Evaluation of Effect of Surgical and Nonsurgical Periodontal Therapy on Serum C-Reactive Protein, Triglyceride, Cholesterol, Serum Lipoproteins and Fasting Blood Sugar in Patients with Severe Chronic Periodontitis
Abstract
Background:
Cardiovascular disease has been associated with multiple risk factors such as dyslipidemia. However, the focus has recently shifted towards some novel risk factors i.e. infection from periodontitis.
Given this background, we aimed to assess the effect of periodontal therapy on some CVD risk factors including Total Cholesterol (TC), Low-Density Lipoprotein(LDL), High-Density Lipoprotein(HDL), Triglycerides(TG) and C-Reactive Protein (CRP). Fasting Blood Sugar (FBS) level has also been measured.
Methods:
Thirty patients (12 male and 18 female) who had severe periodontitis were tested for different blood parameters; namely Total Cholesterol (TC), Low-Density Lipoprotein (LDL), High-Density Lipoprotein(HDL), Triglycerides (TGs), C-Reactive Protein (CRP) and Fasting Blood Sugar (FBS). Enzymatic colorimetric methods were applied to measure all the parameters’ values except for CRP. The first stage of periodontal treatment comprised oral hygiene instruction as well as scaling and root planing. After 1 month, at the next stage, open flap debridement surgery was performed on all 4 quadrants of the mouth. The blood parameters were reassessed and compared with the baseline values after 3 months. Two patients (female) failed to participate in the follow-ups. The Pearson's and Spearman's correlation coefficients were calculated to determine whether changes in laboratory variables are associated with age and average probing depth or not.
Results:
All the assessed parameters related to 28 patients showed mean reduction which proved to be significant for CRP (p=0.011) and cholesterol (p=0.035). Among all parameters, only CRP level was found to have a significant positive correlation with pocket depth. Other blood parameters' relationship with age and probing depth proved to be insignificant.
Conclusion:
Considering the results, periodontal treatment may significantly lower lipid profile serum levels and some inflammatory factors.
1. INTRODUCTION
Cardiovascular Disease (CVD) is regarded as the main cause of myocardial infarction and the leading cause of death worldwide [1]. Age, gender, dyslipidemia, high blood pressure, smoking and diabetes mellitus have been widely accepted as classical CVD risk factors. However, a significant portion of CVD events lacks traditional risk factors which suggest that the classical risk factors are not comprehensive [2]. CVD imposes a great amount of health-related costs and diminishes individual productivity. Because of these problems and additional cost for health provider systems, the search for additional risk factors that may be attributed to these situations has been intensified and recent evidence implicates the causality of chronic infection and inflammation in CVD [3]. The possible role of chronic infection in the development of CVD suggests that atherosclerosis, the major type of CVD might be considered itself as an inflammatory response to injurious factors in all stages, i.e. initiation, growth and complication of the atherosclerotic plaque [4]. One of the earliest signs of atherosclerosis is endothelial dysfunction that is induced by various factors, one of which is the infection from oral bacteria [5].
Periodontitis, as the most prevalent inflammatory state of human beings, is of bacterial origin and leads to the loss of periodontal attachment and alveolar bone [6-8]. When periodontitis is present, bacteria in the form of complex biofilms penetrate into the gingival sulcus and trigger inflammatory reactions by stimulating immunocompetent cells. Virulent bacteria such as Porphyromonasgingivalis, Aggregatibacter actinomycetemcomitans and Treponemadenticola, or the so-called red complex, are held responsible for such robust destructive inflammatory responses [9]. The consequence of such inflammatory chain reactions would be the release of various mediators such as cytokines and chemokines, which contributes to the disintegration of the local periodontal tissues [10]. In addition to host inflammatory response to bacterial antigens, the progression of periodontitis has found to be associated with the interactions and effects of other bacteria, namely “keystone pathogens”, which are fewer in number but are capable of debilitating host defense and promoting microbial community to form resistant pathogenic communities [11]. Nevertheless, in the transition from gingivitis to periodontitis, there are still other determinants that could play a role including poor oral hygiene, smoking, genetic susceptibility, stress and some systemic conditions such as diabetes mellitus.
Nonsurgical periodontal therapy reduces pathogenic biofilm but it could not affect the depth of the pockets and thereby is not considered as effective in the treatment of deep pockets. The existence of periodontal pathogens which belong to red and orange complexes leads to persistence bleeding on probing and pocket depth in these patients. Considering this fact, there is an increasing interest towards adjunctive treatment modalities to enhance periodontal therapy and reduce periodontal microbial biofilm in the long-term. Adjunctive treatment for periodontal therapy has been antibiotic therapy for many years. The use of amoxicillin and metronidazole in addition to scaling and root planning decreased the number of pathogenic microbiota in deep pockets. Complications associated with antibiotic therapy including the detection of resistant pathogens as well as failure to reach a suitable concentration in subgingival area have limited the use of this adjunctive method.
More recently, use of desiccant agent in subgingival area and diode-laser in addition to scaling and root planing has been developed as an adjunct for periodontal treatment. It appears that subgingival microbiota changed in quantity and quality after periodontal treatment. However, recontamination of the treatment sites has reported. For this reason, a full mouth disinfection protocol has been developed. Though, many researchers failed to show advantages for this modality.
With the advent of new methods and techniques such as Polymerase Chain Reaction (PCR) and 16-S ribosome, subgingival microbiota could be better investigated from the clinical and microbiological point of view. However; anaerobic pathogens can be detected and increased at about one-year post-treatment. This signifies the importance of maintenance program for periodontitis patients and the need for adjunctive treatment such as surgical periodontal therapy [12-14].
There is a growing evidence that periodontitis has a direct or indirect correlation with some systemic conditions including diabetes and atherosclerosis, on the grounds that they share the same etiological factors [15]. This association could be readily explained by the possible underlying mechanisms such as direct infection, systemic infection, etc [16]. Different types of oral microbes have been detected in the atherosclerotic plaque, which implies that these microorganisms might be involved in the accumulation of atheromatous plaque on the endothelial surface [17]. Notably, there is sufficient amount of evidence substantiating the presence of periodontal pathogens on carotid endarterectomy specimens as well as their role in atheroma formation [18, 19]. In addition, disparate results show periodontitis causes dyslipidemia. However, different articles have yielded contradictory results and there is still much debate over the potential relationship between periodontitis and dyslipidemia [20, 21] In order to evaluate the effects of periodontal therapy on CVD, many studies have been done and mostly show its effect on reducing inflammatory markers as they are considered novel CVD risk factors rather than the classical ones, i.e. lipid levels [22]. On the other hand, some studies indicate modest or no effect of periodontal therapy on cardiovascular disease [23, 24].
To date, several studies have investigated the systemic effects of periodontal therapy. However, there is still a paucity of conclusive data regarding the possible relationship between cardiovascular disease and periodontal treatment. Therefore, the aim of the present study was to assess this correlation through assessing the effect of periodontal therapy on some classical CVD risk factors including Total Cholesterol (TC), Low-Density Lipoprotein (LDL), High-Density Lipoprotein (HDL), Triglycerides (TG) and C-Reactive Protein (CRP). Fasting Blood Sugar (FBS) level has also been measured and finally, the correlation of periodontal pocket depth with the baseline blood factors has been investigated.
2. MATERIALS AND METHODS
This clinical trial was carried out in the dental research center of Mashhad University of Medical Sciences. Among the individuals referred to the Department of Periodontology of Mashhad Dental School, 30 patients (12 male and 18 female) who had severe chronic periodontitis, based on the 1999 AAP classification, with no diagnosed systemic diseases were enrolled in the study. The patients’ age ranged from 18 to 56 with an average of 36.64. The exclusion criteria were current (including the past 1 month) use of antibiotics, anti-inflammatory or lipid-lowering drugs as well as having diabetes, cardiovascular, cerebrovascular, hereditary storage diseases, malignancies, smoking patients or any other systemic diseases. All patients signed an informed consent form and the ethics committee of Mashhad University of Medical Sciences reviewed and approved the study design.
Complete radiographic examination was conducted and all teeth were examined at six sites for attachment level and probing depth with Hu-Friedy Williams probe and the average and changes in full-mouth probing depth were recorded for each patient.
At baseline estimation, blood samples were collected and the following parameters were assessed:
- 1-TC (Total Cholesterol)
- 2-LDL (Low-Density Lipoprotein)
- 3-HDL (High-Density Lipoprotein)
- 4-TGs (Triglycerides)
- 5-CRP (C-Reactive Protein)
- 6-FBS (Fasting Blood Sugar)
All parameters' levels were measured by enzymatic colorimetric methods except CRP level. TC and TGs were measured by an enzymatic Trinder reaction. Cholesterol fractions were estimated by a homogenous enzymatic colorimetric method and fasting plasma glucose concentration was measured using the Trinder glucose activity test. Immunoturbidimetric assay by an autoanalyzer was utilized to determine the level of hsCRP which is based on the reaction between CRP and its polyclonal antibody. Afterwards, the initial periodontal therapy including full-mouth scaling and root planning, oral hygiene instruction, application of 0.2% chlorhexidine rinses twice daily for three weeks, and removal of other plaque-retentive factors such as overhanging restorations and defective subgingival crown margins was conducted. One month after the initial therapy, all patients were reassessed for the O'Leary plaque index reduction to less than 20% and were scheduled for open flap debridement surgery in all 4 quadrants of the mouth according to their needs. Treatment sessions had 3-week intervals. Surgical flaps were repositioned and sutured using 3-0 silk suture and a periodontal dressing (Coe-pack) was applied to cover the surgical site. Postsurgical care included instructing patients to rinse 0.2% chlorhexidine twice daily and prescription of acetaminophen 325 mg four times a day for one week. Dressing and sutures were removed one-week post-surgery. In case, patients reported discomfort that quadrant was repacked for an additional one week. Followup appointments and supportive therapy were continued for all patients for a period of three months following the completion of active periodontal treatment and finally, blood parameters were reassessed. Two patients (female) quit the follow-up visits and were excluded.
During the maintenance sessions, we assessed the probing depth and attachment level as well as bleeding on probing, as markers for treatment response. All data were measured by a single examiner.
2.1. Statistical Analysis
Data were analyzed using SPSS 13 (SPSS Inc., Chicago, IL, USA). The Shapiro-Wilk test was used for assessing the normal distribution of blood parameters' values, taken at baseline and after three months follow-up. Only CRP and FBS were not normally distributed. Thus, in order to test the mean difference, the paired-t-test was used for data with normality and the Wilcoxon test was used for those with deviations from normality. A secondary analysis was also performed to evaluate whether changes in laboratory variables are associated with age as well as the average probing depth or not. The Pearson's and Spearman's correlation coefficients were calculated to understand linear or monotonic relationship. The significance level was set at p<0.05.
3. RESULTS
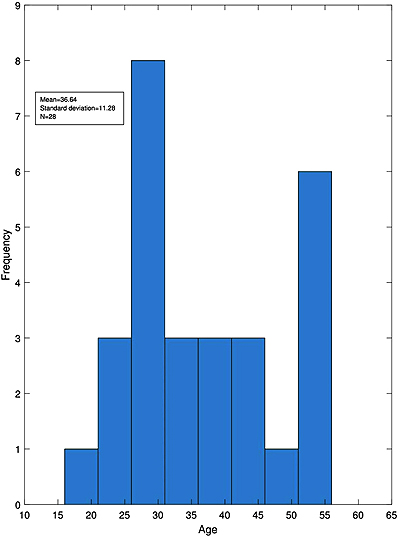
Of all 30 participants included in our study, 28 patients (12 male and 16 female) attended all the visits. As illustrated in Fig. (1), their age ranged from 18 to 56 years and the mean age of the patients was 36/64 ± 11/28.
Table 1 summarizes changes in blood parameters of all individuals before and after surgical and nonsurgical periodontal therapy. All parameters showed a mean reduction, specifically CRP (p=0.011) and TC (p=0.035) which exhibited significant change.
Table 2 shows the correlation of age and the differences in probing depth with the mean change of blood parameters' values. Only CRP had a significant positive correlation with changes in full-mouth probing depth, that is, the deeper the periodontal pocket, the higher the CRP level detected. The strength of other blood parameters' relationship with age and probing depth was considered to be negligible.
| - | Before Treatment | After Treatment | |||||
|---|---|---|---|---|---|---|---|
| Parameter | Range | Median | Geometric Mean±SD |
Range | Median | Geometric Mean±SD |
p-value |
| CRP ,mg/lit | 0/00 to 3/40 | 0/0000 | 0/52 ± 0/93 | 0/00 to 3/00 | 0/0000 | 0/39 ± 0/73 | 0/011 |
| TG ,mg/dl | 64/00 to 225/00 | 150/0000 | 135/46 ± 49/33 | 65/00 to 213/00 | 129/0000 | 126/30 ± 40/43 | 0/245 |
| CHOL ,mg/dl | 107/00 to 233/00 | 187/5000 | 184/71 ± 32/80 | 112/00 to 233/00 | 180/0000 | 178/82 ± 31/81 | 0/035 |
| LDL,mg/dl | 58/00 to 143/00 | 112/0000 | 110/43 ± 20/54 | 62/00 to 150/00 | 106/0000 | 107/07 ± 26/82 | 0/446 |
| HDL,mg/dl | 33/00 to 68/00 | 48/5 | 48/02 ± 8/028 | 34/00 to 68/00 | 52/0 | 51/91 ± 10/87 | 0/106 |
| FBS,mg/dl | 71/00 to 249/00 | 103/0000 | 116/36 ± 40/06 | 71/00 to 205/00 | 100/0000 | 113/54 ± 41/60 | 0/554 |
| - | Age | Probing Depth | ||
|---|---|---|---|---|
| Parameter | r(s) | P value | r(s) | P value |
| CRP | 0/341 | 0/076 | 0/604 | 0/029 |
| TG | -0/087 | 0/659 | 0/0193 | 0/527 |
| TC | 0/175 | 0/374 | -0/174 | 0/569 |
| LDL | 0/075 | 0/703 | 0/232 | 0/445 |
| HDL | 0/287 | 0/139 | -0/076 | 0/806 |
| FBS | -0/114 | 0/564 | -0/101 | 0/744 |
4. DISCUSSION
To date, several studies have investigated the level of classical CVD risk factors in periodontitis patients. Among those, the most relevant one seems to be the inflammatory marker “CRP”. It has been proposed that atherosclerosis is actually initiated and aggravated mostly by an inflammatory process since nearly half of the CVD events happen in individuals without high classical risk factors. The new association between inflammation and atherosclerosis raised a great deal of interest regarding the effect of inflammatory diseases therapy on reducing CVD risk factors. Inflammatory diseases particularly chronic ones came under investigation and among those was chronic periodontitis. Given this background, the present study investigated the effect of periodontal therapy on reducing CRP inflammatory marker in apparently healthy periodontitis patients. Besides, the effect of surgical and nonsurgical periodontal treatment on less associated factors such as TC, LDL, HDL and TG has been studied.
Based on our study, only CRP did have a significant positive correlation with average full-mouth probing depth, that is, the deeper the periodontal pocket, the higher the CRP level detected. All the assessed parameters related to 28 patients showed reduction which proved to be significant for CRP (p=0.011) and cholesterol (p=0.035). Among all parameters, only CRP level was found to have a significant positive correlation with the pocket depth. Other blood parameters' relationship with age and probing depth proved to be insignificant.
Surgical treatment was selected as the periodontal intervention in this study because at baseline visit, after evaluating participants’ periodontitis status, it seemed more reliable to plan thorough periodontal treatment. Moreover, there was not enough evidence about the effect of surgical periodontal therapy on this issue; nearly all previous studies have assessed the relationship of conservative treatment.
CRP level is considered to be a reflection of one's inflammatory state. However, as a weak point, CRP does not necessarily show dose effect or causality relationship with periodontitis status. In two systemic reviews [25, 26], the mean CRP value has been above 2.1mg/l and 1.0 mg/L in only periodontitis patients are a few articles that do not show any increase in serum inflammatory markers in periodontitis patients [27]. Similarly; in our study, patients had normal CRP level although they had chronic periodontitis (mean CRP level=0.53 mg/l).
Reduction in the CRP level has been detected in several studies following the completion of periodontal treatment. For instance, in Paraskevas et al., systemic review, the CRP level between pre- and post-treatment for both test and control groups showed minor reduction with a 0.50 mg/L difference [25]. Likewise, in Ryan T. Demmer et al., meta-analysis, the final CRP values were significantly lower due to the active periodontal treatment. In our study, the mean decrease was about 0.13 mg/l after 3 months of treatment (P=0.011). This inconsiderable level of decrease may be because of the initial low level of CRP at baseline. It has been noticed that in individuals with higher CRP level at baseline, this level of decrease has been higher. In Gupta et al., study, the same surgical intervention was conducted, those with higher CRP level at baseline presented a higher decrease in the end [28]. Notably, in periodontitis patients with cardiovascular diseases, the CRP level at baseline has been higher [29]. Such findings highlight the importance of periodontal therapy in higher CRP level patients particularly individuals with cardiovascular diseases.
It is important to mention that the average decrease in the CRP level following periodontal treatment is considered insignificant as compared with the main ways of CRP decrease including diet, exercise, blood pressure control, and smoking cessation. A recent systemic review has stated modest or no effect of periodontal therapy in systemic CRP reduction, although this seems to be due to the limited sample size in the majority of the studies they have analyzed [30]. This was similarly a limitation in our study. However, Long-term treatment studies evaluating the effect of periodontal treatment on CRP values in relation to healthy controls are scarce.
Paradoxically, there are more mixed findings about lipid profile and periodontitis status as compared with the CRP level and periodontitis. Periodontitis patients are more susceptible to develop hyperlipidemia theoretically [31-33]. But practically, there are contradictory results on this issue and the question whether periodontal therapy can improve one’s lipid profile has received different answers [34-37]. In Shruti et al., study [38], all lipid levels significantly improved except for HDL after conducting periodontal therapy, while in D’Aiuto et al., study, there was no improvement in lipid fractions [37]. In the present research, surgical and nonsurgical periodontal treatment was carried out which resulted in all lipid-related parameters’ improvement; however, the only statistically significant one was the change in total cholesterol. A possible explanation for these contrary results might be that strong confounding factors such as one’s diet, physical activity habits, socioeconomic conditions and stress persist during studies’ course and greatly affect the results.
There are studies which indicate that periodontitis patients show higher blood glucose level [39, 40]. In order to evaluate the hypothesis, we also evaluated alterations in fasting blood glucose level of patients before and after periodontal therapy and the result was not significant.
Finally, we evaluated the association between periodontal pocket depth and some parameters, namely gender and baseline blood factors’ level. The latter proved to be only statistically significant for CRP. The higher the CRP at baseline, the deeper the periodontal pocket. The result was in accordance with several previous studies, highlighting that patients with a more advanced periodontitis status have possibly higher CRP level and accordingly, conducting periodontal therapy may be more essential in these patients particularly if they have concurrent comorbidities like cardiovascular diseases and diabetes [41, 42] There was also no association between participants’ gender and periodontal pocket depth, although it has been proposed that periodontitis is more common among women which is possibly due to the additional role of sex hormones [43].
CONCLUSION
Considering these results, periodontal treatment may decrease lipid profile serum levels as well as inflammatory markers like CRP. Nevertheless, the extent of the effect that this reduction in CRP, as a predictor of CVD events, might have on the prevention of cardiovascular diseases is yet to be determined. Likewise, further research is required to shed more light on the lipid-lowering effects of periodontal treatments. Future studies with larger sample sizes are recommended to be focused not only on the systemic effects of periodontal treatment, but also on the association of its outcomes with the degree of cardiovascular disease prevention, and eventually, whether periodontal therapy and maintenance of periodontal health will reduce the risk for such events.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study design was approved by the Ethical Committee of Mashhad University of Medical Sciences. The reference number is IR.MUMS.REC.1387.950
HUMAN AND ANIMAL RIGHTS
No animals were used in this research. All research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2008.
CONSENT FOR PUBLICATION
All patients signed an informed consent form.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
The authors would like to thank Dr. Farid Shiezadeh for his support and assistance with this project.


