All published articles of this journal are available on ScienceDirect.
Short-Term Analysis of Human Dental Pulps After Direct Capping with Portland Cement
Abstract
This study evaluated the short-term response of human pulp tissue when directly capped with Portland cement. In this series of cases, twenty human third molars that were scheduled for extraction were used. After cavity preparation, pulp exposure was achieved and Portland cement pulp capping was performed. Teeth were extracted after 1, 7, 14 and 21 days following treatment and prepared for histological examination and bacterial detection. Each group had 5 teeth. The results were descriptively analysed. Dentin bridge formation was seen in two teeth with some distance from the material interface (14 and 21 days). Soft inflammatory responses were observed in most of the cases. Bacteria were not disclosed in any specimen. PC exhibited some features of biocompatibility and capability of inducing mineral pulp response in short-term evaluation. The results suggested that PC has a potential to be used as a less expensive pulp capping material in comparison to other pulp capping materials.
INTRODUCTION
Preservation of pulp vitality during restorative procedures is a mater of concern in operative dentistry [1]. Some authors have demonstrated that pulp alterations under different restorative materials were related to the presence of bacteria from microleakage [2,3]. Besides, the major cause of failure in pulp capping treatment has been attributed to bacterial contamination rather than the irritant properties of capping materials. If this hypothesis is valid, pulp healing could be expected if a material maintains the seal against microleakage [4]. Studies have showed that calcium hydroxide is not the only material capable of stimulating reparative dentin deposition [5] or dentin bridge formation [6]. The formation of dentin bridges is an intrinsic response of the exposed pulp in the absence of bacteria [7,8].
Over the last years, studies have compared healing effects and the composition of PC with those of Mineral trioxide Aggregate (MTA). Regarding the healing effects, it was observed that osteoblast-like cells had similar growth and matrix formation when growing on set PC [9], while other authors noted that PC allowed dentin bridge formation after pulpotomy on dogs [10] and induced calcite crystal granulations deposition when placed in dentin tubes that were implanted subcutaneously in rats [11]. Moreover, it has been demonstrated that PC and MTA have a similar effect on pulp cells when used as a direct pulp-capping material in rats [7] as well as comparable antibacterial activity [12]. Such similarities in healing effects induced by these cements may be related to the similarities in their composition. PC has major ingredients in common with MTA such as calcium phosphate, calcium oxide, and silica. MTA also contains bismuth oxide, which increases its radiopacity and is absent in PC [12,13]. However, because of the low cost of the cement, it is reasonable to consider PC as a possible substitute for MTA in endodontic applications.
The purpose of this study was to evaluate short-term pulp response after PC application on exposed pulps of human teeth.
MATERIALS AND METHODOLOGY
Tooth Selection
Teeth were obtained from seven patients from the Semiology Clinic of the School of Dentistry of the University of Pernambuco. Sample consisted of twenty noncarious human third molars which were previously scheduled for extraction for orthodontic reasons. The patients ages ranged from 19 to 31 years (mean age = 24.8 years) for both genders. The patients signed consent forms after receiving a thorough explanation about the experiment, clinical procedures and possible risks. The patients were asked to read and sign a consent form allowing the clinical procedure. Both, the consent form and the research project were approved by the Ethical Committee (protocol178/03) from the University of Pernambuco, Brazil.
Cavity Preparation and Pulp Exposure
Local or regional anesthesia was obtained using 2% mepivacaine (DFL, São Paulo, SP, Brazil). A dental dam was used to isolate the teeth during the treatment. Before cavity preparation, the teeth were cleaned with prophylactic paste.
Class I cavities were prepared using new diamond bur (#1015 – KG Sorensen, São Paulo, SP, Brazil) at high speed, under copious air/distilled water-cooling. The pulpal floor was extended 0.5mm - 1mm from the pulp, based on a previous radiographic examination. Pulp exposures were obtained with a diamond bur (1,2mm diameter – KG Sorensen, São Paulo, SP, Brazil) at high speed and under water. Bleeding control was achieved using a sterile saline solution and sterilized cotton pellets. A new bur was used for each tooth.
Material Placement
Portland cement (CP II – F32, ITAPESSOCA AGRO-INDUSTRIAL S.A., Goiânia, PE, Brazil) was mixed with distilled water to achieve the desired consistency. PC paste pulp capping was performed and the cavities were sealed with glass ionomer cement (DFL, São Paulo, SP, Brazil). Teeth were extracted after 1, 7, 14 and 21 days following pulp capping treatment, and assigned into four subgroups according to the time of extraction. Each group had 5 specimens (n=5) resulting in a sample with 20 teeth.
Hist1ological Preparation
Following postoperative intervals of 1, 7, 14 and 21 days, the teeth were extracted, and the apical third of the root was removed in order to facilitate formalin penetration and fixation (10% buffered formalin solution). Demineralization was obtained with 5% nitric acid after 4-5 weeks. Specimens were paraffin embedded and three-µm sections were obtained, which were stained with Hematoxylin-Eosin and the Brown & Hopps technique [14]. An extracted tooth with necrotic and infected pulp was used as a control for the Brown & Hopps technique to demonstrate the presence of bacteria.
Evaluation Criteria
The sections were blindly evaluated by a pathologist according to the criteria previously established (Cox et al. [4], Cox et al. [15] and Akimoto et al. [16]) and described in Table 1.
| Scores | Inflammatory Cell Response |
|---|---|
| 1 2 3 4 |
None or a few scattered inflammatory cells present in the pulp beneath the exposure site Polymorphonuclear leukocytes (acute) or mononuclear lymphocytes (chronic) in an inflammatory lesion Severe inflammatory lesion appearing as an abscess or dense infiltrate involving one third or more the coronal pulp Completly necrotic pulp |
| Scores | Soft Tissue Organization |
| 1 2 3 4 |
Normal or almost normal tissue morphology below the exposure site and throughout the pulp Lack of normal tissue morphology below the exposure site, with deeper pulp tissue appearing normal Loss of general pulp morphology and cellular organization below the exposure site Necrosis in at the coronal third of the pulp |
| Scores | Dentinal Bridge Formation |
| 1 2 3 |
New barrier tissue directly adjacent to some portion of the restorative material New dentin bridge some distance from the material interface No evidence of any dentin tissue formation in any of the tissue sections |
| Scores | Bacterial Staining |
| 1 2 3 |
Absence of bacterial staining in any section Positive bacterial staining reaction along the cavity walls or within the cut dentin tubules Positive bacterial staining reaction within the dental pulp |
RESULTS
The values related to inflammatory cell response, soft tissue organization, dentin bridge formation and bacterial staining are summarized in Table 2. After 1 day, no dentin bridge formation was observed in any of the 5 teeth in this group. Inflammatory cell response ranged from soft to mild (Fig. 1A). Pulp tissue located below the exposure (1,2mm diameter) was generally disorganized and contained congested blood vessels.
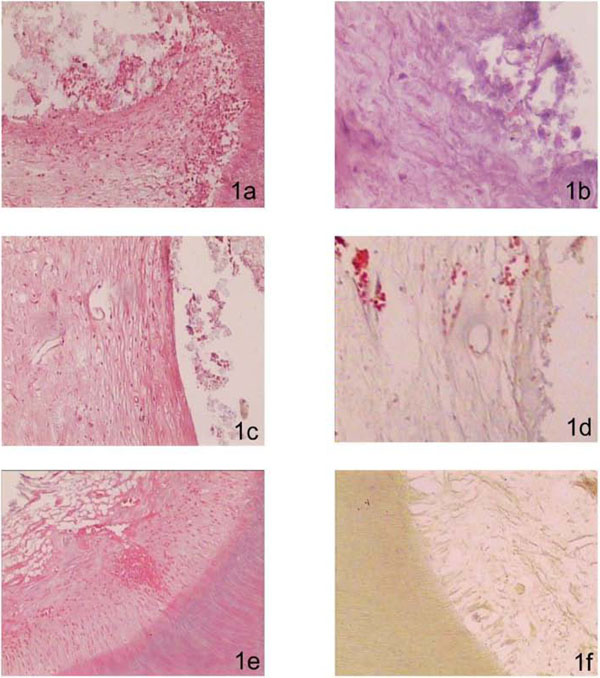
1a no dentin bridge formation was observed. Inflammatory cell response varied from soft to mild (1st day, HE – 100X). In 1b, a necrotic area could be observed underneath the exposure site (7th day, HE – 200X). In 1c, no dentin bridge formation was observed, however, pulpal tissue exhibited organized collagenic deposition (7th day, HE - 100X). In 1d, a dentin bridge distant from the interface pulp-capping material was present. Inflammatory cells were absent (14th day, HE - 200X). In 1e, exposed tissue presented normal characteristics, with new odontoblast-like cells beneath the pulp-PC interface; inflammatory cell response was absent in all cases (21st day, HE - 100X). In 1f, absence of bacteria throughout the specimen was observed (21st day, HE - 100X).
| Specimens | ICR | STO | DBF | BS |
|---|---|---|---|---|
| 1G1 | 1 | 1 | 3 | 1 |
| 2G1 | 1 | 1 | 3 | 1 |
| 3G1 | 1 | 1 | 3 | 1 |
| 4G1 | 1 | 1 | 3 | 1 |
| 5G1 | 1 | 1 | 3 | 1 |
| 1G2 | 1 | 1 | 3 | 1 |
| 2G2 | 1 | 1 | 3 | 1 |
| 3G2 | 1 | 1 | 3 | 1 |
| 4G2 | 1 | 3 | 3 | 1 |
| 5G2 | 1 | 1 | 3 | 1 |
| 1G3 | 1 | 2 | 3 | 1 |
| 2G3 | 1 | 2 | 2 | 1 |
| 3G3 | 1 | 2 | 3 | 1 |
| 4G3 | 1 | 3 | 3 | 1 |
| 5G3 | 1 | 2 | 3 | 1 |
| 1G4 | 1 | 2 | 3 | 1 |
| 2G4 | 1 | 2 | 3 | 1 |
| 3G4 | 1 | 2 | 2 | 1 |
| 4G4 | 1 | 2 | 3 | 1 |
| 5G4 | 1 | 2 | 3 | 1 |
ICR – Inflammatory cell response; STO – Soft tissue organization; DBF – Dentinal bridge formation; BS – Bacterial staining; G – Group.
No dentin bridge formation was observed in any of the 5 teeth in this group after 7 days. Inflammatory cell response was absent in all cases and pulp tissue disorganized and congested blood vessels were observed in one specimen (4G2) below the exposure area (Fig. 1B) while 2G2 specimen exhibited concentrated collagen fibers (Fig. 1C).
After 14 days only one specimen (2G3) of this group exhibited a dentin bridge; however, this bridge was found at some distance from interface pulp-capping material interface (Fig. 1D). Inflammatory cell response was absent in all cases, but pulp tissue below the exposure area was generally disorganized and contained congested blood vessels.
Tissue under the exposure site presented normal pulp tissue characteristics after 21 days, with new odontoblast-like cells beneath the pulp-PC interface. In this period, one specimen exhibited reparative dentin. In the teeth exhibiting no dentin bridges formation, the pulp tissue showed no changes in morphology, and inflammatory cell response was absent in all cases. Area of collagenic fiber deposition was observed in one specimen (Fig. 1E).
Brown & Hopps technique revealed no bacteria in any section of teeth specimens (Fig. 1F).
DISCUSSION
The main aim of this investigation was to assess the short-term response of human pulps capped with PC. This was the first time that the use of PC on human pulps was tested. PC has demonstrated biocompatibility as much acceptable as MTA when applied in deep cavities in animal teeth [10]. Based on the supposition that effective sealing is more important than capping material properties, it was observed partial bridges only in teeth with problems in the coronal sealing indicating that microorganism’s presence was responsible for problems in the healing process [10].
Some authors reported that the initial control of pulpal bleeding was the most critical biologic and clinical issue [16]. One approach to determine the best material used for the initial bleeding control during direct pulp capping would involve short-term histological analysis of materials used as direct pulp capping agents, such as PC and MTA. In our study, bleeding was not detected after PC application. This evidence could indicate an acceptable property of PC considering the fact that the presence of a blood clot between capping material and pulp tissue may jeopardize pulp repair and attract microorganisms, leading to infection [17]. Moreover, as restorative procedures may have a significant influence on the effectiveness of pulp capping and bleeding can compromise the efficacy and durability of adhesion [18,19], the use of PC as direct pulp capping material could ensure proper adhesion of adhesive systems or glass ionomer cements, which in turn could avoid bacterial contamination from any possible microleakage after either shortor long-term.
The biological reaction of the pulp tissue to PC was satisfactory, and the tissues demonstrated that the material is not irritating. Inflammatory cells of the first day were always confined to the area straightly below to the exposure, probably because of the procedure itself. Absence of regular pulp morphology and cellular organization below the exposure site were detected in two cases (4G2, 4G3).
The mechanisms of action of MTA and PC are similar. As both materials have calcium hydroxide. The reaction of calcium from calcium hydroxide with carbon dioxide from the pulp tissue produces calcite crystals [10] when any of these materials is used [20].
An important finding in this study was the relation between the different zones of pulpal reaction to each other. The zones of PC covered the exposed pulp and induced the reaction. The zone of degeneration, although resulting from pulpal injury and chemical effects of the material protected the underlying vital tissues during early stage of healing. PC probably caused denaturation of adjacent cells because of the high surface pH, mainly when freshly mixed.
Finally, the primary calcific bridge, as an early barrier between pulp tissues and the cavity allow the pulp to organize its cellular elements and form the permanent dentin bridge. In our study, new dentin bridge some distance from the material interface was found in one of the specimens (2G3). The response to direct pulp capping with materials such as PC is the formation of a dentin barrier, resulting from the recruitment and proliferation of undifferentiated cells, which may be either stem cells or dedifferentiated and transdifferentiated mature cells. Once differentiated, the cells synthesize a matrix that undergoes mineralization. The extracelular matrix components can induce either reactionary dentin formation or formation of dentin barriers [10, 21].
This finding substantiates the results reported by Berman [22]. The presence of a dentin bridge under PC could be related to an inherent ability of the pulp to heal [3,22-24] regardless of the capping material [3,6,15,17]. However, no tubular dentin formation was observed after 21 days. Using calcium hydroxide, Demarco et al. [21] obtained the formation of 2-layered dentin bridges, an outer osteodentin region, and an inner tubular region, in 90 days. This tubular formation is a signal of maturation of the dentin [19].
Studies using germ-free animals have shown the importance of bacteria in the response of pulp tissue [7,8]. In this current study, no bacteria contamination was detected in any teeth. This means that the bacteriostatic action of CP was enough to reduce the number of viable bacteria near the pulp exposure. However, it is important to emphasize that the absence of bacterial staining did not mean sterility in those cases exhibiting inflammatory responses. A small number of bacteria can produce false-negative results [25,26] or may be removed from the specimen during histological preparation [3,20,22]. Additionally, many bacteria may adhere to the restoration that is lost during the histological process [27].
The results of this study should be carefully evaluated because the capping procedure was accomplished in sound teeth. In most clinical scenarios, the pulp exposure frequently occurs by a carious process, in which the level of inflammation is much higher, and bacteria presence. The idea would be testing these procedures under the aforementioned condition to verify the reproducibility of the findings reported in this clinical evaluation. However, although the use of vital healthy teeth for this kind of study has limitations, it still has the benefit of standardization and can be regarded as acceptable in respect to material selection and handling.
CONCLUSION
Within the limitations of this current study, it is possible to conclude that after short-term histological evaluation:
- Inflammatory cell response was absent in most of cases.
- Dentin bridge was found in 10% of the specimens.
- Tissue under the exposure site presented the characteristics of normal pulp tissue, with new odontoblast-like cells located below the pulp/PC interface after 21 days.
In summary, the findings of our study support the idea that PC has potential to be used as a pulp capping material wich is capable of inducing short-term mineral pulp response. Further, long-term studies are necessary before unlimited clinical use can be recommended.
ACKNOWLEDGEMENTS
The authors are grateful to the Department of Oral Pathology and Semiology of School of Dentistry, UPE. This study was partially supported by CAPES.


