All published articles of this journal are available on ScienceDirect.
Effect of Curcumin Suspension and Vitamin C on Dentin Shear Bond Strength and Durability. A Pilot Study
Abstract
Background:
Preserving the strength and durability of dentin bonds is a concern of dental researchers and practitioners.
Objective:
The aim of this study was to investigate the effect of treating etched dentin with vitamin C and curcumin suspension as cross-linking agents on the shear bond strength and durability of a universal dental adhesive.
Methods:
Extracted human third molar teeth were collected. Fifty-six flat coronal dentin surfaces were divided equally into four groups: (1) treated with adhesives; (2) etched with 37% H3PO4 for 10s before application of adhesives; (3) same as group (2), but with dentin treated with 2.5% curcumin suspension for 60s before the application of adhesive; and (4) same as group (3), but with the use of a 20% vitamin C suspension instead of a curcumin suspension. Filtek Z350XT composite was built for all samples. Half of the samples in each group were subjected to thermocycling. Shear bond strength was measured using a universal testing machine. Scanning electron microscopy was used to investigate the dentinal surfaces after treatment.
Results:
Treatment with curcumin suspension preserved immediate and long-term bond strength. Conversely, treatment of etched dentin with vitamin C solution significantly reduced the immediate bond strength, but a significant improvement was observed after aging. SEM showed that both cross-linking agents resulted in narrowing or closing of the dentinal tubules, with an adhesive mode of failure.
Conclusion:
The application of curcumin or vitamin C suspension to acid-etched dentin resulted in different effects on immediate and long-term shear bond strength when using universal adhesives.
1. INTRODUCTION
Adhesive dentistry is widely practiced in the current era. The quality and durability of adhesive resin-dentin bonds are highly influenced by the structural integrity and mechanical properties of collagen fibrils. The penetration of the adhesive resin into the demineralized collagen fibers produced by acid etching is usually insufficient, especially with total-etch bonding procedures, leaving a weak layer of unfilled collagen fibers that are liable to breakdown and degradation [1]. Furthermore, activation of endogenous proteases, known as extracellular matrix metalloproteinases (MMPs) and cysteine cathepsins (CCs), occurs as a result of acid etching of dentin [2]. When activated, these enzymes lead to hydrolytic degradation of demineralized dentin, resulting in hybrid layer degradation and bond deterioration over time [3-5].
Recently, researchers have focused on natural and synthetic dentin bio-modifiers to inhibit hydrolytic enzymes and enhance collagen crosslinking, with the aim of improving dentin bond durability. Natural dentin bio-modifiers show promising results owing to their biocompatibility, availability, and ease of application [6-8]. Of these, natural bio-modifiers are flavonoid-type polyphenolic compounds, such as proanthocyanidins from grape seeds, epigallocatechin-3-gallate, a green tea extract, genipin from gardenia fruit extract, quercetin, hesperidin from citrus fruit, and curcumin extracted from the rhizome of the plant Curcuma longa L, [6, 8, 9]. These natural bio-modifiers have chemical structures that favor their function as antioxidants, cross-linking agents, and inhibitors of MMPs [3, 6, 10-13]. Collagen cross-linking after etching improves the mechanical properties of dentin and enhances resistance to proteolytic degradation [6, 7]. It has been found to improve the durability of resin dentin bonds through the formation of intra- and inter-molecular cross-links [8, 10, 14].
In addition to collagen bio-modifiers, synthetic agents such as glutaraldehyde, carbodiimide, benzalkonium chloride, and ascorbic acid (vitamin C) have been used to enhance cross-linking [12, 13, 15-19]. Ascorbic acid or sodium ascorbate was used as an antioxidant and inhibitor of both MMPs and cysteine cathepsins, which are implicated in the degradation of composite-dentin bonds [3, 8, 10, 11]. Regarding the effect of vitamin C, however, there are contradictory results on immediate bond strength and bond durability [11, 13, 15, 16].
Recently, universal adhesive systems have gained popularity owing to their simplified procedure and low technique sensitivity [20-23]. They are known as multi-mode systems, as they can be applied in self-, total-, or selective-etch modes [22]. It has been claimed that the self-etch mode produces a high bond strength to dentin but not to the enamel. The total-etch mode has been claimed as associated with high bond strength to both enamel and dentin, but postoperative sensitivity is usually high. Therefore, the selective-etch mode has been employed to obtain high bond strength to both enamel and dentin while reducing postoperative sensitivity. In the literature, there is controversy regarding the action of universal adhesives in different modes, and some studies have shown that there is no significant difference between the three modes in terms of bond strength to dentin [24-27]. However, other studies have confirmed a difference in bond strength and bond durability between these modes of action [28, 29].
Therefore, the aim of the present study was to investigate the difference in the immediate and long-term shear bond strength of a universal adhesive used in both the total and self-etch modes. The effect of treating etched dentin with vitamin C and curcumin suspension as antioxidants and cross-linking agents on immediate and long-term shear bond strength was also investigated and compared with other untreated groups. The failure mode was investigated. The null hypotheses (NH) are as follows: NH1 - There is no significant difference in immediate or long-term shear bond strength when universal adhesives are used in total or self-etch modes; NH2 - Treating etched dentin with vitamin C or curcumin suspension has no significant effect on the immediate or long-term shear bond strength of universal adhesives used in the total-etch mode.
2. MATERIALS AND METHODS
2.1. Sample Preparation
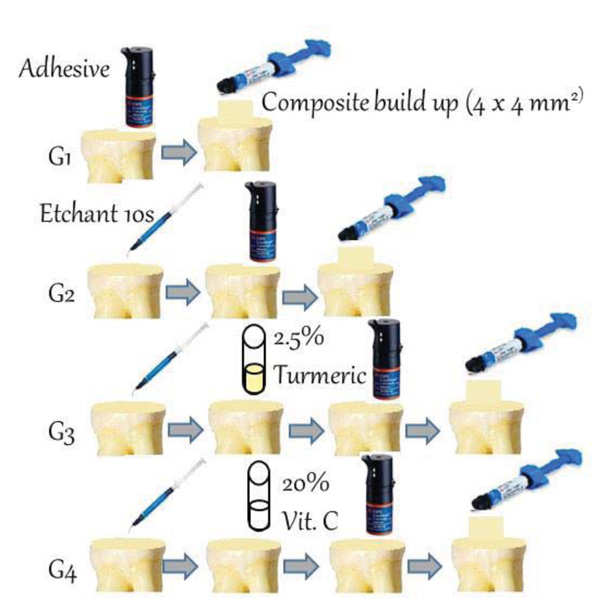
This study was approved by the ethical committee of the Faculty of Dentistry at King Abdulaziz University, Saudi Arabia (Ethical Approval no. 096-09-18). A total of 56 human third molar teeth extracted from an oral surgery clinic at King Abdulaziz University Hospital due to periodontal involvement were used. Teeth were stored in 0.5% chloramine-T solution at 4°C until testing. Roots were sectioned at the cementoenamel junction using diamond metal bond blades (127 × 0.38 × 12.7 mm) mounted on a low-speed diamond saw (Micromet AG, Munich, Germany) rotating at 400 rpm. Water irrigation was used during cutting to avoid heat generation and damage to the dentin. The occlusal surface was flattened perpendicular to the long axis of the tooth, and mid-coronal dentin was used. The teeth were then mounted in a light-cure acrylic resin (Meditray, Promedica Dental Material GmbH, Germany) and smoothed with # 320 and 400 grit silicon carbide papers under water irrigation using a MetaServ 250 grinder polisher (BUEHLER, Lake Bluff, IL, USA) to create a standardized smear layer produced during tooth cutting. The teeth were randomly divided into four groups (n=14 each) according to the treatment used as follows: G1: samples were bonded using a universal adhesive (Single Bond Universal RF, 3M ESPE, USA) in self-etching mode, as per manufacturer’s instructions, wherein the adhesive was applied for 10s on prepared dentin samples. G2: Samples were bonded using a universal adhesive in total-etch mode, wherein dentin was etched with 37% phosphoric acid for 10 s, washed for 10 s, and blot-dried using a mini-sponge prior to the application of the adhesive. G3: Samples were bonded using a universal adhesive in total-etch mode as in G2, then the etched dentin was agitated with a 2.5% curcumin suspension for 60 s immediately before the application of the adhesive; and G4: samples were bonded using a universal adhesive in total-etch mode as in G2, then the etched dentin was agitated with a 20% vitamin C suspension for 60 s immediately before the application of the adhesive. Both curcumin and vitamin C suspensions were prepared by mixing the required amount of powder in distilled water in 10 ml disposable containers (Sterilin Ltd, Newport, UK), followed by agitation for 5 min using a vortex (VM-96A Vortex Mixer, US Plug). Before being applied to dentin, the suspension was vortexed for 60s. The suspension was then applied to the etched dentin surface using a mini-sponge for 60s and then left to air dry for another 60s before application of an adhesive coat. The adhesive was cured for 20 s using blue light (BL) (light-emitting diode curing unit; 3 M ESPE, Elipar, Seefeld, Germany) according to the manufacturer’s instructions. Then, resin composite build-up (Filtek Z350XT composite, 3M ESPE, USA) was carried out using a plastic cylinder 4 mm in diameter and 4 mm in height. The composite build-up was carried out in increments of 2 mm thickness each, and the surface of each increment was pressed smooth and flat using a glass slab. The composite was then light-cured for 20s using a light-emitting diode that was kept at a fixed distance. Fig. (1) shows a schematic representation of sample preparation. Samples in each group were randomly divided into two subgroups (n=7), with one subgroup tested after 24 h, and the second subgroup after 6 months. For 24 h of testing, samples were kept in distilled water after preparation and for 6 months of testing, samples were subjected to 5000 cycles of thermocycling in distilled water at 5°C and 55°C for 30s each using a Thermocycler model THE 1100, SD Mechatronik Gmbh, Germany) [30].
2.2. Measuring Shear Bond Strength
For shear bond strength measurements, samples were sheared with a universal testing machine (Instron model 5944, Canton, MA, USA) using a knife-edge blade at a cross-head speed of 0.5 mm/min until failure. The shear bond strength (MPa) was calculated by dividing the load at failure by the surface area of the sample. After failure, the mode of failure of each sample was examined under a stereomicroscope (Meiji Techno Emz-13TRD-Japan) at a magnification of 50×. The mode of failure was classified as cohesive, adhesive, or mixed failure [8].
2.3. Scanning Electron Microscopy
The morphology of dentin discs treated with 2.5% curcumin or 20% vitamin C suspension after being etched with 37% phosphoric acid was investigated using a scanning electron microscope (AURA100, Seron Technologies Inc., Uiwang, Korea). Etched dentin was used as a control, and samples were coated with gold-platinum using a sputtering machine (SC7620, Quorum Technologies, Laughton, East Sussex, UK).
3. RESULTS
3.1. Shear Bond Strength (MPa)
Fig. (2) shows the 24 h and 6 months shear bond strength of resin composite bonded to dentin after different treatment conditions. Compared to the control (G1), using the universal adhesive in a total-etching mode increased microtensile bond strength. However, this difference was not statistically significant (P >0.05). Agitation of dentin with 2.5% curcumin suspension after etching produced no significant effect on 24-hr shear bond strength. Agitation of dentin with 20% vitamin C produced a significant reduction in 24-hr shear bond strength.
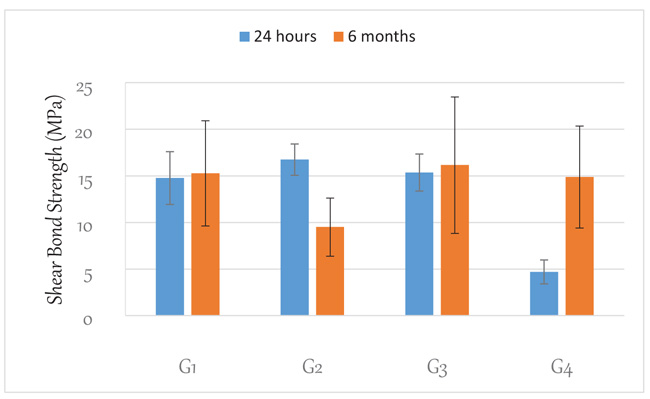
After thermo-cycling for 6 months, the shear bond strength of the samples prepared in the self-etching mode showed no statistically significant difference (p>0.05) from its counterpart at 24 h. For those prepared with the total-etching mode, there was a statistically significant (p<0.05) reduction in microtensile bond strength compared to that at 24 h. The curcumin solution-treated group showed no statistically significant difference (p>0.05) from its counterpart at 24 h. However, the vitamin C-treated group showed a statistically significant (p<0.05) increase in shear bond strength. It was also observed that the samples prepared in the total etching mode showed the lowest shear bond strength.
Regarding mode of failure, after 24h, 67% of the control group (G1) had mixed failure, 33% had adhesive failure, 63% of G2 had mixed failure, and 37% had adhesive failure. All samples in G3 and G4, as well as the thermo-cycled samples, exhibited adhesive failure.
3.2. Scanning Electron Microscopy
Fig. (3) shows the scanning electron microscopy images, with different magnifications, of etched dentin, as well as dentin treated with 2.5% curcumin suspension and 20% vitamin C after etching. Etching of dentin with 37% phosphoric acid for 10 s removed the smear layer and smear plug, as indicated by the presence of opened dentinal tubules (Fig. 3a). Treating the etched dentin with 2.5% curcumin suspension closed the opened dentinal tubules, as shown in Fig. (3b). Treating dentin with 20% vitamin C after etching led to the narrowing of dentinal tubules and closing of the opened dentinal tubules, but not to the same extent as in those treated with curcumin (Fig. 3c).

4. DISCUSSION
In the present study, the shear bond strength of a universal dentin bonding agent used in self-and total-etching, was investigated. In addition, the influence of treating etched dentin with a vitamin C and curcumin suspension on immediate and long-term bond strength was also investigated. Long-term bond strength was investigated after exposing samples for 5000 cycles, which simulates a storage period of 6 months, as stated in the literature [30, 31].
The immediate shear bond strength of the samples bonded using the total-etch mode showed a non-significant increase compared to the self-etch mode. This is in agreement with other studies [22, 28, 29, 32]. However, the difference was not statistically significant.
Additionally, the results after thermo-cycling showed stable bond strength for the self-etch mode, with a significant reduction in the total-etch mode when compared to its counterpart at 24 h. Therefore, the first null hypothesis is partially rejected.
The universal adhesive used in this study is composed of MDP phosphate monomer (10-methacryloyloxydecyl dihydrogen phosphate), dimethacrylate resins, HEMA (2-hydroxy ethyl methacrylate), Vitrebond™ copolymer (methacrylate functionalized polyalkenoic acid), filler, ethanol, water, initiators, and silane [32]. In the total-etch mode, acid etching of dentin removes the smear layer, opens dentinal tubules, demineralizes collagen, and creates enough space for the penetration of the monomer into dentinal tubules and collagen fibers, thus resulting in optimal bond strength. The hydrophilic components of the adhesive resin also facilitate the impregnation of wet dentinal tubules and collagen fibers [28]. However, in the self-etch mode, the acidic monomer demineralizes and infiltrates the dentin without completely removing the smear layer, resulting in shorter resin tags and a thinner hybrid layer. This could explain the slightly lower bond strength observed in the self-etch mode [22]. The reduction in bond strength observed with the total-etch mode in other contradicting studies was explained on the basis that the etching of dentin reduces the calcium and phosphate ions available for chemical bonding with MDP [2].
Treating etched dentin with 2.5% curcumin suspension did not affect immediate bond strength, while a significant reduction was observed with vitamin C application. However, treating etched dentin with 2.5% curcumin suspension resulted in the preservation of the immediate bond strength values while using 20% vitamin C resulted in a significant increase in bond strength when compared to the immediate ones. Therefore, the second null hypothesis is rejected.
Adhesive bond durability is a major concern in the dental applications of bonding systems. In the absence of caries and bacterial ingress, bond durability is mainly affected by the degradation of incompletely resin-infiltrated collagen fibrils caused by the activation of host-derived MMPs [1]. It was proven that acid etching of dentin was one of the activators of these proteolytic enzymes, which could explain the lower shear bond strength observed after aging in the present study. According to the literature [6-8, 12, 13, 16, 33-35], the use of self-etch adhesives and surface treatment of etched dentin with collagen cross-linkers, MMP inhibitors, and antioxidants will improve dentin bond durability. Curcumin, which was used in the current study, is a natural polyphenolic compound known to stabilize the structure of collagen through the formation of multiple hydrogen bonds between collagen polypeptides, and is capable of decreasing its biodegradation [36]. In the present study, the use of a 2.5% curcumin suspension preserved bond strength after aging, which was in agreement with previous studies by Seseogullar-Dirihan et al. [6, 37] wherein curcumin was found to be effective in inhibiting proteolytic enzyme activity and cathepsin K enzyme compared to MMPs. As observed by Seseogullari-Dirihan et al., a significant reduction in MMP-2 and complete inactivation of MMP-9 was observed after application of curcumin [38], with this reduction dependent on the type and concentration of the cross-linking agent [39]. When used as a natural photosensitizer for the disinfection of dentin in combination with LED light, curcumin improved bond strength [40, 41]. The authors related this improvement to its hydrophobicity, polyphenolic nature, anionicity, and ability to bond strongly with calcium ions in dentin [40, 41].
In addition, vitamin C is known for its ability to cross-link collagen I and III, which are antioxidants and inhibitors of MMPs [8, 9, 24]. Antioxidants are capable of counteracting the harmful effects of free radicals, thereby protecting the structural and tissue integrity of the hybrid layer [15]. In the present study, the use of 20% vitamin C significantly reduced immediate bond strength compared to that of the universal adhesive in both modes. Surprisingly, the long-term bond strength of vitamin C-treated dentin improved significantly after aging. This finding is in agreement with other studies [9, 20]. The reduction of immediate bond strength after vitamin C application was inferred from the hypothesis that vitamin C might compete with MDP monomers and form a stable complex with calcium ions and hydroxyapatite, thus reducing the bond with the adhesive system [13]. Another hypothesis was that vitamin C might inhibit the free radicals generated by methacrylate resin, which may reduce their degree of conversion and consequently reduce immediate bond strength. The improved bond strength after aging can be explained by the availability of antioxidants to react with the polymer degradation products even after the initial reaction with the free radicals of polymerization in the process of delayed polymerization, which results in higher resistance to degradation and improved bond strength [9, 20].
The results of the present study were not in agreement with those of other studies [12, 20], which showed that 10% vitamin C for 30s or sodium ascorbate for 10 min preserved the immediate bond strength of self-adhesive systems. This could be explained by the fact that in the present study, a higher concentration of vitamin C was used, which might reduce the available calcium ions for immediate bonding with the resin. After the elimination of degradation products that could possibly occur in the long term, enhanced delayed polymerization might be responsible for the increase in bond strength [8, 10, 20, 21].
SEM showed that the application of the curcumin suspension left some particles on the etched dentin surface without affecting the observed size of the opened dentinal tubules, while the application of vitamin C resulted in obvious narrowing of the dentinal tubules. This indicates that there are different modes of action for both materials, which may explain the difference in the immediate bond strengths between the two materials. This also coincides with the adhesive mode of failure observed in all the samples treated with both materials after immediate testing.
CONCLUSION
Within the limitations of the present study, it can be concluded that an increase in immediate shear bond strength was observed with the introduction of the etching step before the application of the universal adhesive, with a significant reduction in bond strength after aging. The application of 2.5% curcumin suspension to acid-etched dentin does not affect immediate bond strength and can preserve bond durability for 6 months when using a universal adhesive in the etch-and-rinse mode. The application of 20% vitamin C, however, reduced the immediate bond strength, but a significant improvement was observed after aging. This raises concerns regarding bond integrity and microleakage during the aging process. Further investigations are needed to optimize the required surface treatment time and concentrations to be used in order to produce optimum immediate and long-term bond integrity.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was approved by the ethical committee of the Faculty of Dentistry at King Abdulaziz University, Saudi Arabia (Ethical Approval no. 096-09-18).
HUMAN AND ANIMAL RIGHTS
No animals were used in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
Written informed consent was obtained from each participant.
AVAILABILITY OF DATA AND MATERIALS
Not applicable.
FUNDING
This project was funded by the Deanship of Scientific Research (DSR) at King Abdulaziz University, Jeddah, Saudi Arabia under grant no. (G-226-165-1441).
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
The authors would like to acknowledge DSR for providing technical and financial support.
The authors would also like to thank Ebtihal Alahdal and Omar A. Alomem for their participation in conducting the methodology of the present work, as well as acknowledge the “Advanced Technology Dental Research Laboratory” Faculty of Dentistry at King Abdulaziz University.


