All published articles of this journal are available on ScienceDirect.
Healing Process with the use of a New Resorbable Synthetic Membrane
Abstract
Background:
Currently, absorbable membranes tend to be used most frequently for guided bone regeneration. They have many advantages and the most commonly reported complication is early exposure.
Objective:
This retrospective study reports the healing process of soft tissues over a four-week period using synthetic absorbable membranes.
Study Design:
One-hundred and ten cases were included. Soft tissue healing was assessed from anonymized photographs, in accordance with the criteria of the Early Healing Index (EHI) (Watchel et al., 2003). Cohen's Kappa (K) test was used to estimate the reliability of the measures and the variability between the examiners. Chi-squared test and Fisher’s exact test were used to assess the combination of healing outcomes with respect to the type of surgical intervention.
Results:
At 1-week, 81% of the cases showed a Primary Closure (PC) when the membrane was not initially exposed. The score increased to 98% at 4-weeks. Healing at 1-week varied significantly according to the type of intervention, with 73% of primary closure for bone augmentation during implantation, versus 60% for bone augmentation before implantation and 46% for alveolar preservation (Chi-square test, p = 0.049). No statistically significant differences in the healing process were observed between the smoking and non-smoking groups.
Conclusion:
This clinical study shows that the safety and exposure rates of this new synthetic membrane are comparable to the data gathered in the literature concerning non-cross-linked collagenous membranes.
1. INTRODUCTION
There is a vast amount of documented evidence in the literature regarding the safety and efficacy of the use of membranes in guided bone regeneration [1, 2]. The majority of the membranes available on the market are derived from collagen of animal origin, while a limited number of membranes are composed of synthetic polymers [3]. The structure of membranes has evolved over time from a dense single layer to multiple layers having different porosities, which makes it possible to modulate their kinetics of resorption [4]. Resorbable membranes tend to be more advantageous than non-resorbable, for practical reasons and easiness to handle, lack of need for a second operation to remove the membrane, enhanced tissue integration, no need for a second operation to remove the membrane and smaller tendency for exposure [5]. On the other hand, resorbable membranes have certain drawbacks such as barrier effect limitations, space maintenance, and faster resorption upon exposure [2]. Besides, the resorption process of a membrane may interfere with the wound healing of both soft and hard tissues and affect the volume of bone formation [6].
Bone regeneration membranes made of animal-derived collagen have safety and ethical concerns [3]. These collagen membranes are also difficult to handle when wet during surgery and have uncontrolled resorption rates [7]. Since the current generation of GBR membranes is not adequate, a new synthetic resorbable membrane was developed. This new membrane is made of synthetic poly lactic-co-glycolic acid (PLGA), a well-documented and widely used biodegradable polymer [8]. The PLGA 85/15 copolymer formulation was selected based on its degradation time of 4 to 6 months that acts as a barrier for soft tissue and matches the bone regeneration rate. This new membrane has a bi-layered structure with a dense film to prevent gingival fibroblast ingrowth and ensure mechanical function, and a microfibrous layer to support colonization by osteogenic cells and promote guided bone regeneration. The biofunctionality of this membrane was evidenced in different animal models of bone regeneration of critical size defects as well as in patients [9, 10].
There is ample evidence in the literature, to document the healing process of the hard tissues when a membrane is utilised to augment or preserve the volume of the alveolar ridge [11-13]. However, soft tissue healing has been poorly studied even if the major principles of healing in non-oral sites are studied in detail [14, 15]. The way a membrane or bone substitute biomaterials interact with soft tissue healing is even less understood and justified [16, 17]. Soft tissue healing may be compromised when a membrane is used, as the membrane is placed in-between the periosteum of full-thickness flaps and the alveolar process and in most cases, the manipulation of the soft tissues by scoring the periosteum and making vertical incisions that extend over the mucogingival junction to achieve primary wound closure challenges the vascular supply of the flaps.
The aim of this study was therefore to specify the healing process of soft tissues over a four-week period using a synthetic resorbable membrane placement on oral sites.
2. MATERIALS AND METHODS
This observational, retrospective study was based on 110 cases / 108 patients who were referred for a guided bone regeneration procedure between October 2014 and December 2020. All patients were treated in three private offices by three experienced oral surgeons (AH, HLH, FW) for alveolar ridge preservation, bone augmentation before implant placement, and bone augmentation in conjunction with implant placement.
A resorbable synthetic medical grade poly-lactic-glycollic acid membrane (PLGA, Tisseos®, Biomedical Tissues, France) was used in all cases. This membrane has a total thickness of 450 µm and a bilayered structure with a thin dense film intended to face soft tissues and a microfibrous layer for guiding bone regeneration [9, 10]. It maintains its structural integrity and barrier effect for two months and is completely degraded by hydrolysis in 4-6 months [9]. In this study, the the membrane was used in combination with bone grafting substitutes (xenografts, anorganic bovine bone matrix (ABBM). or synthetic beta-tricalcium phosphate (βTCP)).
For each clinical case photographs were taken on day 0, 1 week, and 4 weeks after surgery in a standardized manner with the same equipment (Nikon D800, 100mm macro lens, Nikon macro flash R1C1 R1C1).
A compilation file containing for each case a slide grouping the sutured area during these three stages was used to be noted by three oral surgeons (examiners; HLH, CRB, FW).
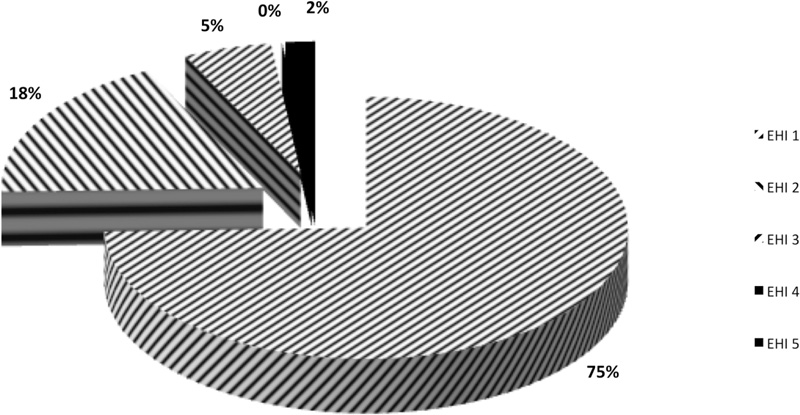
Soft tissue healing was scored by using the early wound-healing index (EHI) [18] by the examiners after calibration. The 5 different index degrees were considered as follows:
(EHI) 1: complete flap closure without fibrin line in the interproximal area.
(EHI) 2: complete flap closure with fine fibrin line in the interproximal area.
(EHI) 3: complete flap closure with fibrin clot in the interproximal area.
(EHI) 4: incomplete flap closure with partial necrosis of the interproximal tissue.
(EHI) 5: incomplete flap closure with complete necrosis of the interproximal tissue.
(EHI) scores of 1, 2 or 3 indicated primary flap closure (PC), (EHI) scores of 4 and 5 indicated secondary flap closure (SC) (Fig. 1). Smoking status was self-reported along with the number of cigarettes smoked a day.
2.1. Statistical Analysis
Cohen's Kappa (K) test was used to estimate the reliability of the measures and the inter-individual variability between the three examiners. Once data’s reliability has been verified, in the event of disagreement between the three examiners, the most frequent answer was chosen.
McNemar Chi-squared tests were used to assess the association between the closure of the surgical site immediately after suturing (initial membrane exposure or no initial membrane exposure) and the 1-week or 4-weeks healing quality (PC or SC).
The Chi-squared test and the Fisher’s exact test were used to assess the association between the healing outcome (PC or SC) and the type of surgical intervention (alveolar ridge preservation, bone augmentation before implantation, bone augmentation in conjunction with implant placement), and the smoking status. Statistical tests were performed with a statistical significance level of 5% (p<0.05) using Systat 10.2 for Windows (SYSTAT Software Inc.).
3. RESULTS
In this study, 110 cases from 108 patients were included. The demographic details of the patients were as follows, 47 women with an average age of 57 ± 14 years and 61 men with an average age of 55 ± 15 years all of the Caucasian origin. Eighteen patients (16.7%) were smokers albeit light users (all < 7 cigarettes a day).
3.1. Reliability of EHI Evaluations from the 3 Examiners
As shown in Table 1, the concordance of observations was very good with a mean agreement of 83.3% (0.614≤ K ≤0.741) at 1 week and 98.8% at 4-weeks with a perfect concordance for two of the three examiners (K=1).
3.2. Clinical Observations
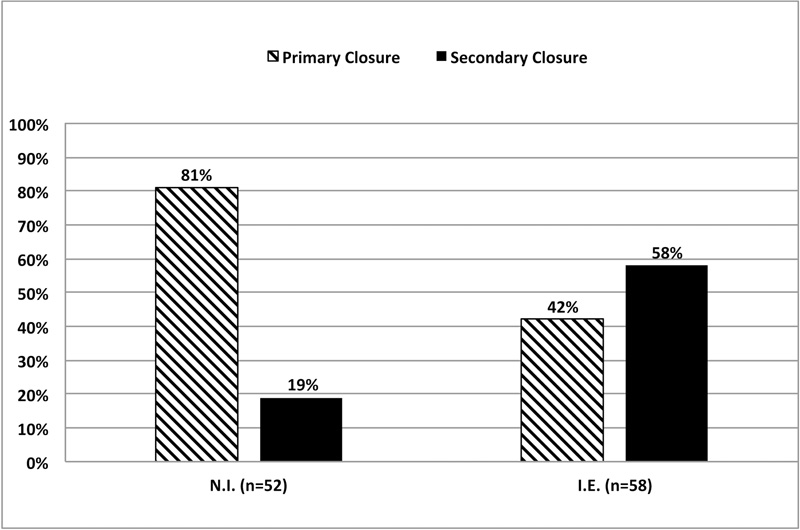
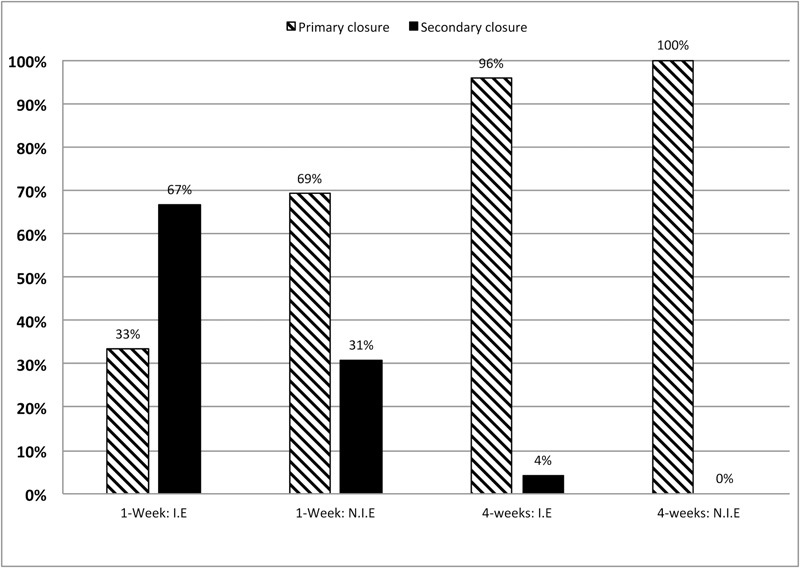
As shown in Fig. (2), at 1-week when the membrane was not initially exposed (n=52), 81% of the cases showed a primary closure (PC). When the membrane was initially exposed (n=58), only 42% of the cases showed a primary closure. Healing mode (PC) or (SC) depends on the initial membrane exposure (Mcnemar’s Chi-square test, p=0.025). Initially exposed sites have shown a lower proportion of 1-week primary closure.
At 4-weeks, 98% of the 110 cases had completed mucosal healing regardless of the initial situation (Fig. 3).
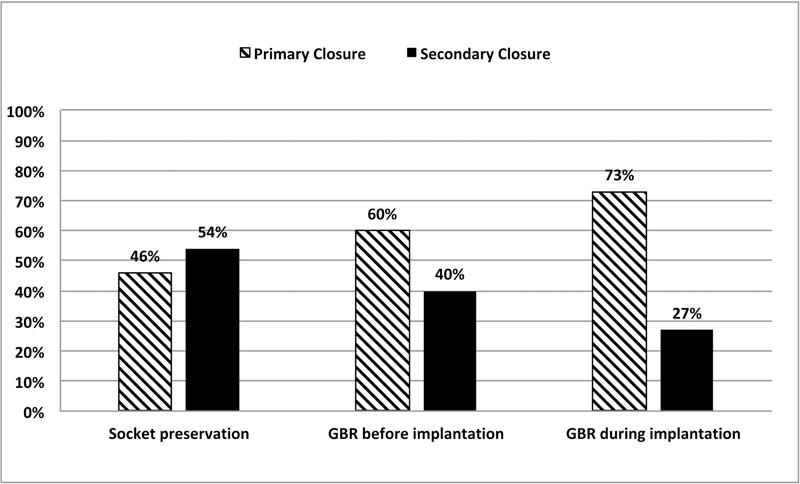
As shown in Fig. (4), the healing outcome at 1 week varied according to the type of intervention and primary wound closure was more frequently observed in the following order: bone augmentation in conjunction with implant placement (73%), bone augmentation before implantation (60%), and alveolar ridge preservation (46%) (Chi-square test, p=0.049).
For alveolar preservation, the aforementioned scores were not the same if we consider the correct closure of the flap during suturing or a possible initial exposure of the membrane.
The 1-week exposure rates significantly decreased from 67 to 31% with an initial exposure versus a correct closure of the flap (Chi-squared test, p=0.036). The 4-weeks exposure rate varied from 4 to 0% according to the same conditions. After 4-weeks, the difference of healing was not statistically different between initial closure of the flap and initial exposure of the membrane (Fisher’s exact test, p>0.05) (Fig. 5).
| Healing Score at 1-Week | ||||
|---|---|---|---|---|
| - | - | - | Inter-investigators Concordance | |
| Observators pairs | % of agreement | Mean of % | Cohen’s kappa K | Agreement |
| Obs.1 X Obs.2 | 81.13% | 83.34% | 0.617 | Substantial |
| Obs.1 X Obs.3 | 87.96% | 0.741 | Substantial | |
| Obs.2 X Obs.3 | 80.95% | 0.614 | Substantial | |
| Healing Score at Week 4 | ||||
|---|---|---|---|---|
| - | - | - | Inter-investigators Concordance | |
| Observators pairs | % of agreement | Mean of % | Cohen’s kappa K | Agreement |
| Obs.1 X Obs.2 | 100% | 98.75% | 1 | Perfect |
| Obs.1 X Obs.3 | 98.13% | unmeasurable | - | |
| Obs.2 X Obs.3 | 98.13% | unmeasurable | - | |
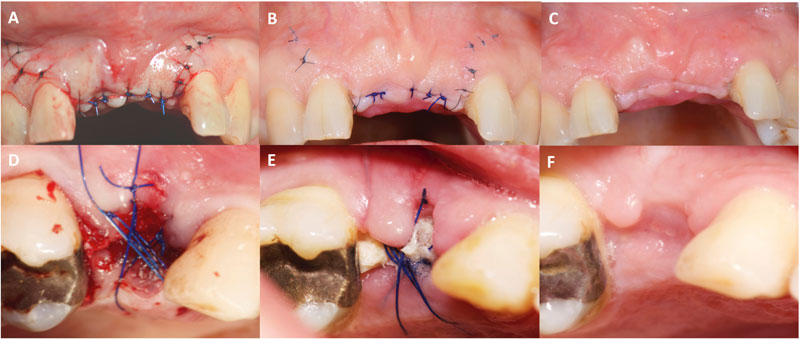
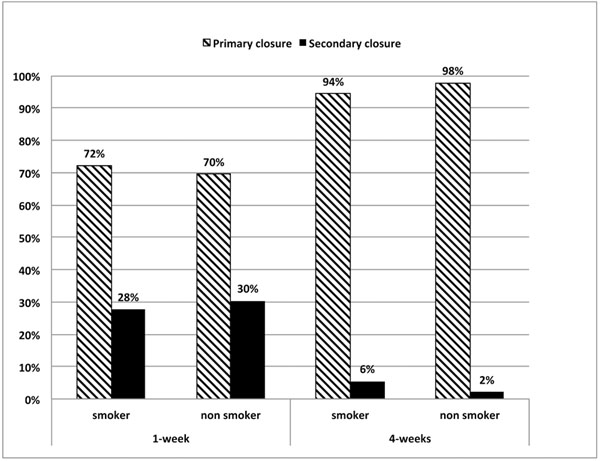
As shown in Fig. (6), smoking albeit light did not affect the soft tissue healing outcome. The proportions of primary closure (72%, 70%) and secondary closure (28%, 30%) between smokers and non-smokers were not statistically different both at 1- week (Chi2, p=0.82) and at 4-weeks (Chi2, p=0.42).
4. DISCUSSION
The principle of guided bone regeneration with or without the simultaneous placement of an implant entails or necessitates the complete coverage of the membrane by the soft tissues provided that flaps are sutured without tension [19-21]. This is not always the case in everyday practice, and this study demonstrated that the healing outcome of the soft tissues with primary closure varied from 81% to 42% at 1-week depending on whether the membrane was initially or not exposed.
At 4-weeks, 100% of the cases of the group with the membrane properly covered demonstrated wound closure. On the other hand, only 2% of cases showed no closure of the soft tissues when the membrane was initially exposed. In these few cases, complete soft-tissue closure was observed at 8-weeks (after the delay of the study period). Interestingly, when the membrane was initially exposed at the alveolar sockets, the membrane appeared to guide re-epithelisation of the site with the mucosa covering the opening of the tissues without losing its integrity, lacking any overt signs of infection or local inflammation of the site or any loss of the graft material as evidenced by the intraoral photographs and confirmed by the patients' reports. Nevertheless, we observed that membrane placement and bone grafting disrupted the natural course of soft tissue healing in 19% of cases at 1-week even when the membrane was properly used.
Although the major steps of soft tissue healing are known [22-24], the closure of gingival tissues has been poorly studied [25]. Considering the great similarities in the healing of the epithelium between the skin and the gingival tissues and the small differences in the chorion between the two origins of tissue, we can argue that the healing phases can be compared between the skin and the gingival tissues [17]. The healing phases have been described as follows: (i) hemostasis (ii) inflammatory phase, (iii) proliferative phase with the development of a granulation tissue and (iv) regeneration phase, including maturation, scar formation, and re-epithelialization. Many cellular interactions, molecular mediators and physical factors modulate these healing phases leading to a first-line healing or delayed healing in particular when the periodontal soft tissues have been damaged. In the case of a persistent inflammatory phase, the granulation tissue would not develop and the remodeling phase of the granulation tissue would delay [26]. The extension of the inflammatory phase induces apoptosis of the cells present in the granulation tissue. Meanwhile, myofibroblasts may persist in preventing both the revascularization of tissues and the reorganization of the extracellular matrix, leading to delayed scarring [15-27].
Different factors may favor inflammation and delayed healing of the gingival tissues. In the hemostasis phase (i) the volume of the graft occupies the space and thus reduces the volume of the blood clot, and in doing so, the number of viable cells that reside within the site. This phenomenon in the cleaning inflammatory phase leads to a reduction in the products of the macrophages degradation, which are important mediators for the conversion towards the proliferation phase (ii) [28]. In the regeneration phase (iii), angiogenesis, being the essential element in the production of the collagen matrix, may be disturbed by the barrier effect of the membrane. Indeed, the membrane isolates the epithelium from the underlying connective tissue and limits the participation of connective tissue in blood capillary proliferation [26, 29]. In the latter part of this regeneration stage, the increase in volume due to the presence of biomaterials can create additional mechanical tension on the tissues. This phenomenon can disrupt the role of myofibroblasts in bringing the wound edges together and the initial closure of the flap [15]. In the regeneration phase (iii), which begins after two to three weeks and ends six months to a year after the intervention, the presence of biomaterials can slow down the remodeling process due to the membrane degradation products and/or the integration of bone substitute [30]. These different factors can explain the differences in results between clinical situations. For instance, the stretching of tissue related to the increase in volume with the use of bone graft before implant placement is generally greater than that observed in the case of bone graft during implantation in respect of GBR indication. (1) The residual tension in the tissues cannot be avoided even by using specific technics of suturing [20, 31, 32]. This tissue tension can counteract the action of the myofibroblasts for bringing together the two edges of the flap and delay its primary closure.
Whatever the exact reasons that disrupt the natural healing of soft tissues, the percentage of exposure of this synthetic membrane (19%) is close to the percentage of exposure observed for membranes made of native collagen with a single layer [33]. Native collagen membranes exhibit good results in tissue integration and biodegradation without exhibiting foreign-body adverse reactions [33]. Their average exposure rates vary between 11% and 32% [34, 35]. However, the major drawback of this type of collagen membrane is that in the proteolytic environment of the oral cavity it loses its integrity in 1-week and fails to occlude the grafted site resulting in loss of the bone graft [36]. Chemical cross-linking of collagen membranes may increase their stability [37] but it has been reported to increase the inflammation in situ in human studies [38] as well as in animal studies [39]. Others evoked that prolonged biodegradation of the membrane is associated with a decrease in tissue integration and vascularization of the tissues whereas an increase of foreign-body reactions [40, 41]. As a result, the exposure rate of cross-linked collagen membranes appears to have increased with reported scores varying from 52.17% to 56% [36]. A recent meta-analysis mentions exposure rates of 28.62% [CI: 14.14 - 49.32) for cross-linked collagen membranes versus 20.74% (CI: 11.16 - 36.19) for non cross-linked collagen membranes [42]. However, spontaneous healing is observed in a few weeks with both types of membranes [43, 44]. In the present study, the rate of exposition of the synthetic membrane was 19% after 1 week. However, its exposition to the oral cavity enzymes did not change its stability and barrier effect as it is observed with collagen membranes that are easily degraded by proteolytic activity. Indeed, the synthetic membrane is degraded by hydrolysis of the polymer chains and not by enzymatic activity.
In our study, tobacco consumption did not have a statistically significant impact on wound healing behavior. This can be explained by the low numbers of smokers and also by the light smoking status (less than 7 cigarettes per day).
A randomized comparative study should be designed to confirm the observations reported in this retrospective study. The gene expression of cytokines in tissue specimens to assess soft tissue inflammation and/or markers of healing during the different healing phases could provide a deeper understanding of the soft tissue healing when a resorbable membrane is used.
CONCLUSION
The soft tissue healing process when using a new synthetic absorbable PLGA membrane was evaluated in three different clinical indications. When the membrane was exposed, the membrane kept its integrity without any loss of the biomaterial granules and without infection. In most cases, the complete soft tissue healing occurred at 4-weeks in the absence of biological complications. This membrane finds its place among the absorbable membranes used in guided bone regeneration procedures by meeting the requirements of patients who – for ethical reasons – do not want medical devices of animal origin.
DISCLOSURE
The authors were fully independent in preparing the protocol, conducting the research, interpreting the results, and drafting the final manuscript.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
Not applicable.
CONSENT FOR PUBLICATION
All patients participated on a voluntary basis and gave their informed consent.
AVAILABILITY OF DATA AND MATERIALS
The data that support the findings of this research are available from the corresponding author [H.A] upon request.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


