All published articles of this journal are available on ScienceDirect.
Integration of Oral Health in Long-Term Care Facilities: Burden, Barriers, and Interventions
Abstract
Introduction
Oral health is integral to person-centered Long-Term Care (LTC), with oral diseases linked to frailty, malnutrition, aspiration pneumonia, and reduced quality of life.
Therefore, the study aimed to synthesize recent evidence on the burden of oral disease in LTC and key barriers, and to review existing interventions that support sustainable oral health integration.
Methods
This study is a narrative review of peer-reviewed literature from PubMed, Scopus, Web of Science, and Google Scholar that used terms related to “long-term care” and “oral health,” limited to English-language studies addressing epidemiology, practice, interventions, or policy in LTC settings.
Results
Across LTC facilities, residents exhibit high treatment needs, hyposalivation, and impaired oral function, including downstream associations with dysphagia, pneumonia, and poor health-related quality of life. Persistent barriers include low organizational priority, workforce and time constraints, limited staff training, and dementia-related care resistance. Promising strategies include professional oral health management programs, structured nursing-led protocols using practical screening tools (e.g., Oral Assessment Guide and Oral Health Assessment Tool), and interprofessional education (e.g., MOTIVATE).
Discussion
At the system level, embedding dental professionals and aligning financing with oral nutrition benefits are associated with improved functional outcomes and facility performance. Cohort data link dental care utilization to lower LTC costs.
Conclusion
Integrating oral health into LTC is feasible and beneficial but demands policy-supported models that combine routine assessment, reimbursed protocols, and sustained interprofessional training to improve function, nutrition, respiratory outcomes, and equity in care.
1. INTRODUCTION
The global demographic shift toward an aging population, increased survival among individuals with chronic diseases and disabilities, and changing family and social structures have expanded the need for Long-Term Care (LTC) beyond older adults [1]. According to the World Health Organization, LTC includes “a broad range of personal, social, and medical services and support that ensures people with, or at risk of, a significant loss of intrinsic capacity—due to mental or physical illness and disability—can maintain a level of functional ability consistent with their basic rights and human dignity” [2]. LTC is provided over extended periods in settings such as homes, communities, residential facilities, or hospitals. Although older adults remain the largest user group, LTC encompasses people of all ages with chronic illnesses, physical or cognitive disabilities, or other long-term functional limitations [1, 2]. Consequently, LTC facilities (LTCFs) have become essential multidisciplinary medical and supportive service providers for individuals with sustained dependency.
Oral health is a fundamental component of overall well-being. The Bangkok Declaration: No Health Without Oral Health (2024) explicitly reaffirms that oral health is an essential component of general and universal health coverage, urging countries to integrate oral health into their health systems and LTC policies [3]. This review argues that oral health is a central determinant of better health outcomes in LTC. Importantly, oral disease is integrally linked to general health and functional status, thereby contributing to frailty, malnutrition, aspiration pneumonia, and diminished health-related quality of life. At the population level, global burden of disease analyses show that although life expectancy is rising, the disability burden in adults aged ≥70 years is increasing, underscoring the relevance of preventable oral diseases [4]. This review aimed to synthesize current evidence on the integration of oral health into LTC systems. In particular, this review focused on burden, reflecting the epidemiological and clinical impact of oral diseases in LTC settings; barriers, including the structural, professional, and behavioral challenges to oral healthcare delivery in LTC; and interventions, encompassing innovative and collaborative models that facilitate the integration of sustainable oral health into LTC policies and practice (Fig. 1). This review consolidated evidence across these domains to inform health policymakers, administrators, and oral health professionals seeking to develop strategies to embed oral health within person-centered interdisciplinary LTC systems.
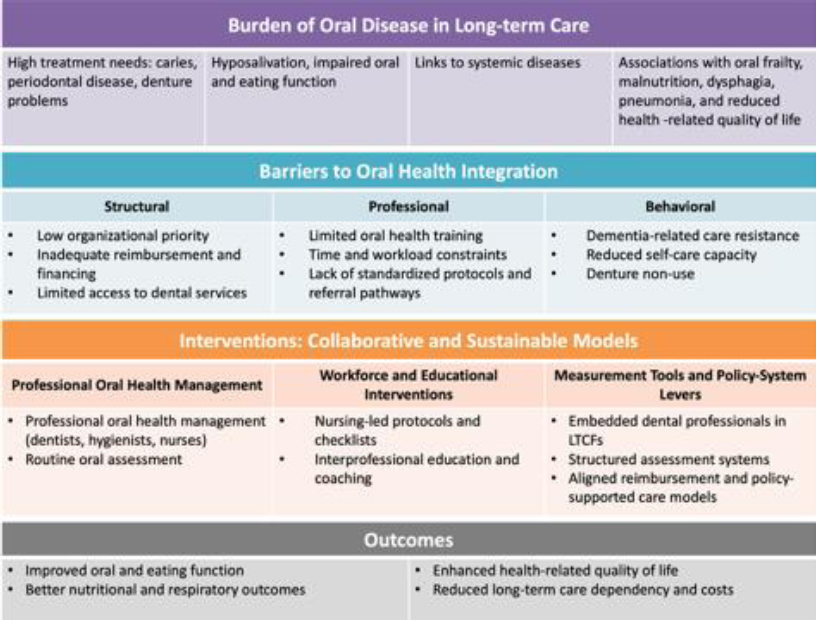
Framework for oral health integration in long-term care facilities (LTCF).
2. METHODS
This review was conducted to identify peer-reviewed papers published between 2016 and 2025 that focused on the integration of oral health into LTC settings. The PubMed, Scopus, Web of Science, and Google Scholar databases were searched. The key search terms employed a combination of “long-term care” and “oral health” across articles published in English. The inclusion criteria were that the articles address oral health in LTC settings and include epidemiological studies, current practices, interventional research, and policy implications. Studies were excluded if they did not address oral health outcomes in LTC settings, were non-peer-reviewed publications, or were not published in English.
3. RESULTS
3.1. Burden of Oral Disease in LTC Settings
3.1.1. Epidemiological Burden
Across LTC settings, treatment needs and functional impairments remain high despite increasing dentition retention. In a Canadian national LTC survey (32 LTCF, n = 559), 79.6% of dentate residents had moderate or severe gingival inflammation in at least one tooth, 43.2% of edentulous residents had poor denture hygiene, and 9% of all residents required urgent dental treatment [5]. In five Austrian LTCFs, denture shortcomings, such as insufficient mandibular denture retention, were reported in 56.0% of lower dentures and chewing problems in 26.7% of denture wearers. Furthermore, 57.1% of dissatisfied denture wearers could not afford to replace their dentures, underscoring the socioeconomic barriers to dental care for LTC residents [6]. In Canadian LTCFs, although tooth retention has improved markedly over ten years, with dentate residents increasing from 56% to 76% and edentulism decreasing from 44% to 24%, new residents had higher dental treatment needs. Dental hygiene treatment needs surged from 43% to 69%, reaching 80% among newly admitted residents, signaling a shift from acute to preventive care and an increasingly dentate resident population [7]. Clinically estimated hyposalivation was also common, with a reported prevalence of 77% among LTCF residents in Finland. This was associated with higher odds of periodontitis (odds ratio [OR] 4.1), lip lesions (OR 2.5), and root caries (OR 1.3) [8]. Microbiome analyses among dentate LTC residents demonstrated a progressive shift toward dysbiotic oral microbiomes with an increasing oral disease burden [9].
3.1.2. Oral-systemic Links and Clinical Consequences
A study of 14 Brazilian LTCFs reported that masticatory impairment was associated with adverse outcomes, including mortality and poor masticatory performance, and predicted all-cause mortality (hazard ratio 1.52) after adjustment [10]. Dysphagia risk was associated with dental and salivary factors among three Brazilian LTCFs, and xerostomia symptoms were associated with dysphagia (prevalence ratios 1.52, 2.86) [11]. A study conducted in nine LTCFs in Japan found that prospective evidence links routine oral assessment scores to better respiratory outcomes. Each 1-point increase in the Oral Assessment Guide (OAG) total scores, indicating poorer oral condition, was associated with a significantly higher risk of pneumonia (adjusted OR 2.29, 95% confidence interval [CI] 1.27-4.14) over 12 months [12]. In a six-month multicenter cohort (31 facilities; n = 1,785), the incidence rates of pneumonia and acute viral respiratory infection (AVRI) were 4.1% and 1.6%, respectively. Furthermore, dysphagia and malnutrition independently predicted pneumonia, while dry mouth and halitosis predicted AVRI [13]. Oral frailty and depressive symptoms have been proven to be associated, suggesting bidirectional interactions between oral dysfunction, mental health, and overall frailty trajectories in older adults [14]. Interventional studies further supported these findings, with a randomized trial demonstrating that combined oral functional exercise and psychological support improved oral frailty while concurrently reducing anxiety and depression in older adults [15]. Together, these findings indicate that oral conditions influence systemic vulnerability through nutritional, functional, and psychosocial pathways.
3.1.3. Quality of Life and Functional Outcomes
Oral symptoms in individuals receiving LTC in Helsinki (n = 2,401) showed consistent associations with malnutrition, lower body mass index, poorer physical function, and poorer perceived health-related quality of life after adjustment [16].
3.2. Barriers: Structural, Professional, and Behavioral
3.2.1. Structural Barriers
Facility leaders perceived a low organizational priority for oral care and cancer screening; limited resident, family, and staff knowledge; regulatory pressures; insurance barriers; and competing demands [17]. In Manitoba, Canada, dentists’ participation in LTC was found to be limited, with only 26.2% providing dental care to LTC residents; they cited barriers such as limited time (60%), lack of invitation from LTCFs (53%), and inadequate equipment (42.6%) [18]. Institutional ethnography across two LTCFs in Canada revealed that frontline oral care was discretionary and weakly guided by institutional protocols. Healthcare aides largely decided when and how to provide oral care based on personal experience rather than standardized instructions. Protocols focused narrowly on toothbrushing and denture cleaning, with little detail provided on disease indicators, referral criteria, or frequency of care [19].
A rapid review found that virtually none of the 59 COVID-19 LTC guidance documents screened contained actionable oral hygiene protocols [20]. Research on LTC faces gatekeeping, reliance on caregiver informants, and protocol burden, reflecting challenges that also affect quality improvement [21]. Population-level insurance claims data further indicate that the onset of the need for home care is associated with a reduction in dental services utilization, highlighting structural access barriers once functional decline occurs [22].
3.2.2. Professional Barriers
Personnel attitudes can be negative. For example, 60% of staff at three Finnish LTCFs reported reluctance to assist with daily oral hygiene, citing resident refusals and time pressure as reasons for this reluctance [23]. Cross-sectional surveys have shown gaps in oral-systemic knowledge among Portuguese LTC workers and New Zealand nursing graduates. Thus, there is an urgent need for updated oral-care protocols and enhanced training [24, 25].
3.2.3. Behavioral Barriers
Alzheimer's disease progression markedly increases behavioral barriers to oral care. The odds of oral care refusal and of dependence on full assistance in performing oral healthcare were shown to increase to 56.35 and 30.52, respectively, at the Functional Assessment Staging Tool stage 7 compared with stages 1-3, with plaque accumulation peaking at severe stages [26]. In Japan, denture non-use among LTC residents was common and independently associated with moderate dementia (OR 4.44, 95% CI 1.18–16.71) and impaired rinsing ability (OR 3.00, 95% CI 1.12–8.06), underscoring how cognitive and oral-motor decline limit denture use and oral-care engagement [27]. Video analyses from two Canadian LTCFs demonstrated that residents brushed for a mean of 60.3 seconds, with 66% omitting brushing of the lingual and occlusal surfaces. This underscores inadequate plaque control and the need for explicit coaching [28].
3.3. Interventions: Collaborative and Sustainable Models in LTCF
3.3.1. Professional Oral Health Management Models
National Japanese data (170,874 residents) showed that enhanced oral care increased the odds of discharge to home (OR 1.07, 95% CI 1.02–1.12) [29]. Moreover, hygienist-led Oral Health Management (OHM) implemented across 25 LTCFs in 17 regions of Japan was associated with better eating function, expressed as twice the odds of maintaining/improving food form compared to those without OHM over one year (OR 2.01, 95% CI 1.03-3.90) [30]. However, a recent Cochrane review (6 randomized controlled trials; 6,244 participants) found insufficient evidence that professional oral care reduces the incidence of nursing home-acquired pneumonia, although low-certainty evidence suggested a 57% reduction in pneumonia-related mortality at 24 months (risk ratio 0.43, 95% CI 0.25–0.76) [31].
3.3.2. Workforce and Educational Interventions
A 14-day quality improvement program implementing a standardized bedside oral-care protocol in a Mississippi LTCF improved staff knowledge (pre-post-test scores increased from 88.8% to 97.7%; p = 0.021) and resident oral health outcomes, as measured by the Oral Health Assessment Tool (p = 0.001). Evidence from a randomized clinical trial in a Swiss LTCF indicated that individualized, visually supported oral health instructions are associated with improved plaque control, denture cleanliness, and oral health-related quality of life among LTC residents, compared with standard verbal instruction alone [32]. Consistent checklist use and staff education led to better protocol adherence, supporting the effectiveness of structured nursing-led oral care models [33]. Recent qualitative evidence indicates that structured oral health assessment tools, such as the International Resident Assessment Instrument for long-term care facilities (interRAI LTCF), enable feasible, systematic oral health assessment by non-dental staff [34]. Interprofessional programs, such as Maine’s Oral Team-Based Initiative Vital Access to Education (MOTIVATE), can enhance staff confidence and increase the application of oral-care skills in nursing homes, leading to gains in knowledge and clinical competence [35]. A pragmatic cluster-randomized trial suggested that caregivers' use of digital tools such as SmartJournal may facilitate incremental improvements in oral hygiene among nursing home residents [36]. Promising expanded-practice hygienist models, such as the Kansas Extended Care Permit initiative, improved access but faced billing and sustainability barriers [37]. Upstream multidisciplinary community programs integrating exercise, oral instruction, and nutrition were found to improve oral diadochokinesis (i.e., motor function and coordination) and swallowing among community dwellers [38], underscoring the value of interprofessional educational initiatives.
3.3.3. Measurement Tools and Policy-system Levers
Practical tools that nurses can use, such as the Oral Health Screening Tool for Nursing Personnel, have demonstrated good sensitivity (0.67 for nurses and 0.71 for caregivers) for natural teeth, dentures, and oral function categories, and adding simple referral rules increases sensitivity from 0.86 to 0.91 [39]. LTCFs that employ dental professionals rather than use private dentists as consultants were found to have higher facility income [40]. At the population level, dental care experience was associated with a 12.4% improvement in LTC dependency (adjusted OR 1.18) among 53,130 Korean LTC beneficiaries [41]. Furthermore, preventive dental visits were associated with lower cumulative LTC insurance costs in an 8-year cohort of 8,429 participants from the Japan Gerontological Evaluation Study, with a reported relative cost ratio of 0.82 (95% CI 0.71-0.95) [42].
4. DISCUSSION
This review highlights converging evidence that integrating oral health into LTC systems yields measurable benefits across functional, clinical, and economic dimensions. However, this remains hindered by systemic fragmentation, professional resistance, and behavioral challenges. Professional OHM programs have demonstrated improvements in eating and swallowing function and reduced pneumonia incidence, demonstrating that regular, protocolized oral care can enhance residents’ health and facility performance. However, large-scale evaluations, such as a Cochrane review [31], underscore the limited certainty and heterogeneity of the evidence for pneumonia prevention when oral care measures are applied, highlighting the need for greater implementation fidelity and standardized outcome measures. In this context, routine use of the OAG has shown promise as a practical nursing tool for pneumonia risk screening and can strengthen collaboration between nursing and dental teams [12]. Workforce and educational interventions, including interprofessional initiatives such as MOTIVATE [35], effectively increase staff confidence and improve the application of skills. However, sustainability continues to depend on reimbursement structures and regulatory support, as illustrated by the Kansas Extended Care Permit model [37]. System-level evidence reinforces the importance of structural integration. Facilities employing dental professionals under Japan’s LTC insurance scheme achieved a higher uptake of reimbursable oral and nutrition benefits and increased income [40], while national data from Korea and Japan linked dental care utilization with improved functional recovery (adjusted OR 1.18) and reduced LTC expenditure [41]. Such findings highlight that even well-intentioned staff require structured evidence-based guidance and continuing education to maintain oral health as a part of holistic care for older adults.
From a research operations perspective, Ramos et al. [21], emphasized that conducting oral health studies in LTC requires trust-building, flexible scheduling, and ethically sensitive data collection approaches, all of which are equally relevant to quality improvement implementation. Collectively, these findings indicate that oral health interventions are most effective when embedded in multidisciplinary policy-supported care frameworks. Future efforts should prioritize scalable quality improvement models, pragmatic trials linking oral and systemic outcomes, and financing mechanisms that incentivize oral health integration as a core element of person-centered LTC.
5. STUDY LIMITATIONS
This review has several limitations. First, the studies varied in study design, country, population, outcome measures, and health system context. This variation made it difficult to compare the studies directly. Second, because this is a narrative review, the selection and interpretation of studies are subject to bias, even though multiple databases and predefined inclusion criteria were used. Third, most intervention studies were either observational or quasi-experimental, with few clinical trials. Nevertheless, the narrative approach brought together epidemiological, clinical, organizational, and policy-level evidence, providing a broad view of the system.
CONCLUSION
Integrating oral health into LTC represents both a clinical imperative and a quality-improvement opportunity. Across diverse settings, evidence indicates that professional oral health management, structured staff training, and interdisciplinary collaboration improve LTCF residents’ oral function, nutritional status, and overall well-being. However, persistent structural, professional, and behavioral barriers, ranging from low institutional priority to limited workforce capacity, continue to constrain its implementation. Scaling successful models will require embedding oral health into broader quality and reimbursement frameworks supported by validated assessment tools and sustained interprofessional education. Ultimately, advancing oral health integration in LTC is pivotal not only for reducing preventable morbidity but also for achieving person-centered health systems that value function, quality of life, and equitable care access.
AUTHOR’S CONTRIBUTIONS
The author confirms sole responsibility for the study conception and design, data collection, interpretation of the results, and manuscript preparation.
LIST OF ABBREVIATIONS
| AVRI | = Acute Viral Respiratory Infection |
| CI | = Confidence Interval |
| LTC | = Long-Term Care |
| LTCF | = Long-Term Care Facility |
| OAG | = Oral Assessment Guide |
| OHM | = Oral Health Management |
| OR | = Odds Ratio |
| MOTIVATE | = Maine’s Oral Team-Based Initiative Vital Access to Education |
ACKNOWLEDGEMENTS
Declared none.


