All published articles of this journal are available on ScienceDirect.
Antifungal Effectiveness between Tricalcium Silicate-White Portland Cements Added Bi2O3 and Mineral Trioxide Aggregate Against Candida albicans
Abstract
Background
Candida albicans is the most dominant fungus found in root canal reinfection cases. This microorganism can withstand extreme pH, low oxygen levels, lack of nutrients, and penetrate the dentinal tubules so that it can resist the intracanal medicament. Root canal cement helps prevent microorganisms and causing root canal reinfection. MTA is one of the root canals cement which is widely used and has an excellent antifungal activity, but it is less beneficial economically. Tricalcium silicate-white Portland cement (WPC) has a similar main composition as MTA, except there is no Bi2O3 content that functions as a radiopacifier.
Objectives:
To fabricate a mixture of tricalcium silicate-WPC with Bi2O3 through a simple solution method and investigate antifungal activity's effectiveness between tricalcium silicate-WPC added Bi2O3 and MTA to C. albicans.
Materials and Methods:
The 80 g of tricalcium silicate-WPC was mixed with 20 g of Bi2O3 through the simple solution method using 99.9% isopropanol as a solution. This sample solution is stirred until homogeneous, then centrifuged. The precipitate was dried until a dry powder was obtained. The powder was analyzed using X-Ray fluorescence spectrometry to identify its chemical composition and concentration. A total of 30 samples were divided into two experimental groups of tricalcium silicate-WPC added Bi2O3 and ProRoot MTA. The vials, which contain cement and C. albicans, respectively, were incubated at 37°C for 24 hours and diluted to obtain a suspension 104, 106 (0.5 in McFarland’s nephelometer) and then inoculated with sterile cotton swabs onto Saboroud Dextrose Agar Plates. The plates were incubated at 37°C for 24 hours. The measurement of colony number of C. albicans was counted by colony counter (CFU/ml).
Results:
The Bi2O3 was revealed in tricalcium silicate-WPC based on XRF characterization, and the antifungal test showed that both materials were effective against C. albicans. There was no statistically significant difference in the number of C. albicans colonies between tricalcium-WPC added Bi2O3 and MTA (p>0.05).
Conclusion:
The mixture of Bi2O3 in tricalcium silicate-WPC was successfully fabricated through a simple solution method, and both samples were effective against the C. albicans.
1. INTRODUCTION
The failure factor in endodontic treatment is caused by persistent microorganisms in the root canal [1-7]. Fungi are microorganisms that can be found in secondary infections with an incidence of between 2% and 40% other than bacteria. The results showed that Candida albicans is the most dominant type of fungi found in endodontic re-treatment [6, 7]. C. albicans is a fungus that can be able to withstand extreme pH, low oxygen levels, and lack of food sources that make microorganisms resistant in the root canal [8]. These microorganisms penetrate the dentinal tubules and interact with other microorganisms to form biofilm complexes and cause reinfection in the root canals [9].
Sealer is an essential material in the stage of filling the root canal as a filling material between guttapercha and the root canal walls and filling gaps and irregularities in the root canal [10, 11]. The ideal cement must-have antimicrobial activity to suppress the remaining microorganisms in the root canal [12-14]. Mahmoud Torabinejad first developed Mineral Trioxide Aggregate (MTA) in 1993 with the main components of tricalcium silicate, tricalcium aluminate, tetra calcium aluminopherite, and Bi2O3. The high alkalinity of MTA can inhibit the growth of microorganisms. Several studies have shown the antibacterial effect of MTA on Enterococcus faecalis and C. albicans [15-20]. MTA is still not widely used because the price is relatively high. Previous research showed the similarity of biocompatibility, sealing ability, and dimensional stability [21-23]. Tricalcium silicate Portland cement produced by Indocement, Ltd., Cirebon, West Java, consists of grey and White Portland Cement (WPC). Tricalcium silicate-WPC has the same composition as MTA, which contains calcium aluminate and calcium sulfate with lower iron content without Bi2O3 [24, 25]. Bismuth oxide (Bi2O3) is a radiopaque material added to a root canal filling material to be seen radiographically. Based on the study results, Guerreiro et al. showed that the addition of Bi2O3 to Portland cement did not change the pH and the antimicrobial effect [26-29].
This study aims to fabricate a mixture of tricalcium silicate-WPC with Bi2O3 through a simple solution method and assess the difference in antifungal activity effectiveness between tricalcium silicate-WPC added Bi2O3 and MTA against C. albicans.
2. MATERIALS AND METHODS
2.1. Synthesis of Tricalcium Silicate-WPC Added Bi2O3
At this stage, the mixing of tricalcium silicate-WPC (Indocement Ltd, Cirebon, Indonesia) was carried out with Bi2O3 as followed: tricalcium silicate-WPC 80 g mixed with Bi2O3 20 g was dissolved using 99.9% isopropanol as a solution. This sample solution is stirred using a magnetic stirrer for 30 minutes until homogeneous, then centrifuged. The precipitate was dried using a vacuum drying oven for 60 minutes until a dry powder was obtained.
2.2. Characterization X-Ray Fluorescence (XRF)
The mixture of tricalcium silicate -WPC added Bi2O3 was analyzed using X-Ray Fluorescence spectrometry (ED-XRF Rigaku Nex CG, The Woodlands, USA) to identify their chemical composition and concentration.
2.3. Antifungal Test
In this study, tricalcium silicate-WPC (Indocement Ltd, Cirebon, Indonesia) added Bi2O3 and ProRoot MTA (Dentsply Sirona, Charlotte, USA) were employed. C. albicans -ATCC 10231 provided by Microbiology Laboratory of Pharmacy School, ITB, Bandung. A total of 30 samples were divided into two experimental groups of tricalcium silicate-WPC added Bi2O3 and ProRoot MTA.
Tricalcium silicate-WPC added Bi2O3 (0.5 g) inserted into a vial of C. albicans suspension (5 ml) and gently shaken to ensure direct contact between fungi and cement as well as ProRoot MTA. The vials were incubated at 37°C for 24 hours and diluted to obtain a suspension 104, 106 (0.5 in McFarland’s nephelometer) and then inoculated with sterile cotton swabs onto Saboroud Dextrose Agar Plates. The plates were incubated at 37°C for 24 hours. The measurement of colony number of C. albicans was counted by colony counter (CFU/ml).
3. RESULTS
3.1. Composition of Tricalcium Silicate-WPC Added Bi2O3
The chemical compositions of the tricalcium silicate-WPC added Bi2O3 by XRF are shown in Table 1. The main chemical compounds of tricalcium silicate-WPC added Bi2O3 were Calcium (Ca) 56.7%, Silica (Si) 14.6%, Bismuth (Bi) 10.6%, Magnesium (Mg) 10.1%, and Aluminum (Al) 5.2% as seen in (Fig. 1). Other minerals were detected under 1% called trace elements. Compared with ProRoot MTA, the amount of Calcium was relatively higher than ProRoot MTA, as well as Aluminum and Magnesium. Tricalcium silicate-WPC added Bi2O3 contained a small amount of silica (Si) than ProRoot MTA. Metal elements were found in both materials, tricalcium silicate-WPC added Bi2O3 and ProRoot MTA at different concentrations. Tricalcium silicate-WPC added Bi2O3 contained a lower amount of iron (Fe) than ProRoot MTA. Zirconium (Zr) was found only in tricalcium silicate-WPC added Bi2O3 at a low concentration.
3.2. Antifungal Activity
Antifungal activity was assessed with the measurement of colony number C. albicans (CFU/ml) used colony counter, as present in Table 2 and the mean number of CFU/ml for each material against C. albicans is shown in Table 3. The growth of C. albicans colonies before and after applying tricalcium silicate-WPC added Bi2O3 and ProRoot MTA incubated for 24 hours is shown in (Figs. 2-4). Among the tested materials, tricalcium silicate-WPC added Bi2O3 and ProRoot MTA present a similar result. There was no statistically significant difference between the fungi count of the Tricalcium silicate-WPC added Bi2O3 and ProRoot MTA after 24 hours incubated since p>0.05, as shown in Table 4.
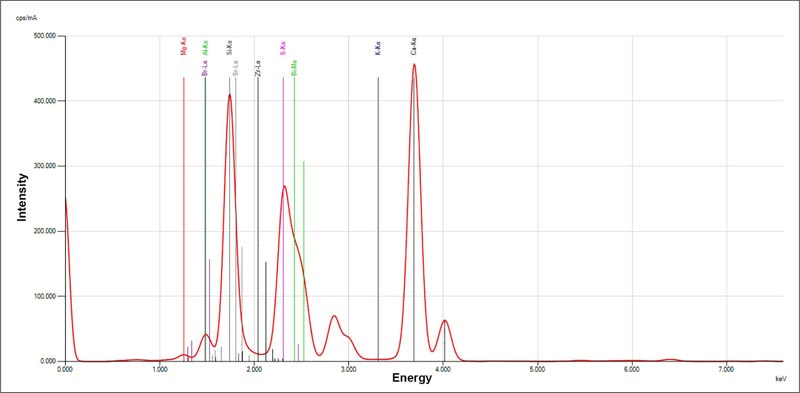
| Minerals | White Portland Cement-Bi2O3 (mass%) | ProRoot MTA[26, 27](mass%) |
| Iron (Fe) | 0.2 | 0.38 |
| Magnesium (Mg) | 10.1 | 5.16 |
| Potassium (K) | 0.3 | 0.45 |
| Calcium (Ca) | 56.7 | 54.6 |
| Titanium (Ti) | 0.05 | 0.09 |
| Chrom (Cr) | 0.003 | 0.003 |
| Aluminum (Al) | 5.2 | 1.19 |
| Zirconium (Zr) | 0.7 | - |
| Sulphur (S) | 1.0 | 1.96 |
| Silica (Si) | 14.6 | 17.6 |
| Bismuth (Bi) | 10.6 | 14.46 |
| Strontium (Sr) | 0.3 | 0.22 |
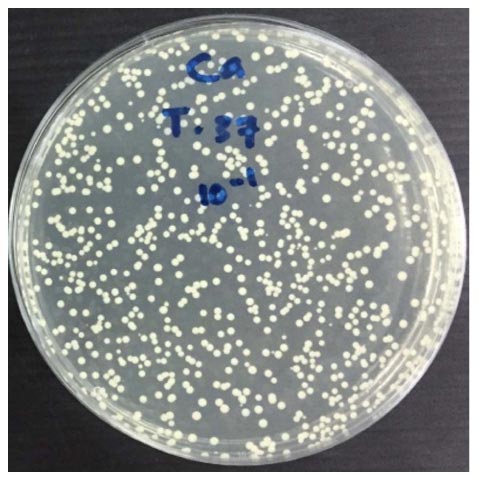
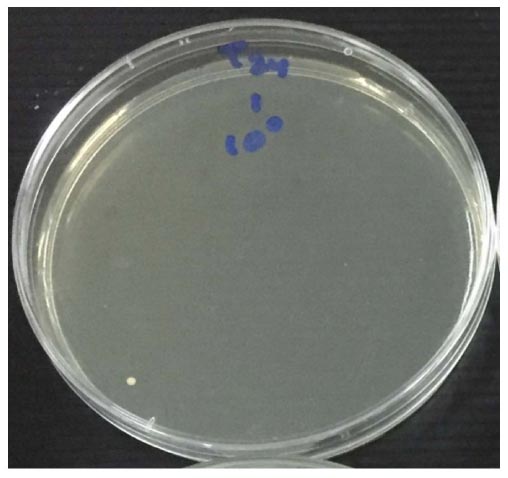
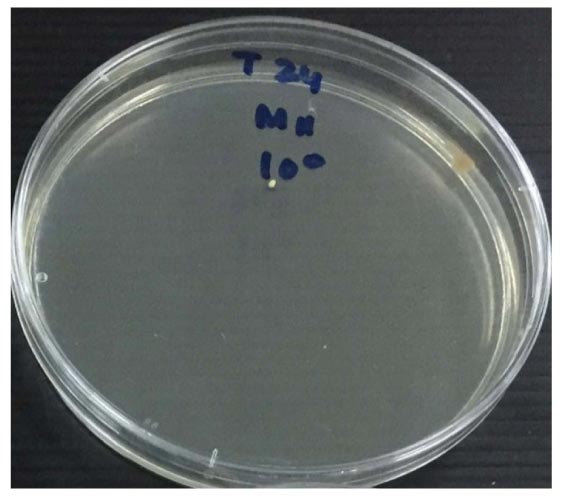
| Tricalcium silicate-WPC added Bi2O3 (CFU/ml) |
ProRoot MTA (CFU/ml) |
| 1 | 2 |
| 1 | 0 |
| 0 | 1 |
| 2 | 1 |
| 2 | 0 |
| 0 | 1 |
| 0 | 1 |
| 0 | 0 |
| 1 | 0 |
| 1 | 1 |
| 0 | 1 |
| 1 | 0 |
| 0 | 1 |
| 0 | 0 |
| 1 | 0 |
| - | Means number of colonies Candida albicans (CFU/ml) |
| Tricalcium silicate-WPC added Bi2O3 | 0.6 |
| ProRoot MTA | 0.6 |
Table 4.
| - | Before 24 h | After 24 h | |
| Tricalcium silicate-WPC added Bi2O3 | ProRoot MTA | ||
| Averages | 88 x 105 | 0.667 | 0.600 |
| Standard deviation | 990.430 | 0.724 | 0.632 |
| n | 15 | 15 | 15 |
| p-value | 0.067* | ||
4. DISCUSSION
Microorganisms are the main factors causing endodontic treatment failure [1-7]. C. albicans are the most dominant fungi found in the root canals and can cause endodontic treatment failures [8, 9]. C. albicans have properties that survive in the root canal after biomechanical preparation and root canal medication [10, 14]. Therefore, the obturation is an essential stage so that a root canal cement with antifungal activity and good sealing ability is needed to prevent microorganisms from entering and causing infection after root canal treatment is completed [15, 16]. MTA is one of the root canals cement widely used and has some ideal properties as a sealer, one of which is an excellent antifungal activity [17-20].
MTA's main content is tricalcium silicate, dicalcium silicate, tricalcium aluminate, and tetra calcium aluminoferrite, also found in Portland cement [20-25]. The similarity of the main content has been studied to provide similar properties in both materials, including the ability of its antifungal activity [26-29]. Tricalcium silicate-WPC also has the same content as MTA [30, 31]. The difference between the two materials lies in the content of Bi2O3 [16, 18, 20]. Bi2O3 is a radiopacifier that can increase radiopacity so that it can be seen radiographically [20, 21]. In this study, Bi2O3 was added to Tricalcium silicate-WPC by 20% according to the amount of Bi2O3 in the MTA. According to Marina et al. (2014) and Paulo (2012), the addition of Bi2O3 to Portland cement has been investigated as a function of a radiopacifier and does not affect pH and the antimicrobial activity which are owned by Portland cement [22, 24]. Guerreiro (2012) also adds the radiopacifiers in Portland cement, which do not change their properties [25].
XRF characterization analysis on tricalcium silicate-WPC added Bi2O3 showed some differences in mineral composition and concentration. The amount of calcium present in tricalcium silicate-WPC added Bi2O3 is higher than MTA, thus maximizing the release of hydroxyl ions obtained when hydration reaction and affect C. albicans. The metal elements are also found in both samples, such as iron (Fe), Magnesium (Mg), Titanium (Ti), Chrom (Cr) and Strontium (Sr) with different concentrations. Tricalcium silicate-WPC added Bi2O3 has a lower iron (Fe) than MTA, minimizing the possibility of staining and toxicity. Iron is one of the heavy metal elements and has an intense color [25, 31, 32]. Tricalcium silicate-WPC added Bi2O3 has magnesium and aluminum higher compared to MTA. Magnesium (Mg) above 6% causes expansion of the material and reduces the risk of shrinkage and a gap formation after setting. Aluminum (Al) affects the setting time of the material. The aluminum found in the Tricalcium silicate-WPC added Bi2O3 is higher than the MTA; this showed that the Tricalcium silicate-WPC added Bi2O3 has a longer setting time than MTA. MTA has less aluminum and more iron (Fe). A decrease in setting time can be achieved by adding iron (Fe) and calcium chloride (CaCl) [14, 19, 36]. Zirconium (Zr) is found only in the Tricalcium silicate-WPC added Bi2O3 and functions as a radioopacifier [32-36].
Dicalcium silicate and Tricalcium silicate contained in Tricalcium silicate-WPC when reacted with water (H2O) then form calcium silicate hydrate (CaO•2SiO2•3H2O) and calcium hydroxide (Ca(OH)2). Calcium hydroxide will break down into calcium ions (Ca2+) and hydroxyl ions (OH-). Hydroxyl (OH-) are active free radicals that can increase the pH level to 12.9 and affect inhibiting the growth of microorganisms [30, 31]. Jafari et al., stated that MTA's antifungal activity comes from Alkaline pH and inhibit the growth of C. albicans [12]. Bhavana et al., also stated that MTA contains calcium oxide, and when in contact with water, it will form calcium hydroxide and increase the pH up from 10.2 to 12.5 within 3 hours [37].
The ability of hydroxyl ions (OH-) inhibits microorganisms' growth through three stages, damaging the cytoplasmic membrane by inhibiting the synthesis of ergosterol found in fungi cell membranes, breaking the DNA chain, and inhibits the process of replication, and causes protein denaturation [38]. Hydroxyl ions are free radicals that can diffuse into the dentinal tubules and cause lethal effects on microorganisms [22, 23, 30, 32, 33]. C. albicans can live in the root canal with an optimal pH of 4.5-8. Environmental pH conditions that rise to alkaline can affect the cell's pH so that pH changes disrupted the activity of ergosterol synthesis enzymes [39, 40]. Hydroxyl ions cause protein denaturation in C. albicans cells. Optimal enzyme activity occurs at controlled pH. Alkalinity induces disrupted ionic bonds, which maintains the protein structure's stability and causes loss of biological activity in enzymes and disruption of cell metabolism [39, 40]. Hydroxyl ions also affected the DNA chain of C. albicans cells. Disrupted DNA chain causes inhibition of the replication of C. albicans cells so that the microorganism can not reproduce itself [39, 40].
CONCLUSION
The mixture of Bi2O3 with tricalcium silicate-WPC could be done through a simple solution method, and there was no statistically significant difference (p>0.05) in the effectiveness of the antifungal activity. In other words, the tricalcium silicate-WPC added Bi2O3, and ProRoot MTA had similar efficacy against the C. albicans.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
No animals/humans were used for studies that are base on this research.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
The data sets analyzed during the current study are available from the corresponding author [A.C] on request.
FUNDING
This research supported and funded by Universitas Padjadjaran with the grant number of 1427/UN6.3.1/LT/2020.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
The authors would like to express special appreciation and thanks to Finders Centre of Excellence, Universitas Padjadjaran, and Integrated Laboratory of Faculty of Dentistry, Universitas Padjadjaran, Bandung, Indonesia.


