All published articles of this journal are available on ScienceDirect.
Effects of Consuming Catha Edulis Forsk (khat) on the Gene Manifestation of CHRM1 and CHRM3 in relation to Salivary Glands, Saliva Flow Rates, pH and Dental Caries in Yemeni Consumers
Abstract
Background:
The current study has been conducted to estimate the impact of Catha Edulis Forsk (simply known as khat) on pH and saliva flow rate in teeth unaffected by caries among patients from Arab, Yemen, consuming the plant. The goal was to evaluate the relation of these indicators with gene manifestation of CHRM1 and CHRM3, which contributes to the regulation of saliva secretions.
Methods:
Overall, 60 respondents aged between 27 and 54 years were selected for the research. They were distributed among two groups, group I and group II, including 30 respondents equally. Group I incorporated respondents consuming no khat, having teeth caries, adequate saliva flow, and generally, healthy oral cavity. In turn, group II incorporated respondents consuming khat having caries-free teeth, inadequate saliva flow and relatively unhealthy oral cavity. Saliva samples were taken from all respondents. They were analyzed for indicators of flow rate as well as pH of saliva. Salivary gland biopsy specimens have also been taken. The whole RNA was allocated, cDNA synthesized and reinforced to estimate the gene manifestation of CHRM1 and CHRM3.
Results:
A considerable rise in the mean salivary flow rate along with a drastic decline in the mean salivary pH within caries-free teeth among the respondents consuming khat was observed during the study. The mRNA expressions CHRM1 and CHRM3 were significantly increased among participants consuming khat. Results were contrasted to the control group of patients not consuming khat.
Conclusion:
The characteristics of salivary flow rate, pH, and caries-free teeth have been related to the gene manifestations of CHRM1 and CHRM3 in respondents consuming khat.
1. INTRODUCTION
Dental caries remain one of the most prevalent chronic diseases affecting the oral cavity of people throughout history [1, 2]. The disease is quite widespread in countries with high industrial potential [3]. The progress of dental caries depends on myriads of inevitable factors, including but not limited to bacteria occupying dental plaque, high-level carbohydrates in the diet, and teeth chronically vulnerable to caries [4]. Caries can develop in all ages, genders, races and socio-economic groups. Oral hygiene and quality of saliva are also essential risk-factors of caries development.
The quality of saliva is highly relevant in supporting oral health. The sum of components in saliva reflects the qualities of lubrication, removal of intruding substances, food digestion, acids/base neutralization, demineralization protection and antimicrobial protection [4-6]. In theory, saliva might prevent dental caries through four potential scenarios: (1) the mechanical purification leading to lower consistency of plaque; (2) the decrease of enamel solubility due to the presence of calcium, phosphate and fluoride; (3) bufferization and neutralization of acids generated by cariogenic agents or delivered through eating habits; and (4) ensuring regular antibacterial activity [5, 6].
Catha Edulis Forskor, commonly known as khat, is a plant referring to the Ceastraceae family. The plant’s leaves and sprouts are chewed to extract chemical agents close to amphetamines in terms of structure and stimulation effects [7, 8]. Some countries from Eastern Africa and the Middle East consume khat as part of their traditions – for example, Saudi Arabia, Yemen, Kenya, Eritrea and others. It is quite ordinary to chew the plant’s leaves at social events. People tend to chew fresh leaves of khat in their mouth with further swallowing the plant’s liquid. The residual components from chewing are usually concentrated in the buccal sulcus in the unilateral or bilateral position; being left in such positions for hours, they still release chemical micro-elements [7, 9]. Khat incorporates stimulants, including Cathinone and Cathine, which affect the consumer’s feelings and cognitive functions.
The extended chewing of khat, however, is associated with a harmful impact on health. The special impact from khat is caused on the brain that is stimulated to increasingly release dopamine, which is a neurotransmitter bringing the feeling of satisfaction and euphoria. Moreover, khat is responsible for actively releasing norepinephrine (stress hormone) making the consumer extremely alert and vigilant [7, 10]. Khat’s adverse effects on health, aside from the brain, also include oral and dental issues. Early studies have revealed that chewing khat might possibly provoke xerostomia (extreme dryness of the oral cavity) because of the amphetamine-like impact of khat that formally exhausts the salivary glands under active chewing [8]. It has been found that Yemeni patients consuming and chewing khat demonstrate high levels of saliva release in their mouth. However, the impact of khat on the salivary glands remains unclear. Therefore, the current study focuses on analyzing khat’s impact on the salivary glands in terms of four certain factors: caries-free teeth, saliva flow rate, saliva pH, and the gene manifestation of CHRM1 and CHRM3 in salivary glands, where both genes technically manage the glandular secretions (including salivary glands) reacting to any parasympathetic stimulation.
2. MATERIALS AND METHODS
2.1. Respondents
The study was conducted by initially involving 74 Yemeni participants (men only) aged between 27 and 54 from 4 private clinics, Riyadh, Saudi Arabia. Their medical histories have been evaluated via self-reports to collect demographic data and personal medical characteristics. All subjects have provided informed consent.
2.2. Exclusion Criteria
In total,14 participants have been excluded from the project. Criteria for exclusion for this study included extended history of smoking, presence of chronic diseases, sialadenitis (sialoadenitis), xerostomia and intake of any agent affecting the level of saliva secretion (namely anticholinergic, antihypertensive, antihistamine, antidepressant, antipsychotic, antiemetic, antispasmotic, and anti-parkinsonian agents).
2.3. Inclusion Criteria
Eventually,60 respondents aged between 27and 54 years were approved for the study.
2.4. Study Design and Clinical Dental Analysis
Following the clinical tests, respondents were distributed in two separate groups. The first group (control group) included respondents who consumed no khat, demonstrated a normal quantity of saliva flow and had a healthy oral cavity (overall, 30 patients). The second group included respondents who consumed khat, demonstrated excessive quantity of saliva flow and had an unhealthy oral cavity (overall, 30 patients) (Table 1, Fig. 1, A and B).
| Covariate | Group I | Group II |
|---|---|---|
| No. of participated patients(%) | 30 of 60 (48.83%) | 30 of 60 (51.16%) |
| Age at diagnosis | ||
| ˂ 40 years (%) | 23 (76.7%) | 19 (63.33%) |
| ≥ 40 years (%) | 7 (23.3%) | 11 (36.66%) |
| Length of hours’ sleep (mean ± SD) | 9 ± 2.64 | 5.4 ± 1.78 |
| Abuse of Khat history (years) | 0 | 7.3 ± 3.26 |
| Frequency of consuming khat/ week (mean of days ± SD) | 0 | 5.5 ± 2.14 |
| Length time of consuming khat/ day (mean of hours ± SD) | 0 | 3.7 ± 2.03 |
| Oral hygiene status (%) | ||
| Good | 26(86.66%) | 12 (40%) |
| Poor | 4 (13.33%) | 18 (60%) |
| Dental health quality (%) | ||
| Good | 19 (63.33%) | 23 (76.66%) |
| Poor | 11 (36.66%) | 7 (23.33%) |
| Gingival health quality (%) | ||
| Good | 30 (100%) | 3 (10%) |
| Poor | - | 27 (90%) |
| Teeth mobility frequency (%) | - | 27 (90%) |
| Missing teeth frequency (%) | 8 (26.66%) | 26 (86.66%) |
2.5. Saliva Collection
10 milliliters of clean, unaffected saliva were taken for analysis. Saliva collection took place in daytime between 10 AM and 12 PM, two hours since the respondents having their breakfast. Prior to taking samples, the respondents had to wash their oral cavities with distilled water after eating. Afterwards, the respondents were placed in a comfortable position within a well ventilated and properly illuminated room. They were asked to expectorate saliva, which was immediately taken within 3 minutes and put in a preliminary-weighed collecting tube. The collection of samples was continued until the tubes were filled with 10 mL of saliva.
2.6. Saliva Analysis
All the samples gathered were evaluated for pH and flow rate potential. The salivary flow rate was assessed by recording the amount of saliva taken within the first 3 minutes of sample collection [5, 6]. The special tool named “Saliva-Check Buffer Testing Mat” (developed by the Japanese company GC Dental Products Corporation) has been utilized to evaluate pH level. For analysis, evaluation of the gene manifestation for the salivary glands was also conducted, implementing collected minor biopsies from the selected salivary glands.
2.7. The Effect of Khat on Gene Manifestation of CHRM1 and CHRM3in the Salivary Glands
2.7.1. Minor Salivary Glands Biopsies
A dissection was made from the lower lip. The purpose was to get a minor biopsy from labial submucosal salivary glands. Biopsy samples used for analyzing gene manifestation have been snap-frozen and held in liquid nitrogen until the start of the RNA extraction. The procedure was undertaken with informed consent from all 60 respondents.
2.7.2. RNA Extraction
Homogenization of tissue samples (10 mg) was performed with the help of a sterilized manual homogenizer (Brinkmann, Westbury, NY). The whole RNA has been successfully derived from the snap-frozen salivary glands utilizing an RNeasyMinikit (manufacturer from Norway), in accordance with original instructions for isolating RNA from tissue [11]. RNA extracted was supported at –80°C before microarray evaluation was initiated.
2.7.3. Synthesis of Fluorescent Complementary DNA (cDNA) from Messenger RNA (mRNA)
In terms of synthesis, a low-level RNA–input fluorescent linear amplification kit (Agilent Technologies, Palo Alto, CA) was exploited in the study to produce fluorescent cDNA to be further utilized in cDNA microarrays from Agilent Technologies. It has been performed in compliance with the developer’s recommendations (Low RNA Input Fluorescent Linear Amplification Kit Protocol version 1.1; March 2003) and following several modifications as proposed [11]. The RNA samples have been accumulated by lyophilization utilizing a vacuum in a rotary at a temperature of 4 °C. After that, the lyophilized RNA samples were re-suspended in 10 µl of Tris–EDTA buffer. Both quality and quantity of RNA samples were calculated with the help of Thermo Scientific™ NanoDrop 2000. For the last cDNA evaluation, 200 ng of RNA customized toward 13.2 µl/sample with nuclease-free H2O was utilized. Fluorescent cDNA was ultimately engineered from the clean and reinforced RNA as mentioned in the manufacturer’s instructions. Cleaning of the marked cDNA was conducted by applying a QIAquick PCR purification kit (Qiagen).
2.7.4. Quantitative RT-PCR
A sum of 100 ng of messenger RNA (mRNA) was eventually reverse-transcribed to complementary DNA (cDNA). The procedure was performed using the reverse transcription reagent kit (manufacturer from the Netherlands). The finished cDNA was reinforced by quantitative real-time PCR applying the recommended Light Cycler system in accordance with the manufacturer’s protocol (Roche Diagnostics). All the reactions took place in a 20μl volume with 0.5 μl of primers, 2 μl of Light Cycler Fast Start reaction mix SYBR Green I (Roche Diagnostics) and normal concentrations of MgCl2. Patterns of PCR primers are demonstrated in Table 2.
| Gene name | GenBank ID | G Genesymbol | Gene Group/function |
|---|---|---|---|
| Cholinergic receptor muscarinic 1 or muscarinic acetylcholine receptor M1 |
M35128 | CHRM1 | Coordinateglandular secretions in salivary glands and other body systems |
| Cholinergic receptor muscarinic 3 or muscarinic acetylcholine receptor M3 |
X15266 | CHRM3 | |
| Glyceraldehyde-3-phosphate dehydrogenase | NM002046 | GAPDH | glycolytic enzyme in cytoplasm (housekeeping gene) |
PCR lasted for 10 minutes at a temperature of 95°C, followed by forty 3-second cycles at a temperature of 95°C, 3 seconds at 55°C, and 10 seconds at 72°C. Gene manifestation was stabilized through HPRT utilized in the process as a housekeeping gene.
2.7.5. Statistical Analysis
The outcomes of the study were statistically analyzed. The mean numbers for flow rate, pH and gene manifestations for two groups of respondents were calculated and evaluated by applying SigmaStat software version 8 (Systatsoftware, San Jose,CA, USA). The outcomes derived from quantitative tests were presented as mean ± SEM. p values < 0.05 were confirmed as statistically valid.
3. RESULTS
3.1. Saliva Flow Rate
Among the two groups under study, there was a considerable distinction in the mean salivary flow rate (P < 0.001). Group II demonstrated an essentially increased mean salivary flow rate as compared to Group I (Fig. 2, a).
3.2. SalivaryPH
Quite predictably, the flow rate caused an impact on salivary pH due to their close interrelation. Therefore, a considerable distinction in the mean salivary pH among the Groups studied was found (P < 0.001). Group I demonstrated an essentially reduced mean salivary pH as compared to Group II (Fig. 2, b).
3.3. The Effect of Gene Manifestation of CHRM1 and CHRM3 in the Salivary Glands
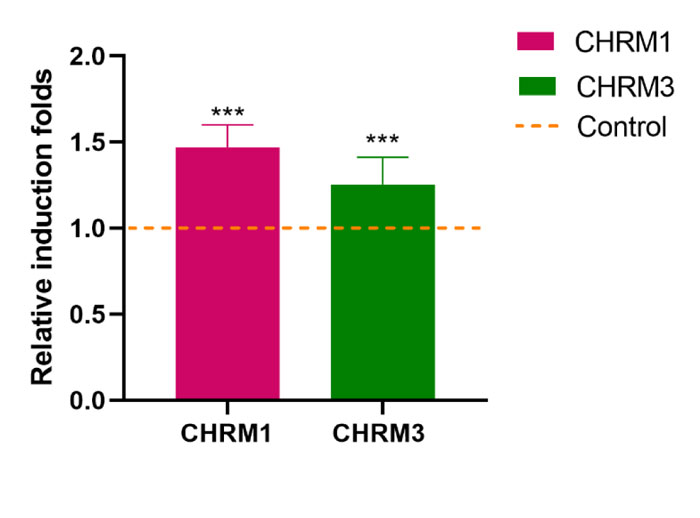
The manifestation of the CHRM1 gene was 1.49 folds relevant (P˂0.001) and greater in consumers of khat contrasted to patients consuming no khat. The plant’s impact was further validated by the manifestation of the CHRM3 gene, demonstrating a differential expression which was 1.25folds relevant (P˂0.001) and higher among participants consuming khat contrasted to patients not consuming khat (Fig. 3).

4. DISCUSSION
The current research is a pioneer in analyzing gene manifestation profiles in relation to the salivary glands of individuals consuming khat with focusing on two relevant and interrelated variables, such as salivary flow rate and salivary pH.
Khat incorporates a diversity of elements, which is why, chewing the plant causes a variety of effects. Mainly, the gastrointestinal system as well as the nervous system, are affected. Constipation, retention of urine and acute cardiovascular complications are viewed here as autonomic (peripheral) effects related to the nervous system; elevated vigilance, dependence, tolerance and psychiatric manifestations are considered effects related to the central nervous system [8, 10-13].
Being a neurosubstance, khat is regarded as a stimulant that might enhance physical and cognitive functionality, increase reactions and performance capacities [9, 12]. Night shift employees tend to chew khat to remain alert, keep awake and fight fatigue. Similarly, students consume khat to prepare for exams and increase cognitive functions. Yemeni khat consumers accept the fact that khat’s benefits outweigh minor challenges and discomfort with health, including headaches, colds, spontaneous pain, fever, arthritis and sometimes depression [13-16].
In the meantime, teeth caries are a highly prevalent chronic oral illness in humans . It is developed at all ages, in all genders, races and socioeconomic groups. Oral hygiene and quality of saliva also contribute to caries development [6]. Although dental science has made significant progress in recent decades, caries remain a popular health issue around the world. Children are especially exposed to caries. Teeth caries represent an infectious bacterial biofilm illness that mainly develops in a pathologic oral setting.This disease has often been considered multifactorial due to its ability to develop as a result of dietary choices and individual factors of the host’s health [7, 17]. Moreover, there are multiple research and clinical evidence confirming saliva’s value in preventing dental caries. Saliva’s defensive effect is secured due to the cleaning cycle inside the mouth, buffering, antimicrobial activity, and calcium and phosphate transportation for remineralization [5]. The relationship between defensive and pathologic variables in saliva and plaque biofilm, including the harmony between the cariogenic and non-cariogenic microbial organisms presented in human saliva, eventually defines how and when caries may develop [3-5, 15]. Principal variables of saliva’s quality that are associated with caries usually include the aciduric/acidogenic bacteria and rates of acid generation. In this sense, variables affecting caries development are, first, the quantity of saliva released in a selected time and, secondly, its acidneutralizing capacity [6, 7, 16].
The findings derived from the salivary pH collected have matched evidence and outcomes from other studies completed by Sardana et al. [14], and Widowatiet al [15]. Another research completed by Zhou et al. [17], demonstrated that the salivary pH derived from children having caries in the early years of life was statistically larger compared to children who did not have caries.
The findings derived from the salivary flow rate analysis have been found interconnected to evidence on the salivary pH in the current study. Our findings have also been correlated to findings from studies performed by Preethi et al. [18], and Prabhakar and Dodawad [19]. On the other hand, the outcomes presented in their research projects were not found to be statistically valid. The salivary flow rate was decreased insignificantly among children diagnosed with caries contrasted to children unaffected by caries. In turn, a study completed by Thaweboon et al. [20], indicated that the mean values for the salivary flow rate were found to be common between unaffected and highly affected children. The salivary flow rate made no impact on the progress of heavy caries, which can be explained by other external variables, including age, oral hygiene, general health, and genetic history.
Hence, supportive molecular experiments were undertaken in this research to add accuracy to the findings. Two relevant genes CHRM1 and CHRM3 were chosen based on the link between their inherent functions and the current research purpose.
The gene manifestations based on RT-PCR demonstrated that mRNA levels of two selected genes CHRM1 and CHRM3 have been associated with regulating the acinar cells responsible for the intense release of saliva in the salivary glands. CHRM1and CHRM3 gene manifestation levels were found to be highly expressive in respondents consuming khat in contrast to respondents not consuming khat.
In addition, primary saliva fluids are secreted in the acinar cells first. A special functional mechanism of regulation takes place to secrete the saliva fluid. It starts from the parasympathetic outflow and then results in the secretion of acetylcholine (ACh) onto M1 and M3 muscarinic receptors. Hence, several effects are observable: a) acinar cells accelerate the release of saliva; b) co-transmitters bring intense blood flow to the salivary glands; and c) contraction of myoepithelium happens, increasing the rates of saliva extrusion [21-23].
Following this logic, khat, as a neuro substance, initiates parasympathetic stimulation leading to an active acetylcholine (neurotransmitter) which then connects M1 and M3 receptors together on acinar cells within the salivary glands, which further trigger the intensification of the salivary flow, making saliva more watery in terms of texture .
Findings from this study derived from the gene manifestation were correlated to a study performed by Gautam et al. [24, 25], which support the idea that a mix between M1 and M3 receptors ensures cholinergic activation of salivary flow, also increasing pH and reducing risks of caries development.
The surplus of saliva accumulated in the mouth, on the other hand, provokes other health risks. Among the threats, there are lung problems, stomach infections, and tumors. Above-the-neck complications include oral infections, strep throat inflammation and tonsil infections, as well as sinusitis, which ultimately affect the swallowing process [16].
CONCLUSION
Concluding all ideas, the evaluation of variance confirmed that people with low risks of caries development deal with higher salivary flow and pH value because of chewing khat in contrast to individuals at a high risk of having caries, as they have low salivary flow and pH. In this sense, khat stimulates parasympathetic system activators, leading to increase in gene manifestation levels of CHRM1 and CHRM3. That, in turn, elevates the salivary flow rates via the effects of M1 and M3 receptors on acinar cells located in the salivary glands. In the meantime, elevated salivary flow stimulates the increase in salivary pH 7.5, thereby, decreasing the risk of caries development yet increasing other health risks and concerns.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was approved by the Ethics Committee of Princess Nourah Bint Abdul Rahman University, Saudi Arabia (IRB Registration Number with KACST, KSA: H-01-R-059)
HUMAN AND ANIMAL RIGHTS
No animals were used in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
All subjects have provided informed consent.
AVAILABILITY OF DATA AND MATERIALS
The data supporting the findings of the article are available within the article.
FUNDING
This research was funded by the Deanship of Scientific Research at Princess Nourah Bint Abdulrahman University through the Fast-track Research Funding Program.
CONFLICT OF INTEREST
The author declares no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
This research was funded by the Deanship of Scientific Research at Princess NourahBint Abdulrahman University through the Fast-track Research Funding Program.


