Gingival Biotype Assessement: Visual Inspection Relevance And Maxillary Versus Mandibular Comparison
Abstract
Objective:
This clinical study was aimed at evaluating the accuracy of gingival visual inspection procedures during clinical examination and determining whether differences existed between the maxillary and mandibular gingival biotypes.
Materials and Methods:
The study included 53 patients and 124 clinicians. The clinicians were asked to assign to each subject, using photographic documents, one of three biotypes: thin-scalloped, thick-scalloped, or thick-flat gingival biotype. A total of 19716 responses were collected for statistical analysis.
Results:
Identification accuracy of the gingival biotype and the intra-examiner repeatability presented poor highlighting of the limited relevance of visual inspection. In addition, the percent of agreement between classifications based on the global view of both the maxilla and mandible and the classification based on the individual mandibular or maxillary anterior teeth was not statistically significant.
Conclusion:
Based on the above results, it can be concluded that a simple visual inspection is not effective for the identification of gingival biotype. Furthermore, evidence suggests that a difference of biotype between the maxilla and the mandible in the same patient is conceivable. Therefore, orthodontic clinical examination should incorporate a reproducible method of determining the individualized gingival biotype for each group of teeth that will be moved.
INTRODUCTION
Reduced gingival thickness is one of the factors that can cause periodontal attachment loss and marginal tissue recession in a patient [1]. This is particularly true during orthodontic treatment [2]. The clinical careful examination of gingival biotype helps practitioners to make appropriate decisions concerning the degree of incisor inclination [3] or intrusion [4]. Nevertheless, clinicians face difficulty associated with the correct identification and categorizing of the patient’s gingival biotype; this is owing to the several classifications that have been established, though with none serving as a reference. Indeed, these classifications depend on numerous observations and measurements, such as the height of keratinized tissue, the bucco-lingual thickness of the gingival [5], or the alveolar process thickness. Other classifications refer to the dental form, gingival thickness, and the height of the gingival [6]. The multiplicity and variability of these parameters are the source of some confusion. Thus, orthodontists require more relevant, reproducible, sensitive, predictive, and easy-to-use classification methods for detecting and diagnosing high risk patients in terms of marginal tissular recession prior to orthodontic treatment.
Recently, De Rouck et al., in 2009, revisited the subject of gingival biotype and developed for simple visual inspection a new method for the classification of gingival biotype based on the following four clinical parameters: crown width/crown length ratio, gingival height, papilla height, and gingival thickness. This classification is based only on a maxillary observation regardless of the mandibular parameters and the following biotypes were identified: thin-scalloped, thick-scalloped, and thick-flat scalloped gingival biotype [7].
On the other hand, it is observed that no distinction has been made between the maxilla and mandible within any of the existing classifications of gingival biotype. It is thus clear that in classifications it is considered that the gingival biotype is unique for a patient. However, to our knowledge, no study has provided a comparison of the biotype of the maxilla and the mandible in order to prove this assertion. Nevertheless, the risk for the development of dehiscence is more important during the labial movement of the mandibular incisors [8]. Interestingly, studies on humans [9] and dogs [10] have shown that the gingival thickness varies according to the dental arch, gender, and age.
This study was aimed at evaluating the accuracy of a simple visual inspection based on the De Rouck classification and determining whether differences existed between the maxillary and mandibular gingival biotypes.
MATERIALS AND METHODS
Subjects
A total of 53 subjects (25 females and 28 males with a mean age of 17.5 years) were included in the present study. Among these, 36 were volunteer dental students from the Nantes dental school, and 17 were patients receiving clinical orthodontic treatment at Nantes hospital. All subjects provided informed consent for participating in the present study.
The inclusion criteria were as follows:
- Subjects presenting all maxillary and mandibular incisors and canines;
- Subjects having good oral hygiene without any clinical signs of gingival inflammation or attachment loss (periodontal probing does not exceed 3mm).
The exclusion criteria were as follows:
- Subjects with crown restorations or fillings that involved the incisal edge on the anterior teeth;
- Pregnant or lactating women;
- Subjects taking medication with any known effect on periodontal soft tissues.
Clinical Parameters
The gingival biotype for each of the subjects was determined by two experienced clinicians following the method described by De Rouck et al. These clinicians had acquired training in periodontology (LF) and orthodontia (ML). The classification done by the clinicians was used as the gold standard.
The following four clinical parameters were recorded by the two examiners (Fig. 1) and were used to establish the gold standard as described by De Rouck et al. [7]:
- Using a caliper, the crown width/crown length ratio was measured for the two maxillary and mandibular central incisors according to the procedure described by Olsson and Lindhe [11]. The crown length was measured between the incisal edge of the crown and the free gingival margin or, if discernible, the cement enamel junction. The length of the crown was divided into three portions of equal height. The crown width, i.e., the distance between the proximal tooth surfaces, was recorded at the border between the middle and the cervical portion.
- The gingiva height was measured using a periodontal probe (PCP UNC 15 probe, Hu Friedy, Chicago, IL). This parameter was defined as the distance from the free gingival margin to the mucogingival junction.
- The papilla height was assessed using the same periodontal probe at the mesial and distal aspect of the four central incisors. This parameter was defined by Olsson et al. [11] as the distance from the top of the papilla to a line connected to the midfacial soft tissue margin of the adjacent teeth.
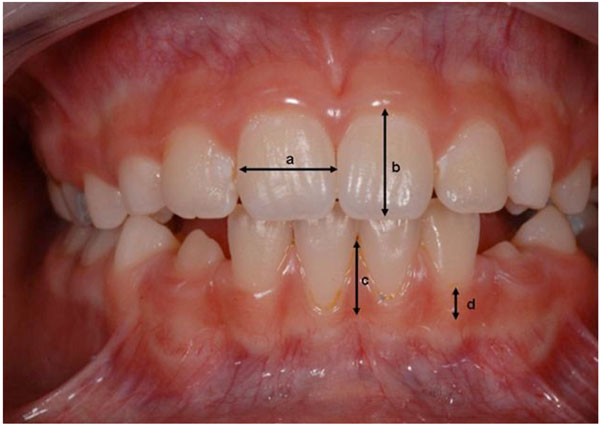
Quantitative measurements made by the referenced clinicians: (a) crown width, (b) crown length, (c) papilla height, (d) gingival height.
De Rouck etal. [7] had identified three gingival biotypes (Fig. 2a, b ,c):
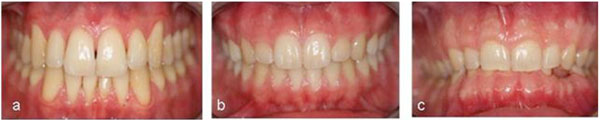
(2a). Cluster A1, (b) Cluster A2, (c) Cluster B.
- Cluster A1: thin-scalloped gingival biotype (ThinS) with a slender tooth form;
- Cluster A2: thick-scalloped gingival biotype (TS) with a slender tooth form;
- Cluster B: thick-flat gingival biotype (TF) with a quadratic tooth form.
Finally, three clinical pictures were taken for each patient using a Nikon D60 in macro mode with flash and a magnification of 0.5 (a total of 159 pictures). The first picture focused on the front maxillary and mandibular teeth in occlusion (Fig. 3a), the second focused on the maxillary anterior teeth only (Fig. 3b), and the last picture was of the mandibular anterior teeth only (Fig. 3c).
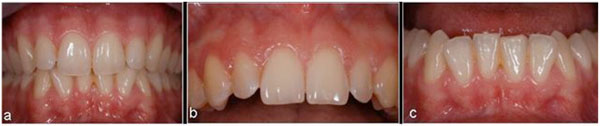
Pictures of the teeth of subjects for diagnosis of gingival biotype by participating clinicians: (a) front views of the occlusion of the maxilla and mandible, (b) the maxillary anterior teeth only, (c) the mandibular anterior teeth only.
After collecting all these quantitative and qualitative data, the experienced clinicians (ML and LF) together identified for each subject the gingival biotype according to De Rouck methodology [12].
Visual Inspection by Clinical Staff
As it is not realistic to conduct a direct clinical examination of the 53 subjects by 124 examiners, all clinicians were asked to identify the gingival biotype in the same 53 subjects based on visual inspection alone. Among the practitioners, nine were specialists in orthodontia (group C) and nine were residents in dental surgery (group I). Moreover, 48 were dental students in the final year of the graduate program (group T), and the remaining 58 were undergraduate dental students (group D).
Scoring
The examiners were installed in a dark room. Explanations of the cluster analysis determined by De Rouck et al. were provided by a periodontist (AS). The classifications were discussed with a focus on the specificities of each cluster using unambiguous clinical pictures from the paper by De Rouck et al. The clinicians were presented with 159 slides assigned in random order (three clinical slides of each subject: the front view of the occlusion for maxillary and mandible, the maxillary anterior teeth, and the mandibular anterior teeth) on a large screen, with each slide projected for eight seconds.
All clinicians were invited to attend a class for an update on gingival biotypes. The clinicians were asked to assign one of the three possible biotypes to each case.
A total of 19716 responses were collected; this significant range of data was entered for statistical analysis.
STATISTICAL ANALYSIS
Accuracy in Identifying the Gingival Biotype
The accuracy in identifying the gingival biotype was evaluated for the occlusion, the maxilla, and the mandible as compared to the gold standard.
For each clinician, the accuracy in identifying the gingival biotype was assessed using percent of cases correctly identified. The overall average percent of cases correctly identified and the average percent of cases correctly identified by type of clinicians were estimated with a 95% confidence interval. The overall average percent of cases correctly identified in each was tested using a mixed linear model with a clinician random effect. The p-values were adjusted with the Tukey procedure.
The accuracy in identifying each of the three gingival biotypes was detailed (ThinS/ TS/ TF).
Agreement Between Occlusion, Maxilla, and Mandible
For each clinician, the percent of agreement and the kappa statistics were determined. The overall average percent of agreement and the average percent of agreement by type of clinicians were estimated with a 95% confidence interval. The overall agreement was evaluated using the mean of kappa statistics with a 95% confidence interval.
RESULTS
The results of the gold standard (ML and LF) were as follows:
- Cluster A1 thin-scalloped gingival biotype: 24.5%;
- Cluster A2 thick-scalloped gingival biotype: 56.6%;
- Cluster B thick-flat gingival biotype: 18.8%.
In order to evaluate the reliability of the simple visual inspection, a calculation was made as to how many cases had been correctly identified for the gingival biotype by each clinician in various situations (in occlusion of both maxilla and mandible, maxillary teeth only, or mandibular teeth only) according to the gold standard classification.
ACCURACY IN IDENTIFYING THE GINGIVAL BIOTYPE IN OCCLUSION
Table 1 shows the average percent of cases in which the 124 clinicians correctly identified the gingival biotype in occlusion view compared to the gold standard classification. No significant differences were observed among the responses of the four groups of clinicians. The percent of agreement was only 36.68% (CI at 95%: 35.43% - 37.93%), which is indicative of slight to fair precision.
ACCURACY IN IDENTIFYING THE GINGIVAL BIOTYPE IN MAXILLARY AND MANDIBULAR VIEW
Table 1 shows the average percent of cases in which the 124 clinicians correctly identified the gingival biotype in maxillary and mandibular view compared to the gold standard classification. The results were 41.10% (CI: 39.86% - 42.34%) for maxilla and 35.58% (CI: 34.54% - 36.62%) for mandible.
Fig. (4) resumes the results of Table 1 and shows that there is a significant difference (p<.0001) between the classification made in maxillary view and occlusion or mandibular classification. However there are no significant differences between occlusion and mandibular results.
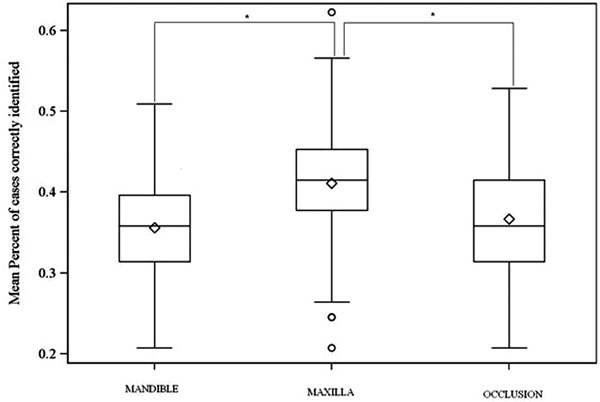
Boxplot illustrating the mean percent of cases correctly identified for mandible, maxilla and occlusion (* P-Value< 0.0001).
Average Percent of Cases Correctly Identified for each group of Clinicians and in Different Situations (Pictures in Occlusion, Maxilla and Mandible View)
| Occlusion | Maxilla | Mandible | |
|---|---|---|---|
| C (N=9) | 34.25% [31.2% - 37.48%] | 46.75% [43.14% - 50.36%] | 37.27% [32.83% - 41.72%] |
| D (N=58) | 38.19% [36.17% - 40.21%] | 41.30% [39.63% - 42.96%] | 37.43% [35.92% - 38.94%] |
| I (N=9) | 37.50% [32.57% - 42.43%] | 31.12% [25.38% - 36.85%] | 31.12% [25.38% - 36.85%] |
| T (N=48) | 35.15% [33.28% - 37.02%] | 41.67% [39.82% - 43.53%] | 33.86% [32.47% - 35.25%] |
| Total (N=124) | 36.68% [35.43% - 37.93%] | 41.10% [39.86% - 42.34%] | 35.58% [34.54% - 36.62%] |
REPEATABILITY OF EACH CLINICIAN IN GINGIVAL BIOTYPE IDENTIFICATION
To evaluate the number of cases in which the most clinicians identified the same gingival biotype, we compared in one hand the front view in occlusion with the maxillary anterior teeth of the same patient, and in another hand the front view in occlusion with the mandibular anterior teeth and, finally, we compared the results between the maxillary and mandibular anterior teeth. The results are shown in Table 2.
Repeatability of each Clinician in Gingival Biotype Identification: for each Clinician the Percent of Agreement Between his own Responses in Occlusion, Maxilla and Mandible was Calculated
| Occlusion Versus Maxilla | Occlusion Versus Mandible | Maxilla Versus Mandible | |
|---|---|---|---|
| Percent of agreement [IC 95%] (N=124) | 48.68% [47.18%- 50.18%] | 43.54% [42.23%-44.85%] | 44.97%[41.94%- 47.99%] |
| Kappa value [IC 95%] | 0.2102 [0.1870- 0.2334] | 0.1392 [0.1195- 0.1589] | 0.1695 [0.1244- 0.2146] |
The percent of agreement for each case was about 48.68%, 43.54%, and 44.97%, respectively, with kappa values 0.2102 (CI at 95%: 0.1870 - 0.2334), 0.1392 (CI at 95%: 0.1195 - 0.1589), 0.1695 (CI at 95%: 0.1244-0.2146). These results are indicative of poor reliable responses.
CLUSTER IDENTIFICATION IN DIFFERENT VIEW
Table 3 shows the average percent of cases in which each cluster (ThinS, TS, and TF) were correctly identified in occlusion, maxilla, and mandible.
Average Percent of Cases in which each Cluster Thin-Scalloped, Thick-Scalloped and Thick-flat (i.e.ThinS, TS, TF) were Correctly Identified in Occlusion, Maxilla and Mandible
| Total (N=124) | Occlusion | Maxilla | Mandible | |
|---|---|---|---|---|
| ThinS | 33.19% [30.22% - 36.16%] | 33.33% [30.56% - 36.11%] | 34.77% [31.94% - 37.59%] | |
| TS | 38.21% [36.34% - 40.07%] | 40.64% [38.46% - 42.82%] | 39.35% [36.87% - 41.84%] | |
| TF | 36.88% [34.92% - 38.85%] | 44.87% [42.41% - 47.34%] | 32.65% [30.33% - 34.96%] |
The thick-scalloped biotype was the best identified in occlusion (38.21%) and in mandible (39.35%). In maxillary view, the clinicians better classified the thick-flat biotype (44.87%). The most difficult biotype to recognize was the thin-scalloped.
DISCUSSION
The results indicate that a simple visual inspection is not effective for identifying gingival biotype, which is not a real surprise. Further, the results suggest that the biotype might be different between the maxilla and mandible in the same patient.
This study was aimed first at determining whether the experience of the clinicians could influence the ability to identify among different biotype clusters. As shown in Table 1, no difference was observed in the identification ability of the clinicians. These results are in agreement with the results obtained by Eghbali et al., who found no significant differences among the four groups of clinicians in their study [12]. Thus, it can be noted that the experience of the clinician did not make a difference in identifying the gingival biotype.
These results suggest that a simple visual inspection is not an accurate method for the identification of the gingival biotype; indeed, the probability for identifying the correct cluster is less than 50%. These results are once again consistent with the results described by Eghbali et al.
Interestingly, it was found that the thin-scalloped biotype was the most difficult to identify by visual inspection. This is significant because this biotype is the most at risk to gingival recession [13, 14]. Thus, it is clearly suggested that to prevent gingival recessions, the orthodontist must not be satisfied with the visual inspection, and must resort to other complementary methods. Several studies have attempted to refine the analysis of gingival biotype [1, 14] using cone beam computed tomography (CBCT) as a novel method for the measurement of gingival thickness. This quantitative technique allows the visualization and measurement of the dimensions and the analysis of the relationship of several structures of the periodontium and dentogingival attachment apparatus. Eger et al. (1996) applied an ultrasonic device for measuring gingival thickness and for defining three gingival phenotypes. In the present study, Eger et al. (1996) [6] found that the characteristics of the maxillary front teeth could be extrapolated to other parts of the masticatory mucosa, in particular the palatal mucosa.
These two methods have not been adapted for ordinary practice; however, they suggest that additional examination is necessary for correct identification of the gingival biotype. Using a probe test to complement a visual inspection, similar to that of Kan et al. (2010), seems to be a reliable and objective method for the evaluation of the gingival biotype, and further studies might be necessary for improving the use of this probe test [15].
Regarding the differences between the maxillary and mandibular gingival biotypes, the present study demonstrates that the thick-flat biotype is most frequently identified in the maxilla, while it is less frequently identified in the mandible (44.87% versus 32.65%), suggesting either greater difficulty in classifying biotype in the mandible or existence of a biotype difference between maxilla and mandible in the same patient. To date, all gingival classifications use the maxillary reference for determining the gingival biotype for both dental arches; however, this method is not discriminating. In fact, it has been shown in the present study that the results of agreement seem more relevant for identifying gingival biotype in maxilla than in the mandible. These results suggest that the classification of gingival biotype must be revisited once again, introducing a mandibular reference.
In 2005, Vandana and Savitha used transgingival probing for demonstrating that the gingiva was thicker in the mandibular arch as compared to the maxillary arch [9]. This is in contrast with the study conducted by Müller et al. (2000), in which a thicker gingiva was found in maxilla and the thinnest facial gingiva was found in the maxillary canines and first mandibular premolars [13]. Moreover, in a study of temperature patterns of the gingival [16], Holthuis and Chebib (1983) showed temperature differences between the arches (the mandible versus the maxilla) and regions (the posterior versus the anterior). On the other hand, it was indicated that the blood flow to mandibular gingiva in healthy and inflamed sites is statistically different, whereas the blood flow to the maxillary gingiva in healthy and inflamed sites is not statistically different in maxilla [17]. All of these results reinforce the present hypothesis and indicate that a difference should be observed between the gingival biotypes in the maxilla and mandible. In addition, individualizing the gingival biotype for each of the arches is seemingly a new outcome that needs further investigation. Moreover, as shown by Olsson and Lindhe, varying form of the teeth is associated with the variation in the gingival biotypes [18]. Further studies with larger cohorts are needed to confirm these results.
CONCLUSION
A precise and careful examination of the gingival biotype is necessary to guide treatment and monitoring of the patient during dental treatment.
Within the limits of the present study, it can be concluded that a visual inspection is not an accurate method for gingival biotype diagnosis. It is clear that an assessment of the gingival biotype should incorporate an easy and reproducible method for discriminating a thin gingiva from a thick one.
Thus, according to the results obtained in this study, the gingival biotype should be individualized to a group of teeth or even a tooth. Such an approach is appropriate for orthodontic practice. In fact, this approach is aimed at moving teeth, sometimes one tooth and at other times a group of teeth. The mechanism suitable for each case is determined; however, it seems that individualized data on the tooth gingival biotype could help to better control the side effects of their treatment.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflicts of interest.
ACKNOWLEDGEMENT
Declared none.


