All published articles of this journal are available on ScienceDirect.
Resin Infiltration into Differentially Extended Experimental Carious Lesions
Abstract
Resin infiltration of initial caries lesions is a novel method of caries therapy. However, it has some limitations. Therefore, further experimental studies are needed to improve resin infiltration. It was the aim of this investigation to study resin infiltra-tion into different experimental carious lesions. Caries-free extracted human molars and premolars were demineralized for 3, 6, 9 and 12 days and infiltrated with resin. Prior to infiltration, the teeth were incubated with sodium fluorescein. After em-bedding, serial sections were cut through the experimental lesions, and the penetration of the resin was measured with fluo-rescence microscopy. Two infiltrated teeth from each time interval were not embedded and cut. Infiltration of the resin was then studied with EDS element analysis. The results showed that with increasing demineralization time, the lesion expansion was also increasing, and the resin infiltration was always almost complete. From these results it can be concluded that artifi-cial standardized caries-like lesions are suitable for experimental studies of resin infiltration.
INTRODUCTION
In modern operative dentistry, there is a focus on noninvasive treatment of initial caries lesions. For decades, attempts have been made to infiltrate initial caries lesions with resin to stop further caries progression instead of excavating those lesions and placing fillings [1-5]. During the past decade, a very promising resin has been developed that is now in clinical use [1, 6-15]. Infiltration of this resin into natural initial carious lesions has been studied, and it was found that those lesions are permeated more or less completely [8, 11, 15-17]. A disadvantage of resin infiltration is the need for surface conditioning of the initial lesion [15, 18, 19]. Surface conditioning with 15% hydrochloric acid is necessary because the hypermineralized surface layer of natural initial caries lesions hampers resin penetration into the demineralized body of the lesion. A comparative study showed that phosphoric acid is less effective than hydrochloric acid and results in less complete infiltration [20]. Another problem of the resin is its extreme hydrophobicity. Therefore, the resin has to be applied in absolutely dry conditions. Wet conditions within the body of the lesion also hamper resin infiltration. This makes the use of a rubber dam necessary.
Further experimental studies will be needed to optimize resins and their infiltration behavior. It was therefore the aim of this investigation to study the infiltration of resin into experimental caries lesions. Artificial caries-like lesions have the advantage that they can be produced under standardized reproducible conditions. Thus, experiments with resins can be carried out under standard conditions.
MATERIALS AND METHODS
Thirty-two caries-free premolars and third molars were randomly divided into four groups of eight teeth each and were used for this investigation. The extracted teeth were anonymized and the patients gave their informed verbal consent to use the teeth for this in vitro study. The protocol conformed to the principles outlined in the Central German Ethics Committee's statement (2003) focusing on the use of human body material in medical research and was approved by the Ethical Committee of Witten/Herdecke Universtiy (116/2013). Exclusion criteria for the use of teeth were initial subsurface caries lesions. They were stored in 0.9% NaCl containing 0.1% thymol. The crowns were covered with dental wax leaving a 4x4 mm window on the buccal and oral surface. The teeth in the different groups were then demineralized in 1.6% hydroxyethylcellulose for 3, 6, 9 and 12 days at pH 4.8. The pH was adjusted with acetic acid. As infiltration resin, Icon® (Dental Materials Company, DMG, Hamburg, Germany) was used. After demineralization, the buccal windows of six teeth out of each group were treated with the resin as follows. The oral window remained untreated as a control:
1. Application of Icon-Etch (15% hydochloric acid) for 2 min.
2. Wash out for 30 s with distilled water.
3. Imbibition of the whole tooth in 1% aqueous fluorescein solutions.
4. Wash out in distilled water for 5 minutes.
5. Air drying.
6. Application of Icon-Dry (99% ethanol) for 30 s.
7. Intensive air drying to simulate rubber dam.
8. Application of Icon-Infiltrant (dimethacrylate) for 3 min followed by 40 s light curing.
9. Application of Icon-Infiltrant for 1 min followed by 40 s light curing.
10. Wash out unbound fluorescein for 10 min in water.
11. Finally, the teeth were stored in a moist chamber at 37°.
Dental wax was removed, and the roots of the teeth were then embedded into Technovit 3040 (Heraeus-Kulzer, Wehrheim, Germany) to achieve a base for cutting serial sections of 80 μm thickness through the lesions with a saw microtome (Leica 1600, Leica). Three sections in the center of each lesion were selected for further evaluation.
Two teeth of each group were treated with Icon® without imbibition in the fluorescein solution. These teeth were cut horizontally through the middle of the experimental lesion into two halves and the lesions were investigated directly with SEM and EDX analysis.
POLARIZED LIGHT MICROSCOPY
The lesions were investigated using polarized light microscopy (PLM) with the optical path from the bottom to identify the morphology of the caries lesions; fluorescence microscopy, with the optical path through the objective and from the top, was used to visualize the infiltration of the resin by the trapped fluorescein. Finally, simultaneous microscopy with both optical paths was used to combine the morphology of the lesions with the infiltration depths of the resin. The extension of the demineralized zone and the fluorescent area was measured quantitatively and the data compared statistically. 10 single measurements of the extension of the demineralized zone were determined in each section.
SCANNING ELECTRON MICROSCOPY AND EDS ANALYSIS
The same sections and the directly treated, sectioned two teeth of each group underwent a scanning electron microscopic investigation combined with quantitative EDS element analysis. The content of Ca2+ and C4+ was determined using line scans and EDS element mapping. Scanning electron microscopy was carried out using a Zeiss Sigma VP scanning electron microscope in the variable pressure mode at 30 Pascal pressure, with 20 kV acceleration voltage and an AsB backscattered electron detector. EDS spectroscopy was performed with an EDAX Apollo XL system with an active area of 30 mm2 and the Team V3.3 software. The reading of the line scans was carried out with a dwell time of 25 msec and an amplification time of 12.8 µsec with a distance between each reading point of 1 µm. The EDS mappings were measured with a dwell time of 200 µsec per reading point and a frame resolution of 1024x800 pix. A total number of 256 frames were recorded resulting in a scan time of approximately 13 hours.
RESULTS
Quantitative measurement of the extension of the experimental demineralization revealed that the lesion depth increased slightly from 3 to 12 days of demineralization. However, the increase was not significant (Fig. 1). Polarized light microscopy and fluorescence microscopy showed in all investigated sections that the size of the lesions and the resin infiltration was identical (Fig. 2). The range of the lesion depth and the infiltration of the resin into the lesion were between 23.47 (±10.20) µm after 3 days demineralization and 40.81 (±10.11) µm after 12 days demineralization. EDS element mappings and the respective line scans demonstrated an irregular infiltration of the resin into the lesions (Fig. 3). This irregular infiltration corresponded with differences in the pore volume in the body of the lesion, which could be noticed in PLM. All lesions were completely infiltrated.

Extension of the lesions after demineralization of various time intervals.
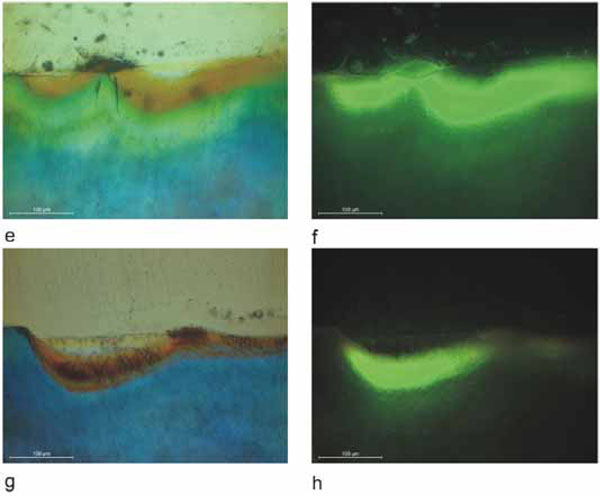
PLM and fluorescence microphotographs of the lesions after different demineralization times and resin infiltration. After three days of demineralization, a) and b), a rather faint fluorescence could be detected. After six days of demineralization, c) and d), intense fluorescence in the whole demineralized zone was observed. After nine days of demineralization, e) and f), very strong fluorescence of the whole demineralized zone was observed. After twelve days of demineralization, g) and h), again, very strong fluorescence in the whole demineralized zone was observed.
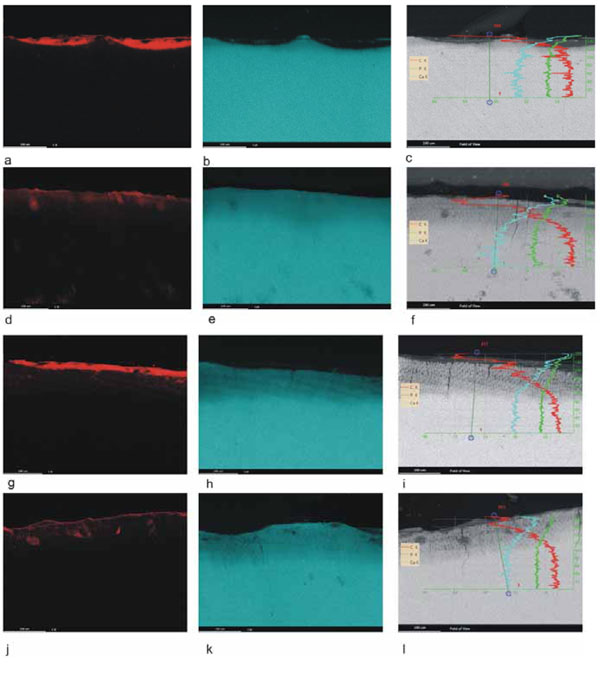
Element mapping of carbon (red) and calcium (blue) and line scans of the lesions after different demineralization times and resin infiltration. a) through c) show the results after three days of demineralization. a) There is a resin deposit on top of the lesion but almost no carbon signal within the demineralized enamel. b) The calcium map shows a very distinct demineralization of the enamel surface. c) The line scan demonstrates a thin demineralized surface zone with decreasing carbon content from the surface to the bottom of the lesion. d) through f) show the results after six days of demineralization. d) Carbon infiltration into the lesion can be seen but is not strong. f) The line scan shows carbon infiltration into the whole lesion. g) through i) show the results after nine days of demineralization. g) On top of the lesion there is a resin deposit. The carbon signal is weak but can be detected in the entire lesion. h) The calcium map shows a more extended demineralized zone. i) The line scan demonstrates a homogenous linear infiltration of the lesion with abrupt carbon decrease at the bottom of the lesion. j) through l) show the results after twelve days of demineralization. j) There is a very clear carbon signal in the whole demineralized area. k) The calcium map demonstrates an extended demineralized enamel surface. l) The line scan shows complete carbon infiltration into the lesion with decreasing concentration with increasing mineralization.
There was a clear discrepancy between the results of fluorescence microscopy and the element mappings and line scans. Fluorescence microscopy always demonstrated more intensive and complete infiltration whereas element mapping was very discrete. The line scans showed a clear distribution of the carbon infiltration into the lesion.
DISCUSSION
A number of studies have been performed on the infiltration of resin into natural caries lesions [7, 9-11, 13, 15-18, 20, 21]. Only a few studies used experimental caries lesions [5, 22, 23] but for different research purposes. For a standardization of infiltration experiments, reproducible artificial lesions are beneficial. Therefore, in the present study, the infiltration of resin into artificial caries-like lesions was studied under standardized experimental conditions.
There is still a debate whether large natural initial caries lesions are infiltrated completely [7, 16, 17, 20]. Complete infiltration of the lesion is important for the stability of the initial lesion and it should prevent further caries progression [10]. It is dependent upon several factors, such as surface porosity, porosity of the body of the lesion, dryness of the lesion and capillary forces [14]. Surface porosity is achieved by conditioning with 15% hydrochloric acid [18, 19]. However, this destroys the surface integrity of the enamel [24]. Porosity of the body of the lesion is dependent on the caries activity and stage of demineralization. Infiltration into active lesions is deeper than infiltration into inactive lesions [20]. The degree of porosity decreases from the surface towards the enamel-dentin junction. Therefore, the deeper areas of the lesion may not be penetrated with resin. Furthermore, the infiltration into natural caries lesions appears to be inhomogeneous depending on irregularities of the porous volume of the body of the lesion [7, 8, 11, 13, 15, 16, 20].
Artificial caries-like lesions are homogenous demineralized areas of enamel. Varying depth can be achieved by different demineralization times. However, prolonged demineralization periods result in surface destruction of the enamel. Therefore, artificial demineralization is limited and the artificial lesions are more shallow than natural lesions. In this investigation, surface destruction was observed after twelve days demineralization. All artificial lesions were completely penetrated by the resin, which could be shown with fluorescence microscopy. These results are in accordance with other studies on natural caries lesions [7, 8, 13, 15-17, 20]. Reproducibility is a great advantage of artificial lesions, as a variety of experiments can be carried out with the same type of lesion.
As for chemical restrictions, it is not possible to stain the resin directly with dyes. Indirect methods have to be used for studying infiltration into enamel lesions. Usually the teeth are stained with fluorescent dyes [10, 11, 13, 15-17, 20, 25] and then infiltrated with the resin which traps the dye in the demineralized enamel pores. However, the fluorescent dye could penetrate much deeper into the lesion than the resin itself. The use of EDS element analysis may be a more precise method to study resin infiltration [16]. A limitation of this method is the sensitivity. Line scans of the sectioned lesions appeared to be the most promising method to detect resin infiltration.
EDS analysis is a powerful method to study resin infiltration [16]. Using the line scan method, the infiltration of the whole lesion could be demonstrated. It could be shown that the artificial lesions were infiltrated completely. In early lesions, the penetration decreased from the surface to the bottom of the lesion indicating an increasing pore volume towards the bottom of the lesion. The EDS mapping also demonstrated infiltration of the lesions but with rather weak signals for carbon. The sensitivity of the EDS mapping may be the limitation of this this method.
In conclusion, in this study it could be demonstrated that artificial caries-like lesions can be used, within the limits of the shallow artificial lesions, to perform experimental studies on resin infiltration into lesions.
CLINICAL RELEVANCE
Minimally invasive treatment of initial caries lesions comprises oral hygiene instructions, fluoridation and resin infiltration. Resin infiltration has been developed during the past decade, and numerous studies investigated resin infiltration into natural initial caries lesions. However, there are some limitations of this technique that are surface conditioning, porosity of the lesion and the extreme hydrophobicity of the resin. For further optimization of resin development and infiltration studies, standardized experiments are necessary. Therefore, we investigated resin infiltration into artificial caries-like lesions in human premolars and molars under standardized conditions using polarized light microscopy, fluorescence microscopy and SEM-EDX element analysis.
AUTHORS' CONTRIBUTIONS
WHA wrote the manuscript and was responsible for the project planning
LB carried out EDS analysis
KB carried out the morphometric measurements
EAN calculated the statistics
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
The authors would like to thank Mrs. Susanne Haussmann for her technical support regarding the SEM specimen preparation and cutting the teeth.


