All published articles of this journal are available on ScienceDirect.
Effect of SARS CoV-2 Infection on Periodontal Tissue: A Systematic Review
Abstract
Background:
Coronavirus disease 2019 (COVID-19) is caused by severe acute respiratory syndrome coronavirus 2 (SARS CoV-2) infection in human respiratory cells. The first spread of this disease first occurred in Wuhan, China, in December 2019, and on March 11, 2020, the World Health Organization declared the spread of the infection as a global pandemic. People who are infected experience mild to moderate clinical symptoms which are characterized by an increase in T cell, CD8, and IgM/IgG levels. The occurrence of severe clinical symptoms reflects an excessive and uncontrolled increase in the secretion of pro-inflammatory cytokines, which impacts the systemic condition and periodontal tissues of affected patients.
Objective:
The aim of this study is to describe the impact of SARS CoV-2 infection on periodontal tissues.
Methods:
In this study, we searched the NCBI, PubMed, Google Scholar, and Research Gate databases. The keywords used were “coronavirus disease” or “COVID-19” or “SARS-CoV-2” with “oral manifestations” and “periodontal,” and we screened the articles using the PRISMA flow diagram.
Results:
Patients with COVID-19 present with a variety of systemic symptoms, as well as manifestations in the periodontal tissues, especially in the gingival area.
Conclusion:
SARS CoV-2 infection can impact periodontal tissues in the form of necrotizing ulcerative gingivitis, aphthous stomatitis, gingival hyperpigmentation, edema and erythema, lichen planus, and aphthous-like ulcers.
1. INTRODUCTION
From December 2019 until now, countries worldwide have been dealing with the new virus, which causes a severe acute respiratory syndrome known as coronavirus disease 2019 (COVID-19) [1]. COVID-19 is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The first spread of this disease occurred in Wuhan, China, in December 2019, and on March 11, 2020, the World Health Organization (WHO) declared the disease a global pandemic [2]. Based on global case reports from the WHO [3], there were 594,367,247 confirmed COVID-19 cases including 6,451,016 deaths as of August 23, 2022.
The spread of SARS-CoV-2 infection can occur either directly or indirectly. Direct transmission begins with the spread of droplets that spread when someone with COVID-19 coughs, sneezes, or talks to other people without wearing a mask [4, 5]. The indirect route of infection occurs through exposure to surfaces and objects contaminated by droplets from an infected person [6].
During dental procedures, dentists, dental assistants, dental hygienists, and patients can be at high risk of cross-infection with SARS-CoV-2 [7]. This may be due to various factors, including the use of dental hand pieces and ultrasonic scales capable of spreading contaminated aerosol droplets or inadequate personal protective equipment (PPE) [8, 9].
Early preventive measures against COVID-19 implemented in dental clinics include assessment of patient’s body temperature using infrared thermometers and questioning of patients about the history of fever, flu, cough, respiratory and recent travel to and from outside regions [10, 11].
Once it is confirmed that a patient has no symptoms or risk factors, dental care can be provided while maintaining standard infection control protocols such as washing hands before and after treatment and using advanced PPE (goggles, face shields, surgical caps, and disposable gowns). It is also appropriate to ask the patient to gargle with 0.2% hydrogen peroxide before treatment and appropriate to use a rubber dam during the procedure [10, 12].
Individuals infected with COVID-19 may experience a variety of clinical symptoms such as fever, dry cough, headache, weakness, diarrhea, shortness of breath, anosmia, loss of taste, and sore throat [13]. Clinical symptoms can be mild, moderate, or severe. In mild to moderate clinical disease states, there is an increase in T cell, CD8, and IgM/IgG levels, while in a severe clinical disease state, there will be an increase in pro-inflammatory cytokines (IL-1β, IL-2, IL-6, IL-7, IL-10, and TFN-α), triggering the occurrence of a “cytokines storm” [14-16].
The severity of SARS-CoV 2 infection, if not treated immediately, can cause various periodontal tissue diseases [17]. A person with periodontal disease can experience damage to the jawbone structure, cause the teeth to fall out of their sockets, pain, and, in a more severe phase, can worsen the systemic condition and lead to death [18, 19].
Based on this background, the authors conducted this systematic review with the aim of evaluating the impact of COVID-19 on the periodontal tissue.
1.1. Coronavirus Disease 2019
COVID-19 is a clinical condition that is characterized by severe acute respiratory syndrome. This virus initially appeared in Wuhan, Hubei Province, China, in December 2019, and quickly spread to all countries, including Indonesia. According to data from the Ministry of Health of the Republic of Indonesia, in Indonesia, there were 6,329,143 confirmed positive cases as of August 24, 2022, of which 6,123,342 had recovered and 157,438 had died [20].
The name coronavirus is from the Latin word “corona,” which means crown or halo as the spike observed on the surface of the virus is shaped like a crown [21]. Coronavirus belongs to the Coronaviridae family (sub family Coronavirinae) and can be classified into four genera, namely Alphacoronavirus, Betacoronavirus, Gammacoronavirus, and Deltacoronavirus [22].
When the virus enters a human body, the spike glycoprotein (a part of the virus), binds to the cell surface of angiotensin-converting enzyme 2 (ACE2), which causes a down regulation of ACE2 receptors and increases the production of angiotensin-2 (AT2). The increase in AT2 production can further increase pulmonary vascular permeability and cause lung injury [5, 23].
An individual who is infected with SARS-CoV-2, begins to experience signs and symptoms of COVID-19 approximately 5 days after the incubation period. These signs and symptoms can last up to 11.5 days. Patients may experience a broad range of symptoms, and the duration of the symptoms may be influenced by the severity of the infection and the age of the patient [24, 25]. Some mild symptoms of COVID-19 include fever, dry cough, and fatigue. Some severe symptoms are shortness of breath, anosmia, ageusia, diarrhea, headache, sore throat, nausea, vomiting, chest pain, and periodontal tissue disease [17, 26].
1.2. Periodontal Tissue
According to Reddy [27], periodontium is formed from two Greek words, namely “peri,” which means “around,” and “odont,” which means “tooth.” Periodontium is defined as the tissue that surrounds the teeth and functions to hold the teeth in place. Periodontal tissue consists of four components, namely the gingiva, periodontal ligament, cementum, and alveolar bone [28].
Recently, WHO reported that healthy periodontitis can be described as a periodontal condition that is free from inflammatory disease and allows affected individuals to perform their usual activities [29]. Periodontal tissue health can be reviewed through several examinations, such as gingival status, gingival index, bleeding on probing (BOP), periodontal pocket depth (PPD), and tooth mobility [27-30].
In general, periodontal tissue disease can be classified into gingivitis and periodontitis [31, 32]. Gingivitis is a mild inflammatory condition that affects only the gingival tissue and does not affect other supporting tissues, and in periodontitis, the inflammation affects the supporting tissues under the gingiva [33]. Periodontal disease is caused by both local factors (bacterial plaque) and systemic factors [34].
2. METHODS
2.1. Research Strategy
This review used the NCBI, PubMed, Google Scholar, and Research Gate databases. The keywords were “coronavirus disease” OR “COVID-19” OR “SARS-CoV-2” AND “oral manifestations” AND “periodontal.” The authors used the PRISMA flow diagram to screen the titles, abstracts, and full text of the articles to ensure that they met the eligibility criteria, retrieved those that met the inclusion criteria, and excluded some reports.
2.2. Inclusion Criteria
The inclusion criteria were (1) articles published from 2020 to 2021, (2) research articles, observational studies, case reports, and case series, (3) articles published in English, and (4) fully accessible articles.
2.3. Exclusion Criteria
The exclusion criteria were (1) articles on congenital systemic disease with and without COVID-19, (2) articles on periodontal disease due to the impact of drug use or due to poor oral hygiene, and (3) articles that discussed the impact of COVID-19 on systemic conditions without providing an overview of its impact on the periodontal tissues.
2.4. Systematic Review Stages
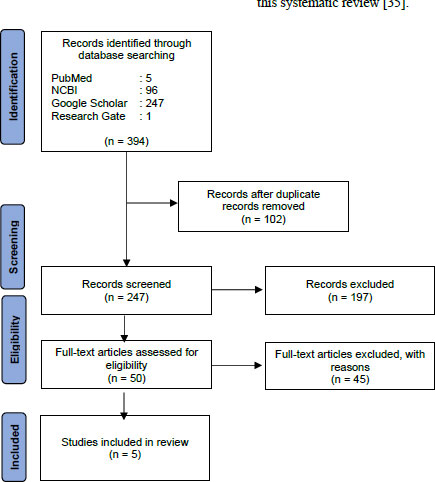
We obtained 349 articles in the first stage of the article searches in the four databases stated earlier. Fig. (1) provides the PRISMA flow chart depicting the article screening process, which resulted in the selection of five articles for inclusion in this systematic review [35].
| Authors/Refs | Publication Year | Source | Article Type | Location | Number of Samples | Gender | Age | Periodontal Tissue Features |
|---|---|---|---|---|---|---|---|---|
| Samoilenko et al. [36] | 2021 | Google Scholar | Case Report | Ukraine, Eropa | 1 | F | 32 years | 1. Generalized erythematous 2. Edematous with ulceration 3. Necrosis of the interdental papillae of the mandibular sextant 4. Gingival sulcus bleeding, without loss of attachment Case diagnosis: Necrotizing Ulcerative Gingivitis |
| Corchuelo and Ulloa [4] | 2020 | PubMed | Case Report | Kolombia, Amerika Selatan | 1 | F | 40 years | 1. Apthous stomatitis in attached gingiva in relation to 34 2. Melanin pigmentation (Gingival hyperpigmentation) |
| Favia et al. [37] | 2021 | Google Scholar | Research Study | Italy, Eropa | 123 | F/M | - | Ulcero-necrotic gingivitis |
| Biadsee et al. [38] | 2020 | PubMed | Case Series | Israel, Asia | 140 | F/M | - | 1. Gingival swelling (gingival edema), n: 2 2. Oral cavity swelling, n: 8 |
| Fidan et al. [39] | 2021 | NCBI | Observational Study | Turkey, Asia | 74 | F/M | 19–78 years | 1. Apthous-like ulcer (n: 4) 2. Erythema (n: 3) 3. Lichen planus (n: 4), Total Lesions in the Periodontal Tissue, gingiva (n:11) |
3. RESEARCH RESULTS
Table 1 summarizes the characteristics of the five studies included in the systematic review.
4. DISCUSSION
The lungs and oral cavity are the main targets of SARS-CoV-2 after it enters the human body. The lungs and oral cavity are involved in the mechanism of virus entry, become a site for virus replication, and induce an inflammatory response [15]. This viral infection, if treated early and properly, can trigger an increase in the secretion of pro-inflammatory cytokines [14, 15].
This excessive and uncontrolled secretion is referred to as a cytokine storm, which impacts patients’ periodontal tissue [40]. This is consistent with a statement by Janani et al.. [39] that elevated pro-inflammatory cytokine levels detected in the gingival tissues of patients with COVID-19. The statement shows that the presence of periodontal disease is a negative effect of COVID-19.
In the five selected articles, the patient had different features of the impact of COVID-19 on their periodontal tissues.
4.1. Necrotizing Ulcerative Gingivitis
The first article was a case report by Samoilenko et al. [35] of a 32-year-old woman with COVID-19 in Ukraine, Europe. The woman reported to the Therapeutic Dentistry Department at Dnipro State Medical University with complaints of severe gingival pain, spontaneous bleeding for two days, and difficulty in chewing. After intraoral clinical examination, the periodontal tissues showed generalized erythematous, edematous gingiva with ulceration and necrosis of the interdental papilla on the mandibular sextant, and gingival sulcus bleeding, without loss of attachment. She was diagnosed with necrotizing ulcerative gingivitis (NUG). Another study by Favia et al. [37] supports the finding that patients with COVID-19 demonstrate features of NUG (they found as many as 7 cases out of 123 patients with COVID-19 in Italy, Europe.
NUG is a periodontal disease with an acute clinical presentation and characteristic features of rapid gingival pain, necrosis of the interdental gingiva, and bleeding. This disease is associated with several risk factors or predisposing factors, such as poor oral hygiene, psychological stress, malnutrition, smoking, and pre-existing gingivitis conditions [41].
4.2. Apthous Stomatitis and Gingival Hyperpigmentation
Another case report by Corchuelo and Ulloa [4] described the effect of SARS-CoV-2 infection on periodontal tissues in a 40-year-old woman in Colombia, South America. Initially, the patient complained of dark discoloration in the gingival area and pain in attached gingiva of tooth 34. After history taking and several examinations, the patient was diagnosed with COVID-19 and had periodontal tissue lesions including apthous stomatitis on attached gingiva of tooth 34 and gingival hyperpigmentation. In addition, the case report by Biadsee et al. [38] that collected information using a web-based questionnaire, showed that of 140 patients with COVID-19, two had gingival edema and eight had oral cavity swelling.
4.3. Apthous-like Ulcer, Erythema, and Lichen Planus
The changes observed in the periodontal tissue due to SARS-CoV-2 infection were mostly in the gingival area, which is consistent with the case report by Fidan et al. [39]. In their report, 74 patients with COVID-19 aged 19–78 years (45.6 ± 12.8 years) who reported to the clinic from April 2020 to October 2020 were observed. The results showed that there were cases of aphthous-like ulcers (n: 4), erythema (n: 3), and lichen planus (n: 4), and most were located in the periodontal tissue, specifically the gingiva (n: 11). The importance of early examination of the oral cavity and of the provision of instructions for maintaining oral hygiene in patients with COVID-19 is to determine the appropriate management of COVID-19 and to help improve the patient’s condition, which will ensure better treatment prognosis [42].
Of the five selected articles, three articles explained how periodontal tissue lesions are managed. In the first article, medications, such as metronidazole 400 mg (taken 3x1 day for 5 days), Iruxol Gum (taken 2x1 day for 7 days), and 0.05% chlorhexidine gluconate mouthwash, were used to manage NUG. After 7 days, post-case management of periodontal tissue lesions showed that the lesions could heal and also instructed to maintain regular oral hygiene [36].
In the second article, the following instruction, were followed to manage aphthous stomatitis and gingival hyperpigmentation: brushing of teeth regularly as a control measure for oral hygiene, changing the toothbrush frequently, soaking the toothbrush head in 5% sodium hypochlorite solution for 30 minutes, and gargling with chlorhexidine gluconate 0.12%; dental teleconsultation was performed after 20 days. The results after these instructions showed that the lesions had healed successfully with no aphthous stomatitis and pigmentation found on the gingiva [4].
In the third article, management for periodontal tissue lesions such as ulcero-necrotic gingivitis included instructions for using hyaluronic acid gel and 2% chlorhexidine gel or mouthwash, which can be used 2x1 day for 14 days. Biopsy under local anesthesia was performed if the lesion did not heal [37].
Periodontal tissue, especially the gingiva, is very important in supporting the aesthetic value of individuals when they smile. Therefore, it requires special attention. Gingival hyperpigmentation, as previously discussed, can greatly interfere with the aesthetic value of an individual’s face. The color of the gingiva depends on the number and size of blood vessels, the thickness and degree of keratinization, and pigmentation in the gingival epithelium. Gingival pigmentation is described by a change in color to purple brown, light brown, or black spots, which are due to melanoblast production [43]. In addition, various conditions of the gingiva, such as aphtous stomatitis, gingival edema, gingival erythema, lichen planus, and aphthous-like ulcers, interfere with the gingiva appearance and function, and can affect the quality of life of such individuals.
In patients with periodontal disease, apart from identifying the main causative factors, it is necessary to identify other risk factors. Treatment must also be performed sequentially, and acute phase treatment must be given immediately to prevent a relapse. In treating periodontal disease, good patient compliance during the treatment phase and maintenance of oral hygiene can guarantee a better and more stable healing process [41].
5. CONCLUSION
Based on this systematic review, we can conclude that COVID-19 has an impact on periodontal tissues by causing NUG, aphthous stomatitis, gingival hyper pigmentation, gingival edema, gingival erythema, lichen planus, and aphthous-like ulcers, although these conditions are not specific to only patients with COVID-19.
LIST OF ABBREVIATIONS
| WHO | = World Health Organization |
| COVID-19 | = Coronavirus disease 2019 |
| ACE2 | = Angiotensin-converting enzyme 2 |
STANDARDS OF REPORTING
The PRISMA guidelines have been followed.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
The authors confirm that the data supporting the findings of this study are available within the article.
FUNDING
The research was supported by Universitas Muhammadiyah Surakarta fund. (Funder ID : 061008) and https://pddikti.kemdikbud.go.id/data_pt/NkQxMjQxNDItRTc5OC00RjYyLTg3NEItQ0U0MzVCNTQwOUYx Awards/Grant number : 609/A.3-III/FKG/VIII/2022.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
The author heartfully thanks and appreciates Prof. Dr. Supriatno., DDS., MDSc., Ph.D, for the helpful discussion and diligence in the correction of the manuscript. The authors thank TopEdit (www.topeditsci.com) for its linguistic assistance during the preparation of this manuscript.
SUPPLEMENTARY MATERIAL
PRISMA checklist is available as supplementary material on the publisher’s website along with the published article.


