All published articles of this journal are available on ScienceDirect.
Acidogenic Potential of “Sugar-Free” Cough Drops
Abstract
A patient presented with extensive marginal ditching around restorations recently placed during whole-mouth rehabilitation. The patient was not xerostomic and was otherwise normal except for the self-reported excessive use of “sugar-free” cough drops sweetened with sorbitol and Isomalt® (an equimolar mix of glucosyl-mannitol and glucosylsorbitol). This prompted an in vitro investigation to determine whether Streptococcus sobrinus 6715, a cariogenic streptococcus, could grow and produce acid in growth medium containing an aqueous extract of such “sugar-free” cough drops. The results indicate that S. sobrinus 6715 uses Isomalt® and sorbitol extensively, producing terminal culture pH as low as 4.2 when grown on medium with cough drop extract containing these sugars. This pH is sufficient to demineralize dental enamel. Patients should be cautioned against the chronic overuse of “sugar-free” cough drops and other “sugar-free” confections sweetened with a mixture of Isomalt® and sorbitol.
INTRODUCTION
Dental caries remains one of the three most common infectious diseases in the world [1]. A major risk factor for this disease is the frequent ingestion of sugars (especially sucrose) that can be rapidly fermented by oral bacteria, producing sufficient acid to dissolve dental enamel [2, 3]. While highly acidogenic and adherent organisms such as Streptoccoccus mutans and S. sobrinus have been implicated as primary pathogens, it has become evident that other acidogenic bacteria are present in significant numbers in carious lesions and may be important in lesion progression. These include so-called low-pH non-mutans streptococci as well as members of the genera Lactobacillus, Actinomyces, Atopobium, Bifidobacterium, Propionibacterium, and Veillonella [4].
According to the Ecological Plaque Hypothesis, frequent ingestion of sucrose and other fermentable sugars can produce regular and prolonged low pH in dental plaque. These acidic conditions provide a selective pressure that allows acidogenic and acid-tolerant bacteria such as the mutans streptococci and lactobacilli to become dominant in the dental plaque microbial community, while at the same time suppressing non-cariogenic components of the oral microbiota [5, 6]. Thus, the suppression of acid-producing bacterial metabolism by metabolic inhibitors and the replacement of sucrose with non-fermentable sweeteners in foodstuffs have been among the goals of caries control [2, 5-7].
Non-fermentable sweeteners can be high-intensity (saccharin, aspartame, sucralose) or carbohydrate [2]. Non-fermentable carbohydrate sweeteners are polyols (sugar alcohols) and include sorbitol, hydrogenated glucose syrups, Isomalt® (an equimolar mixture of α-D-glucopyranosyl -1,6-sorbitol and α-D-glucopyranosyl -1,6-mannitol) and others. These polyols are regarded as “tooth-friendly”; i.e., they do not promote dental caries [2, 8], and studies have demonstrated minimal, short-duration pH falls after brief exposure of dental plaque to polyols in plaque telemetry experiments [9] and very low cariogenicity compared to sucrose in animal experiments [10].
An adult patient at the Louisiana State University School of Dentistry in New Orleans was treated for rampant caries with a complete full-mouth restoration. Approximately one year later the patient returned to the clinic with extensive ditching around the margins of the new restorations. Staining and ditching at margins of restorations have been suspected as indicators of marginal or secondary caries [11]. Margins with ditches of 0.4 mm or greater contain high numbers of bacteria, mutans streptococci, and lactobacilli compared to clinically intact margins or margins with narrow ditches [12], suggesting a possible microbial etiology for at least some marginal ditching. The patient in question had normal salivary flow and normal dental and medical histories, except for the use of 8-12 so-called “sugarless” or “sugar-free” cough drops per day during the preceding year. The brand used by the patient was sweetened with aspartame, hydrogenated glucose syrup, and Isomalt®, and was marketed as “sugar-free”.
The purpose of the present study was to examine in vitro whether a known cariogenic streptococcus could metabolize sweeteners from “sugar-free” cough drops to produce acid sufficient to demineralize dental enamel.
MATERIALS AND METHODS
Cough Drop Extract
“Sugar-free” cough drops of the same brand and flavor used by the patient in question were purchased at a local pharmacy. The listed sweetening agents included aspartame, hydrogenated glucose syrup, and Isomalt®; amounts or concentrations were not given. Other ingredients were menthol, canola oil, FD&C Blue #1 Aluminum Lake, and flavors.
Cough drops from several packages were unwrapped, pooled, weighed, and dissolved in deionized water. After filtration (Whatman #1) to remove oil, extracts were slightly turbid with a faint blue to blue-green color. In separate preparations, extracts were diluted with deionized water to give concentrations of 280 and 500 mg/ml, based on weight of the unwrapped cough drops. Extracts were stored at -20°C. Aliquots were thawed and sterilized by membrane filtration immediately before use.
Bacteria and Bacterial Growth
Streptococcus sobrinus 6715, a highly cariogenic strain [13], was obtained from the culture collection of the Department of Microbiology, Immunology, and Parasitology, Louisiana State University Health Sciences Center, New Orleans. Cultures were grown essentially as previously described [14] using a Trypticase peptone medium [15]. Briefly, basal medium was sterilized (autoclave), after which separately sterilized (membrane filtration) growth sugars were added aseptically. Growth sugars included glucose, mannitol, sorbitol, and cough drop extract, with sugar concentrations as indicated in Fig. (1) and Tables 1 and 2. Growth was determined by measuring culture absorbance at 660 nm (A660) and culture terminal pH was determined with a pH meter.
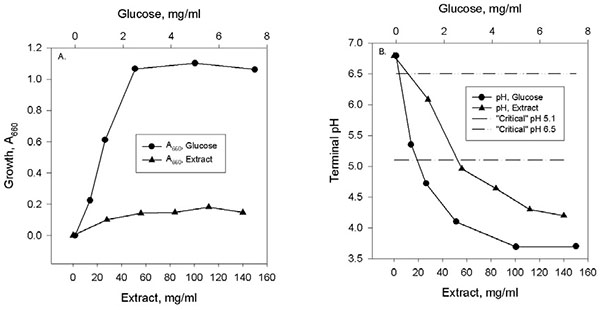
Growth and terminal pH of cultures of Streptococcus sobrinus 6715 grown in liquid medium containing varying concentrations of glucose or Cough Drop Extract (CDE). Note dual X-axes above (glucose) and below (CDE) each graph. Fig. (1A): Growth measured as A660. Fig. (1B): Terminal pH. Also shown in Fig. 1B are the extreme values for the so-called “critical pH” for the dissolution of dental enamel in oral fluids [17].
Growth and Terminal pH in Cultures of Streptococcus sobrinus 6715 Grown in Trypticase Broth Supplemented with Sugars or Cough Drop Extract (CDE)
| Sugar | A660 | pH |
|---|---|---|
| None | 0.27 | 6.79 |
| CDE, 50 mg/ml | 0.40 | 5.12 |
| Glucose, 4 mg/ml | 1.31 | 3.90 |
| Mannitol, 4 mg/ml | 0.90 | 4.42 |
| Sorbitol, 4 mg/ml | 0.57 | 4.86 |
Concentrations of Isomalt® and Other Sugars in Culture Fluid Before and After pH-Controlled Growth (pH 6.5) of Streptococcus sobrinus 6715
| Sugar | Source | Concentration, Moles/Liter | |
|---|---|---|---|
| Sterile Medium | Spent Culture Fluid | ||
| Glucose | Trypticase Broth | 0.022 | 0.015 |
| Sorbitol | CDE* | 0.062 | None detected |
| Mannitol | CDE | None detected | None detected |
| Isomalt | CDE | 0.119 | 0.034 |
* CDE: Cough drop extract.
For growth-terminal pH experiments, cultures (10 ml, 18x150 mm culture tubes) were incubated at 37°C without mixing for 48-72 hours. For pH-controlled growth, a 500 ml culture containing cough drop extract (50 mg/ml) was grown as described [14] for 62 hours at 37°C with gentle stirring. The pH was maintained at 6.5 by periodic addition of 0.2 M KOH using a pH controller (Cole-Parmer). After removal of bacterial cells by centrifugation, the supernatant fluid was sterilized (membrane filtration) and submitted for analysis of residual sugars. A sample of sterile uninoculated growth medium was retained as a control for sugar analysis.
Sugar Analysis
Sugar analysis was done by high-pressure liquid chromatography (HPLC) essentially as described [16], using a Dionex HPLC with CarboPac PA10 column (4x250 mm), Dionex ED40-1 pulsed amperometric detector, and 100 mM NaOH as the eluent. Chromatographic standards (glucose, mannitol, sorbitol, and Isomalt®) were obtained from Sigma.
RESULTS
Growth and Acid Production in Medium Containing Cough Drop Extract (CDE)
Growth and terminal pH of Streptococcus sobrinus 6715 in medium containing either CDE or other sugars are shown in Fig. (1) and Table 1. Growth and terminal pH as functions of sugar concentration are shown in Fig. (1). Growth, measured as final culture turbidity (A660) is shown in Fig. (1A); values are corrected for the scant growth that occurred in medium with no added sugar. The results indicated that S. sobrinus 6715 could grow in medium containing CDE as the sugar source, although the biomass yield was less than 20% of that obtained with the preferred substrate glucose.
Acid production, measured as terminal culture pH, is shown in Fig. (1B). Measurements of pH were done on the same cultures for which A660 had been measured in Fig. (1A). CDE was shown to be an acidogenic substrate for S. sobrinus 6715. Terminal pH decreased as CDE concentration increased, reaching a terminal pH as low as 4.2. Glucose was even more acidogenic, with a low terminal pH of 3.7. Also shown in Fig. (1B) are reference lines representing the possible range of the so-called critical pH at which dental enamel could dissolve in dental plaque fluid (pH 5.1) or in saliva with low concentrations of calcium and phosphate ions (pH 6.5) [17].
Growth and terminal pH of S. sobrinus 6715 cultures grown in medium supplemented with CDE or its constituent sugars is shown in Table 1. Glucose, mannitol, and sorbitol are components of Isomalt®, and sorbitol is the major free constituent of hydrogenated glucose syrup [18]. Aspartame, the remaining sweetener, is not metabolized by bacteria to produce acid [9]. Scant growth and approximately neutral pH were observed in medium with no added sugar, while all other sugars produced both growth and terminal pH at or below that which would dissolve enamel under plaque at a tooth surface. An average cough drop weighed 3.65 grams (data not shown). Thus, 50 mg of CDE per ml of culture corresponded to approximately 1.4% of a single cough drop.
Analysis of Residual Sugars After Growth of S. sobrinus 6715 in Medium Supplemented with CDE
HPLC analysis of sugars in CDE-supplemented medium before and after bacterial growth is shown in Table 2. Culture pH was maintained at 6.5 to prevent inhibition of growth by low pH, thus amplifying possible use of CDE during growth. Sterile medium contained glucose, sorbitol, and Isomalt®; mannitol was not detected. Spent culture medium after growth contained reduced concentrations of glucose and Isomalt®; sorbitol and mannitol were not detected. Approximately 71% of Isomalt® was consumed during growth.
DISCUSSION
Dental caries is one of the most widespread human infectious diseases [1]. A diet high in fermentable sugars, especially sucrose, is regarded as a major risk factor for this disease [2, 3, 19]. Furthermore, according to the Ecological Plaque Hypothesis frequent and prolonged contact of these sugars with dental plaque alters the microbial composition of plaque in favor of bacteria that are both acidogenic and acid tolerant, thus increasing the cariogenic capacity of dental plaque [5, 6]. Therefore a major effort in food technology has been to produce confections and snack foods in which sucrose has been replaced by sweeteners that are metabolized very slowly or not at all by oral bacteria, the goal being to greatly reduce or eliminate acid production by plaque following consumption of such foods [7, 20].
A number of such sweeteners have been tested and identified as having reduced or low caries risk [9, 21]. Most relevant for this study are the polyols sorbitol, mannitol, and Isomalt® (an equimolar mixture of α-D-glucopyranosyl –1,6-sorbitol and α-D-glucopyranosyl –1,6-mannitol). By in situ measurement of plaque pH, these compounds have been shown to produce minimal drop in plaque pH following a single brief oral exposure to the agent [9], and studies with pure cultures of oral bacteria have shown that acid production from these sweeteners is quite slow compared to sucrose or glucose [10, 19]. However, frequent ingestion of sorbitol can lead to increased acid production from dental plaque [22], and “excessive” use of confections containing sorbitol and mannitol can lead to increased levels of mutans streptococci in plaque [23]. Thus it appears that the Ecological Plaque Hypothesis [5, 6] could be invoked in the apparent alteration of dental plaque composition and acidogenicity following frequent and prolonged exposure to at least some slowly metabolized “non-cariogenic” sweeteners.
The present study was precipitated by the occurrence of extensive marginal ditching in an adult patient with a large number of recent restorations. Marginal ditching may be an indicator of marginal or secondary caries, and the bacterial flora of marginal ditches can be enriched for potentially cariogenic mutans streptococci and lactobacilli [4, 10, 12]. The patient was not xerostomic and had an unremarkable history except for the daily use of 8-12 “sugar-free” cough drops over the period of approximately one year. The cough drops were sweetened with the non-metabolizable agent aspartame as well as hydrogenated glucose syrup and Isomalt®. The major free constituent of hydrogenated glucose syrup is sorbitol [18], and both sorbitol and Isomalt® are regarded as low-risk for caries [9]. However it has been pointed out that hard candies (e.g., cough drops) are long lasting in the mouth and thus produce extended exposure time in the oral cavity as the sweeteners are gradually released [2]. According to the Ecological Plaque Hypothesis [5, 6], this frequent and prolonged exposure could select for plaque bacteria able to metabolize sorbitol and Isomalt®.
The component disaccharides of Isomalt® both are α-glucosides and α-glucosidase activity is widely distributed in oral bacteria [24-27]. In a survey of 210 strains of mutans streptococci, 100% of S. sobrinus strains and 91% of S. mutans strains were positive for α-glucosidase activity [24]. Thus it is reasonable to presume that S. sobrinus 6715, used in this study, could degrade Isomalt® to produce glucose, sorbitol, and mannitol. The data show production of acid from CDE and its constituent sugars (Fig. 1 and Table 1) and the consumption of Isomalt® during bacterial growth (Table 2), supporting this contention. Thus, a putative catabolic pathway would begin with α-glucosidase-mediated cleavage of Isomalt® to produce glucose, mannitol, and sorbitol, which would be further catabolized via glycolysis yielding a mixture of lactic, acetic, and formic acids [19, 22]. While mannitol and sorbitol are thought to be catabolized slowly, glucose is a rapidly fermented substrate.
As shown in Fig. (1), CDE is both a growth and an acidogenic substrate for S. sobrinus 6715, with a terminal pH of 4.2 in medium containing 140 mg CDE per ml; this corresponds to approximately 3.84% of a single cough drop per ml (data not shown). Table 1 shows that CDE and sugars derived from CDE also are acidogenic with terminal pHs ranging from 5.12 (CDE) to 3.90 (glucose); the concentration of CDE used in this experiment corresponds to approximately 1.37% of a single cough drop per ml.
Dental decay occurs when dental enamel is dissolved by acids made during bacterial metabolism, and the pH at which this process begins often is described as the “critical” pH. The so-called critical pH in fact does not have a fixed value but is inversely proportional to concentrations of calcium and phosphate in oral fluids, and can range from 6.5 in a person with low salivary phosphate and calcium to as low as 5.1 in the fluid phase of dental plaque [17]. As shown in Fig. (1) and Table 1, in vitro growth of S. sobrinus 6715 in medium with CDE can produce culture pH as low as 5.1, even with only 50 mg CDE per ml. Even lower pHs were produced with increased concentrations of CDE (Fig. 1).
This study raises a number of questions. Clinical details of tooth damage in affected patients would be valuable. Unfortunately these are not available for the present patient, whose dental records were destroyed and lost when the Louisiana State University School of Dentistry was flooded in the aftermath of Hurricane Katrina. However, there are anecdotal reports of similar tooth damage in other otherwise healthy dental patients who overuse “sugar-free” cough drops (J. Newman, D.M.D., personal communication). It would be interesting to document whether prolonged dietary exposure to Isomalt® would in fact lead to a more acidogenic and acid-tolerant oral microflora as predicted by the Ecological Plaque Hypothesis [5, 6]. However, human and animal trials of excessive use of “sugar-free” cough drops could be both experimentally and ethically problematic. Nevertheless, production of α-glucosidase activity is widespread in cariogenic oral streptococci [24], and it would be feasible to examine both the ability of these bacteria to grow on and produce acid from Isomalt® and the enzymatic cleavage of Isomalt® by purified streptococcal α-glucosidases.
The clinical picture of our patient and the ability of S. mutans 6715 to produce acid from CDE (Fig. 1 and Table 1) and to consume Isomalt® during growth (Table 2) are provocative especially when interpreted in light of the Ecological Plaque Hypothesis [5, 6]. It would seem only prudent to counsel dental patients against chronic and excessive overuse of “sugar-free” cough drops, confections, and snack foods sweetened with Isomalt® and hydrogenated glucose syrup.


