplotype analysis suggested that a specific TLR2 haplotype, not the analysed SNPs, conferred protection against AgP. Because this is the first report of an AgP-associated TLR2 haplotype, the association should be validated in other cohorts. Finally, detailed analysis of variations in TLR2 gene including rare alleles is required to further clarify the responsible SNP(s) for protection against AgP. Growing knowledge about the genetic factors that predispose individuals to AgP will hopefully open the novel insights in developing for prediction and prevention of the disease in high-risk individuals.
CONFLICT OF INTEREST
None declared.
ACKNOWLEDGMENTS
This study was supported in part by Grant-in-Aids from the Ministry of Education, Science, Sports, Culture and Technology (MEXT) of Japan, a grant for Japan-Korea collaboration research program from Japan Society for the Promotion of Science (JSPS), and research grants from The Institute of Seizon and Life Sciences. This work was also supported in part by the follow-up grants provided from the Tokyo Medical and Dental University. All authors have no conflicts of interests.


