Fibroblastic Variant of Osteosarcoma: A Challenge in Diagnosis & Management
Abstract
Osteosarcoma of the jaws is a relative rare malignant bone tumor. Like, its counterpart in the long bones, osteosarcoma affecting the head and neck region shows distinct yet diverse clinical, histologic and prognostic characteristics. Its diagnosis is a challenge to histopathologists and is especially important in early stages to improve its prognosis. In the initial phase, it may present as nondescript swelling with an indolent growth rate, only to become overtly aggressive and malignant towards the later phase of the disease. This article reports on a case of an advanced osteosarcoma of the maxilla in a 42 year old woman who came for the evaluation of a swelling. The case was submitted for surgical intervention and was followed by one recurrence till the time of reporting. This case illustrates that immunohistochemical staining of Vimentin, S-100 and CD 68 markers are useful to confirm the histologic diagnosis of osteosarcoma, along with radiographic evaluation using CT scan and 3D imaging.
INTRODUCTION
Although, the incidence of osteosarcoma (OS) among malignant tumors is low, it is still the most common primary tumor of bone [1]. It belongs to a family of lesions which have considerable diversity in histologic features and grades [2]. OS arising from the jaw comprises 2.1% of all malignant oral and maxillofacial tumours [3]. OS of jaw differs from OS of the long bones in its biological behavior, presenting a lower incidence of metastasis [4] and a better prognosis with approximately 40% 5-year survival rate as compared to 20% for non-jaw lesions [5].
In the initial phase of the disease, OS may present as an unremarkable slowly progressing bony swelling, only to become overtly aggressive and malignant towards the latter phase of the disease, thus imposing a challenge to accurate diagnosis [1]. Histologically, OS is classified into three subtypes: osteoblastic, chondroblastic, fibroblastic [6]. However, it may have various degrees of differentiation and produce various kinds of extracellular matrix, thus producing a histologic pattern that may vary significantly, not only from one case to another, but also from area to area within the same case. OS are believed to arise from immature bone-forming cells or through neoplastic differentiation of other immature mesenchymal cells into osteoblasts. Three main factors generally are purposed to be etiologically significant in their development -irradiation, preexisting benign bone disorders and trauma [4]. OS tumorigenesis has been linked to alterations in several genes. The first association of OS with an inherited predisposition was observed in patients with bilateral retinoblastoma. This association was confirmed by identification of the retinoblastoma susceptibility gene (RB1) on human chromosome 13 which showed high percentage of mutations in OS [7]. The second gene associated with OS was the p53 gene where the mutations in the p53 gene were first observed in sporadic OS [8, 9].
Here, we present a rare case report of OS in a 42 year old female patient, who had a swelling in the maxillary region with varied histologic appearance mimicking proliferative reactive lesions, thus posing a challenge to pathologic diagnosis and effective surgical management of the case.
CASE REPORT
A 42 year old female patient presented with a solitary swelling in the left middle-third of the face. The swelling had been present for a month. Extraorally, the swelling measured around 2x2cms (Fig. 1A). It was firm in consistency, tender on palpation and the overlying skin appeared stretched and shiny. There was associated lymphadenopathy. Intraorally, the swelling extended anterioposteriorly from distal aspect of 23 to distal aspect of 27 (Fig. 1B). Lateral extension revealed the obliteration of the buccal vestibule, medially up to the midpalatal area. A root stump was present in relation to upper left first premolar. Investigations like intra oral periapical radiograph showed periapical rarifying osteitis with increase in the width of periodontal ligament membrane in relation to 26 (Fig. 1C). Orthopantamograph revealed a root stump in relation to 24 and increase width of periodontal ligament membrane with loss of lamina dura in relation to 26 (Fig. 1D). Considering the findings, a provisional diagnosis of infected periapical pathology in relation to 26 was given. Patient was advised extraction of the root stump and endodontic therapy for 26. Following treatment, patient reported back to the hospital within two weeks with significant increase in the size of the swelling from 2x2 cms at the first visit to 6x5 cms (Fig. 2A). On examination, a foul smelling, reddish black mass measuring 6x5x6 cms, pedunculated swelling was seen arising from the extracted socket of 24 (Fig. 2B). Palpatory findings revealed the swelling to be firm in consistency, tender on palpation and slight bleeding on probing. These features suggested a provisional diagnosis of central giant cell granuloma. Additional investigations were done that included hematological investigation which revealed increase in blood glucose and ESR levels. Radiographic investigations of paranasal sinus showed haziness of the left maxillary sinus, with erosion of infra orbital, medial and lateral walls. Computed tomography revealed a large expansile lesion arising from the lateral wall of the superior alveolus of the maxilla of the left side, extending into the left maxillary sinus and eroding all the walls of the maxillary sinus (Fig. 2D). CT morphology suggested the possibility of a giant cell tumor.
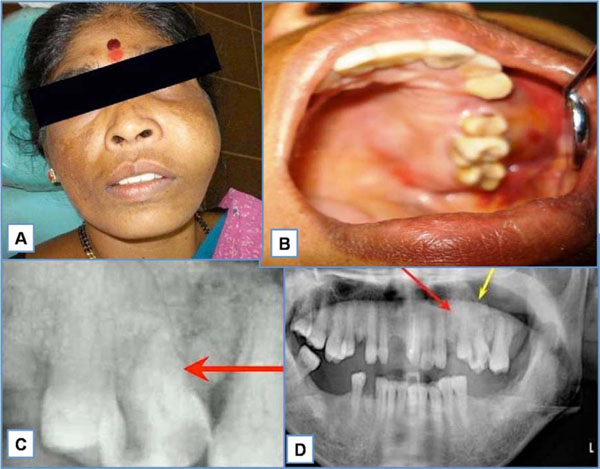
A. Extra oral swelling measuring 2x2 cms in the left middle third of the face. B. Swelling extending anterioposteriorly from the distal aspect of 23 to distal aspect of 27. C. Intraoral periapical radiograph showing periapical rarifying osteitis with increase in width of periodontal ligament membrane in relation to 26. D. Orthopentamograph showing root stump in relation to 24 and increase width of periodontal ligament membrane with loss of lamina dura in relation to 26.
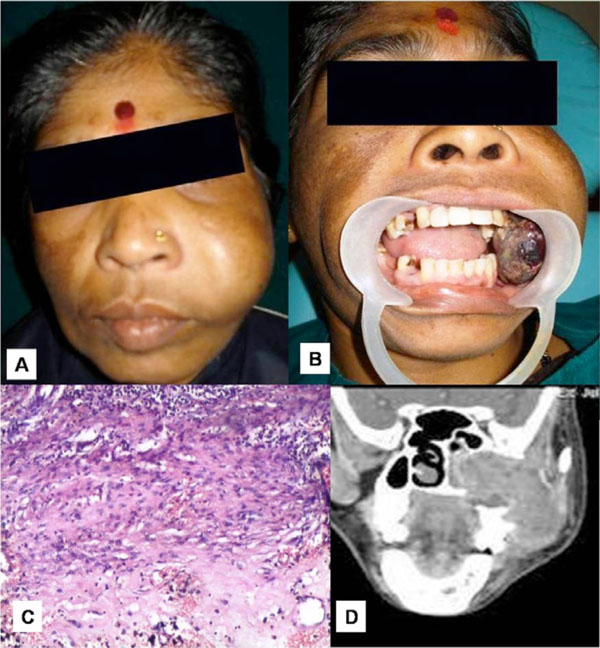
A. Swelling measuring 6x5 cms. B. Reddish black pedunculated mass measuring 6x5x6 cms from the extracted tooth socket 24. C. Histopathology of incisional biopsy showed a non specific infection. D. Computed tomography showing the extension of the swelling into the left maxillary sinus & causing erosion of all the walls.
An incisional biopsy was performed and histopathological findings showed a nonspecific infection suggesting an inaccurate biopsy site (Fig. 2C). A repeat biopsy was requested in relation to deeper areas. However, patient reported back within two days of incisional biopsy with a further increase in the size of the swelling with the lesion protruding out of the oral cavity causing inability to close the mouth (Fig. 3A). An immediate partial maxillectomy was done based on the clinical features, computed tomography findings, to avoid further encroachment of the base of the skull (Fig. 3B). The resected mass weighed about 250gms, measuring 14x8 cms (Fig. 3C). Histopathology showed the lesion to be composed of proliferating fibroblasts along with bundles of collagen fibers. A markedly cellular lesion comprising of mature proliferating fibroblasts with infrequent mitosis and occasional atypia was seen (Fig. 4A). There was a presence of abundant inflammatory cells along with few reactionary giant cells, interspersed with a myxoid background (Fig. 4B). Storiform pattern of arrangement of fibroblasts was also seen, with the fibroblasts showing atypia in relation to size and shape. Invasion into the adjacent adipose tissue was also noted. Immunohistochemical staining was performed with vimentin markers and S-100 protein. Vimentin positivity proved the myofibroblastic nature of the spindle cell lesion (Fig. 4C). S-100 was negative, thus ruling out neural tumours (Fig. 4D). Based on the clinical picture, rate of growth of the lesion and histopathological findings, a final diagnosis of fibrosarcoma was given. Patient reported back with an uneventful healing following which functional and esthetic reconstruction was done by giving an obturator. After hemimaxillectomy, chemotherapy was administered which included Cysplatin 80 mg/m2 on day 1 along with 5 Fluorouracil - 500mg/m2 which was administered on day 1 to 4. This resulted in partial response of the tumour with stoppage of bleeding. Patient also underwent a course of radiotherapy (60 Gy over 6 weeks). The decision to give radiotherapy was made taking into consideration the large size of the lesion along with a history of recurrence. After surgery, an acrylic plate obturator was placed which was to be replaced by a cast partial hollow bulb closed obturator.
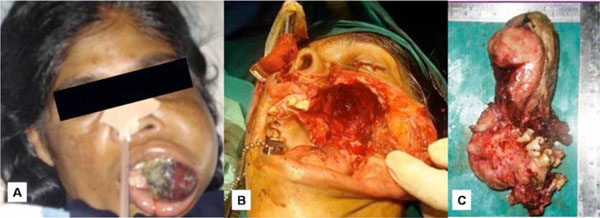
A. Further increase in the size of the swelling causing inability in closing the mouth. B. Immediate partial maxillectomy being carried out. C. Complete resected mass measured 14x8 cms.
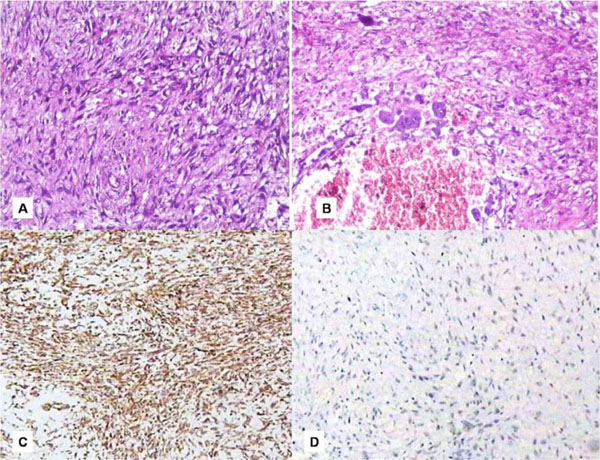
A. Histopathology showed a markedly cellular lesion with proliferating fibroblasts. B. Presence of few reactionary giant cells. C. IHC showing positivity to Vimentin marker. D. Immunohistochemistry with S-100 marker being negative.
There was no evidence of recurrence for six months after which patient reported back with a complaint of pain in the same region with difficulty in eating and carrying out routine oral hygiene procedures. Extra orally a diffuse solitary swelling measuring 2x2 cms was seen in the left middle-third of the face with associated lymphadenopathy. Intra orally, the swelling was measuring 2x3 cms in the region of earlier maxillectomy (Fig. 5A). CT scan was performed for the same, and it revealed a soft tissue density present heterogeneously in the region of left pterygopalatine fossa, extending superiorly and medially into the maxillary sinus and there was loss of fat plane with lateral pterygoid muscle. With 3D imaging, erosion of floor of the left orbit, left medial and lateral pterygoid plates were evident, suggesting a residual lesion (Fig. 5B).
Elaborate hematological investigations with electrolyte levels were done which revealed an increase in lymphocyte, ESR rate and hemoglobin level. The duration of the lesion with clinical behavior suggested possibilities of giant cell reparative granuloma, chronic fungal or bacterial infections, fibrosarcoma and osteosarcoma.
Treatment rendered was immediate resection to prevent the encroachment of the mass. The resected soft tissue mass was irregular in shape, grayish white in colour, measuring 4x3 cms with white solid areas seen on the cut surface. Histopathology revealed storiform pattern of arrangement of fibroblasts interspersed with atypical spindle cell to round to oval cells (Fig. 5C). Faint eosinophillic areas representing tumour osteoid was seen within the tumour mass. Osteoclasts like giant cells were seen in close association to tumour osteoid (Fig. 5D). Few areas suggested histiocytic type cells admixed with atypical fibroblasts and mitotic figures were also seen (Fig. 5E). The entire lesion varied in histological appearance from area to area. An immunohistochemical study was also carried out with CD68 to know the nature of histiocytes, but its focal positivity ruled out the malignant fibrous histiocytoma variant (Fig. 5F). Proliferating fibroblasts along with the typical tumour osteoid was more favoring a sarcomatous lesion like OS. A final diagnosis of fibroblastic variant of OS was given. Alveolar bone graft with osseointegrated implants has been considered on this patient at a later date.
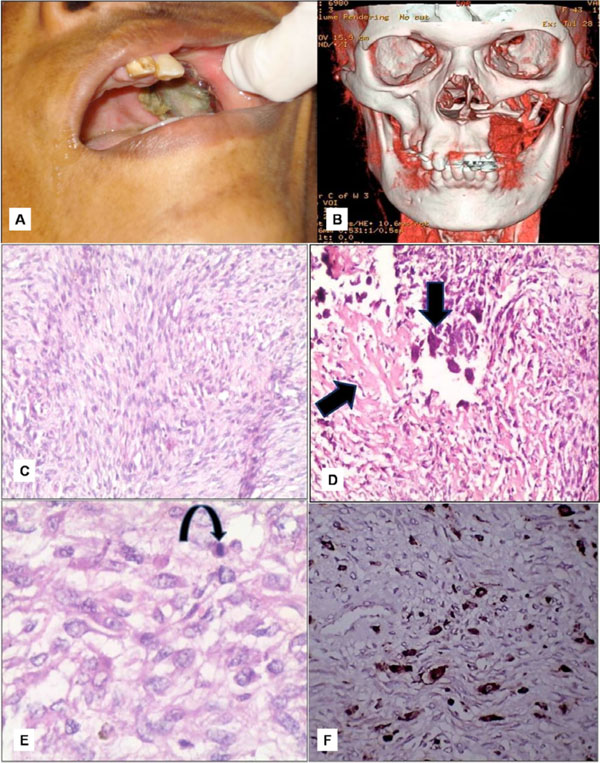
A. Recurrent lesion measuring 2x3 cms in the maxillectomy region. B. 3D-imaging showing erosion of the floor of the left orbit. C. Storiform pattern of arrangement of fibroblasts with atypical; spindle cells. D. Osteoclasts like giant cells closely associated with tumour osteoid. E. Histiocytic type of cells admixed with atypical fibroblasts and atypical mitotic figures. F. Immunohistochemical CD68 marker study showing focal positivity for histiocytes.
DISCUSSION
OS are primarily bone producing malignant tumours arising most frequently in the long bones. Jaw OS usually occur one to two decades later than that of OS of other regions. Patients with OS usually have nonspecific clinical symptoms, the most common of which is pain associated with the swelling presenting from weeks or months. It has a bimodal age distribution, with a major peak in the second decade and a somewhat smaller peak after the age of 50. However, bimodal distribution of jaw lesions differ from those in the apppendicular skeleton, with the first peak occurring some-what later, in the third decade [10]. The present case, with a critical clinical appearance of pain and swelling, is in accordance with the literature related to jaw OS, where patient presents with clinical symptoms in 3rd and 4th decade of life. These neoplasms often show typical behavior in the initial phase presenting as a nondescript swelling, only to become overly aggressive in the later phase of the disease [11]. This was seen in our case as well, where the initial swelling was an unremarkable swelling leading to a preliminary diagnosis of periapical pathology. However, the swelling became significantly aggressive within a short span of time which led to emergency management of the lesion.
Histologically, although OS of jaws are almost similar to that of long bones, they are always better differentiated than the latter [12]. It has been reported that production of osteoid by malignant cells, even in small amounts, is diagnostic of OS [13, 14]. Depending on the relative amounts of osteoid, cartilage, or collagen fibres present in the extracellular matrix, OS are categorized histopathologically into osteoblastic, chondroblastic, or fibroblastic subtypes [6]. In reality, most OS exhibit varying amounts of these three cell types and matrix [1]. Therefore, division into any one of these types is arbitrary and is generally meant to signify greater than 50% prevalence of any of these histologic types [1]. OS are known to vary in histologic pattern within same case with areas showing storiform pattern of fibrosarcoma to atypical elongated histiocytic like cells representing histiocytic tumours. Storiform pattern of fibroblasts with proliferating fibroblasts along with bundles of collagen fibers led to the diagnosis of fibrosarcoma in the initial biopsy in the present case. Further, there was a predominance of spindle cell proliferation in sweeping fascicles along with infrequent mitosis, occasional atypia and with focal areas showing giant cells, which again favored a diagnosis of fibrosarcoma.
Immunohistochemistry forms an integral part of pathologic diagnosis that aids in arriving at an accurate histopathological diagnosis. Vimentin, S-100 and CD68 markers were used to help in reaching the diagnosis. Vimentin was constantly positive with S-100 showing negativity, thus ruling out the possibility of neural tumours. Focal positivity with CD68 added on to the fibrohistiocytic nature of the tumours as quoted in the literature, to be one of the variants of OS [15]. Such challenging cases often pose a problem in executing a definitive surgical treatment option.
Radiographic evaluation is important in diagnosing OS as clinical symptoms like pain, swelling, paresthesia, and loosening of teeth are not specific [16]. Better knowledge of the radiological features can lead to an earlier diagnosis thus improving its prognosis [17]. Characteristic radiographic features include destruction of the cortical plates, widening of periodontal ligament space and perpendicular spiculated pattern of new periosteal bone formation [18]. However, these characteristic features are often missing and thus, OS is difficult to interpret as a result of varied appearance ranging from purely osteolytic or osteoblastic lesion, to a mixture of both [10]. The recurrent nature of the lesion with its aggressive nature, in our case, was confirmed by CT scan, where extensions into the orbit promoted a thorough sampling of the lesion. The classic appearance of tumour osteoid was the characteristic finding that led to the diagnosis of OS. Radiographs suggested a lytic destructive lesion while CT findings ruled out any metastasis.
The recurrence rate in large, bulky tumours is 80% within 24 months. Early diagnosis and radical surgery are the key factors for the better survival rate in this condition. Treatment of this lesion is radical surgery consisting of complete resection along with a margin of normal surrounding tissue, usually accompanied by chemotherapy. The best survival rate of five years was noted with radical surgery [12]. Anatomic limitations in face can cause some difficulties in achievement of uninvolved margins [19]. The combination of closed margins and poor histologic response appears to be the reason for high local recurrence of these lesions. The prognosis for patients after local recurrence of OS is poor.
Recent studies have focused on new markers for early detection of OS. Park et al. in their recent study showed by immunohistochemistry that high-grade OS of the jaws had a higher expression rate of proteins involved in regulation of growth and metastasis of cancer cells (ezrin and Metastatic tumour antigen) suggesting that their positivity can be used as additional prognostic markers in OS of the jaw [20]. Another study analyzed the clinicopathological features and immunohistochemical expression of p53, MDM2, CDK4, PCNA and Ki67 proteins in 25 head and neck OS and found 52% positivity for p53, 24% for MDM2, 84% for CDK4, 92% for PCNA and 88% for Ki-67 suggesting PCNA as one of most favourable prognostic marker [21] . Another study suggested the role of 12q13-15 genes in OS of the jaws with amplification and overexpression of these genes might help in detecting high-grade tumours [22].
Response to chemotherapy (CT) is best seen in fibroblastic subtypes and poorest in chondroblastic subtype [11]. Multimodality therapy using chemotherapy and radiation treatment (RT) has shown improvement in survival rates in the OS of the extremity, from 20% to 70%, which is better than the 40% survival rate reported for jaw OS [23]. A recent study which was performed to evaluate the outcomes of multimodality treatment in patients with OS of the jaw/craniofacial region with positive/uncertain resection margins, found that combined modality treatment, comprising of surgery and RT (median dose, 60 Gray) significantly improved local control (P = 0.006) and overall survival (P < .0001) as compared to surgery alone [24]. In the case presented here, patient was subjected to partial maxillectomy coupled with RT & CT and one episode of recurrence of tumour was reported at the time of write up, at a follow up for past one year. Multidimensional approaches including conventional surgical and prosthetic procedures, however, should be considered in accordance with the individual patient and specific defect.
Various new treatment modalities are being investigated for the treatment of OS of the jaws. A recent study focused on the expression of inducible nitric oxide synthase (iNOS) in OS of the jaw, and its relationship with tumour angiogenesis and clinicopathological characteristics. They concluded that iNOS may promote tumour angiogenesis in OS of the jaw and may present an important target in anti-tumour therapy [2]. Development of microvascular tissue transfer now offers various reconstructive options and has greatly improved the result of mid-face reconstruction [25].


