All published articles of this journal are available on ScienceDirect.
A Comparative Study of Root Coverage using OrACELL™ Versus Subepithelial Connective Tissue Graft: A Randomized Controlled Trial
Abstract
Background:
Gingival recession is defined as a mucogingival deformity that includes the apical displacement of the marginal soft tissues below the cemento-enamel junction, loss of attached gingiva, and exposure of root surfaces. The present study aims to compare root coverage outcomes between autogenous Connective Tissue Graft (CTG) and decellularized human dermis (OrACELL™) in areas of facial gingival recession.
Methods:
Twenty-four non-smoking, healthy patients, with 2mm or greater facial gingival recession at a minimum of one site that is classified as Miller Class I, II, or III recession defects were included. Patients were randomly assigned to either control (CTG) or OrACELL™ (test) groups, which were treated with identical surgical techniques. All root coverage clinical parameters were evaluated at baseline, 3, and 6-months.
Results:
Eleven patients received CTG while 13 patients received OrACELL™; 23 of the 24 total patients had Miller Class III defects. Baseline mean Vertical Recession (VR) (CTG = 3.27±0.68 mm, OrACELL™ = 3.50±0.89 mm) and Clinical Attachment Level (CAL) (CTG = 4.86±0.74 mm, OrACELL™ = 4.73±0.90 mm) showed no significant difference between groups. At 6 months, mean VR (CTG = 0.59±0.70 mm, OrACELL™ = 1.19±1.07 mm) significantly decreased in both groups, whereas CAL (CTG = 1.90±1.00 mm, OrACELL™ = 2.42±1.17 mm) significantly increased in both groups. Differences between group means were not statistically significant.
Conclusion:
VR and CAL improved significantly in both the CTG and OrACELL™ groups from baseline to 6 months post-operatively, with no significant differences between groups regarding VR or CAL over the course of the study. In Miller Class III recession defects, OrACELL™ provided a viable alternative to CTG with similar results.
1. INTRODUCTION
Gingival recession is a condition that affects an extensive range and percentage of people. In an evaluation of epidemiological data, Kassab found that 50 percent of people between the ages of 18 to 64 have gingival recession of at least 1 mm [1]. Furthermore, 88 percent of those over 65 have lost gingival attachment on at least one site, concluding that the trend for increasing recession appears to progress with age. It is likely that gingival recession will become an increasing concern for patients, given the aging and esthetically-minded populations of today [2].
The main indications for root coverage procedures include esthetic demands, root sensitivity, preventing future progression of recession, and replacing class V carious lesions [3]. According to Miller, the ultimate goal of these procedures is root coverage that reestablishes both esthetics and function with a sulcus exhibiting no bleeding on probing and less than 2 mm probing depth [4].
Numerous techniques have been used to treat gingival recession. The current gold standard for root coverage is the subepithelial Connective Tissue Graft (CTG) technique, which requires tissue harvested from the palate and placed over an appropriate recession defect with coronal advancement of the flap over the donor graft [5]. This procedure requires a donor and recipient site leading to greater patient discomfort, potential bleeding issues post-surgically and increased surgical time [3]. However, given the reluctance of patients to tolerate additional surgical sites and the necessity to treat multiple recession defects, periodontists have turned to allograft substitutes.
It was demonstrated in a study by Harris that Acellular Dermal Allograft Materials (ADM) like AlloDerm™ are viable substitutes for CTG in the treatment of gingival recession defects [3]. Within the current body of literature, support of the efficacy of AlloDerm™ is abundant. AlloDerm™ has been successfully utilized in treatment for a range of procedures including: (1) increasing keratinized tissue width, (2) root coverage, (3) guided tissue regeneration, and (4) guided bone regeneration. New decellularized dermis products have recently been introduced as another alternative. In contrast to AlloDerm™, little research has been conducted on the efficacy of decellularized dermis.
The manufacturer of a decellularized dermis material, LifeNet Health, promotes that OrACELL™ retains native growth factors along with collagen and elastin [6]. Therefore, OrACELL™ is potentially an excellent candidate for use in guided tissue regeneration, guided bone regeneration, and the treatment of oral soft tissue recession [6]. OrACELL undergoes the patented MATRACELL process which includes the following 4 steps: 1) Decellularization with an anionic, non-denaturing detergent, N-Lauroyl sarcosinate, to remove donor cells and Benzonase®, a recombinant endonuclease, to degrade DNA; 2) Rinsing with water to remove residual reagent and donor cell remnants; 3) Preservation with a solution that is comprised of USP Glycerol and USP Saline to allow for room temperature storage; and 4) Sterilization with low-dose gamma irradiation at low temperatures. This process achieves a final Sterility Assurance Level (SAL) of 10-6.
This proposed study was designed to compare the differences in clinical parameters of root coverage surgeries using OrACELL™ versus CTG in an adult population presenting with gingival recession.
2. MATERIALS AND METHODS
2.1. Patient Population
Non-smoking, healthy patients undergoing treatment in the Graduate Periodontics clinic at Texas A&M University College of Dentistry were chosen for the study based on their existing recession. The inclusion criteria were as follows: (1) patients must be between 18 and 80 years old, with recession present in one quadrant as either single or multiple buccal vertical recession sites; (2) the defect must be at least 2 mm in length, (measured from the CEJ to the midfacial gingival margin) and classified as either Miller Class I, II or III; and (3) the study is limited to vital and nonvital incisors, canines, and premolars. If teeth adjacent to the site to be treated had recession as well, they were included in the grafting procedure but not included in the data collection. Plaque control defined as modified O’Leary Index of 85% was established before surgical intervention [7]. Only sites with probing depths of 3mm or less and no bleeding on probing were accepted for surgery.
The exclusion criteria were as follows: (1) subjects who smoke more than ten cigarettes per day or use nicotine replacement therapy; (2) previous history of surgery performed at surgical sites included in the study; (3) subjects who have uncontrolled or poorly controlled systemic conditions that could compromise or contraindicate periodontal surgery; (4) non-English speakers; (5) pregnant or lactating females; and (6) subjects taking immunosuppressant medications. All patients were required to sign an informed consent in order to participate in the study.
To establish controls, all patients received a dental examination, dental prophylaxis when indicated, and oral hygiene instructions to address habits related to disease etiology and to demonstrate effective plaque control prior to surgical procedures. Teeth in the quadrant involved in the study received periapical radiographs, if not previously available, and were tested for vitality. Within each patient, the quadrant with qualifying recession was randomly assigned to either the experimental or control group using a randomization table.
Random numbers were assigned to each patient as they were recruited for the study based on a potential study population of thirty. The patients assigned to the test group were treated with OrACELL™. The patients assigned to the control group were treated with an autogenous connective tissue graft. Factors such as oral hygiene, compliance, and varied healing responses were therefore controlled by randomization.
Twenty-four patients (15 females, 9 males, aged 26 to 78 years; mean age: 50.5 years) had one defect treated, for a total of 24 defects. All twenty-four patients that enrolled in the study and completed the study, between April 2015 and May 2016, with no dropouts. There were 23 Miller Class III defects and one Miller Class I defect which consisted of 12 incisors, 4 canines, and 8 premolars. Eleven sites were treated with CTG (control) while thirteen sites were treated with OrACELL™ (test).
2.2. Clinical Parameters
The following clinical parameters were evaluated: Vertical Recession (VR) measured as the distance from the Cemento-Enamel Junction (CEJ) to the Free Gingival Margin (FGM) in mm; Horizontal Recession (HR) measured at the CEJ in mm; Probing Depth (PD) on the midfacial aspect measured as the distance from the FGM to the bottom of the sulcus in mm; Clinical Attachment Level (CAL) measured as the distance from the CEJ to the bottom of the sulcus in mm; presence or absence of Bleeding On Probing (BOP) on the midfacial aspect; Papillary Height (PH), defined as the distance from the tip of the papilla to the base of the papilla at level of the CEJ; Papillary Width (PW) measured at the base of the papilla at the CEJ level for both the mesial and distal papilla adjacent to the recession; and the width of Keratinized Tissue (KT) from the FGM to the mucogingival junction in mm.
The distance from the CEJ to bone at the midfacial aspect was measured after flap reflection. All measurements were made with a UNC periodontal probe (15 mm). Clinical parameters VR, HR, PD, CAL, BOP, PH, PW, and KT were recorded prior to surgery (baseline) and again at 3 and 6 months post operatively. When the CEJ was obliterated by a non-carious cervical lesion, the most coronal aspect of a non-carious cervical lesion served as a reference point for VR measurements. All parameters, including Miller recession classification, were evaluated by 2 blinded Board Certified Periodontists (GRG & JAR).
2.3. Surgical Procedure
The surgical procedure was identical for both test and control groups with the only difference being the graft material. On the day of surgery, the exposed root surfaces were thoroughly root planed by means of curettes to ensure a smooth biocompatible surface. This was performed prior to flap reflection to ensure soft tissue adhesion to the previously exposed root surfaces, no further root conditioning mechanical nor chemical was performed. The sites were rinsed with 0.9% normal saline solution. The surgical site was anesthetized with (2%) lidocaine HCl with 1:100,000 epinephrine. The surgery began with incisions described by Zucchelli, which consist of making “horizontal incisions of an envelope flap consisting of oblique submarginal incisions interdentally continuous with intrasulcular incisions at the recession defects” [8]. Full thickness flaps were reflected approximately 3mm apical to the alveolar bone crest at each defect site using a periosteal elevator. Starting at the coronal aspect, a split-thickness flap dissection was performed and extended mesially, distally and apically to facilitate adequate mobility for the coronal advancement of the flap without tension. The anatomic papillae coronal to the incisions were then de-epithelialized to ensure a good vascular connective tissue bed. In the test sites OrACELL™ graft was trimmed to fit in the recession area. The graft was adjusted to completely cover the defect and the superior graft margin was positioned at the CEJ, while the inferior and lateral borders of the graft were extended at least 3 mm beyond the osseous defect margins. OrACELL™ was placed against the root surface and sutured using the double sling suture technique with 5-0 chromic gut suture. The flap was coronally positioned to cover the entire allograft and papilla area and sutured using the double sling suture technique with additional interrupted 6-0 Prolene® sutures, as needed, to ideally position the papillae. Flap closure was accomplished with 6-0 Prolene® sutures (Fig .1).
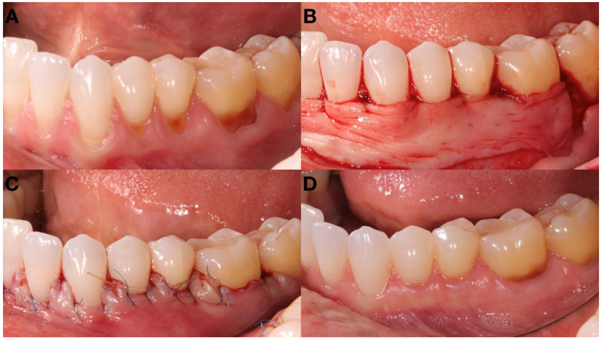
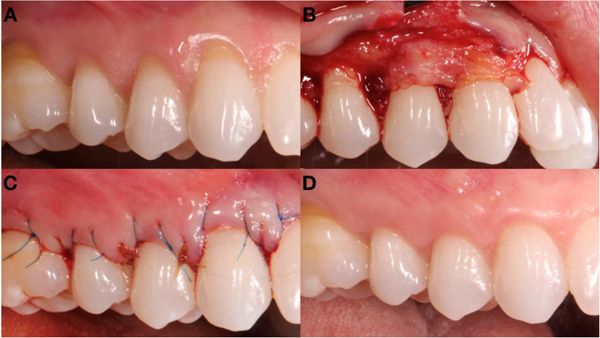
The control group underwent the same surgical procedure as the test, substituting OrACELL™ for an autogenous Connective Tissue Graft (CTG). In the control group, the CTG was harvested from the palate in a surgical approach described by Langer [5] taking tissue between the cuspid and first molar sites and apically to the marginal tissue on the palate by 3 mm. The CTG was thinned to 1.5 - 2 mm thickness prior to placement at the recipient bed. The suturing technique was the same as described above for the test sites (Fig. 2).
2.4. Post-Surgical Care
After the procedure, patients were prescribed 500 mg amoxicillin, three times daily for 7 days, or 300 mg clindamycin, three times daily for 7 days if the patient was allergic to penicillin. In addition, 50 mg Tramadol was prescribed for post-operative analgesia. Ice pack application was used immediately after surgery on an intermittent basis for the first 3 to 4 hours at the surgical sites. All patients were advised to discontinue mechanical oral hygiene measures in the surgical areas for 2 weeks and avoid trauma to the surgical sites. A liquid diet was recommended for the first 48 hours followed by a soft diet for the next 2 weeks. Chlorhexidine gluconate 0.12% rinse was prescribed for 2 weeks with instruction to rinse on a daily basis. Gentle tooth brushing was resumed at 2 weeks using a roll technique until the 1-month follow-up appointment. Professional plaque control was performed at the 1 week, 1 month, 3 month, and 6 month recall appointments.
This study was conducted in accordance with the Helsinki Declaration of 1975, as revised in 2013, approved by the institutional review board TAMU College of Dentistry (Study number: 2014-0833-BCD-FB), and registered under Clinicaltrials.gov (ID: NCT03226600). Written informed consent for participation was obtained from each patient.
2.5. Statistical Analysis
Data were tabulated and analyzed as described above using IBM SPSS software (IBM Corp, New York, USA). Analysis of Variance (ANOVA) was utilized to test the effect of procedure (CTG vs. OrACELL) over time for the clinical measures. If the ANOVA showed statistical significance, paired t-tests were used to compare change in clinical measures from baseline to 3 month and 6 month evaluations. A significance of α = 0.05 was utilized for all testing.
3. RESULTS
All 24 enrolled patients completed the study. No patients experienced post-operative infections, dehiscences, or noted sequelae over the course of the study. Baseline, 3 month, and 6 month clinical measurements for test and control groups are summarized in Table 1. Furthermore, root coverage and defect coverage were calculated and reported in Tables 2 and 3, respectively. At baseline there was no statistically significant difference in parameters between the CTG and OrACELL™ groups. The clinical presentation of both test and control sites can be seen in Figs. (1 and 2).
| - | Baseline | 3 Month | 6 Month | |||
|---|---|---|---|---|---|---|
| - | CTG | OrACELL™ | CTG | OrACELL™ | CTG | OrACELL™ |
| VR | 3.27±0.68 | 3.50±0.89 | 0.64±0.78α | 1.35±1.26α | 0.59±0.70α | 1.19±1.07α |
| HR | 3.50±1.02 | 3.35±0.66 | 1.91±1.77α | 2.08±1.5 α | 1.86±1.61α | 2.31±1.44α |
| PD | 1.59±0.58 | 1.23±0.39 | 1.23±0.68 | 1.39±0.55 | 1.23±0.52 | 1.31±0.43 |
| CAL | 4.86±0.74 | 4.73±0.90 | 1.86±0.87α | 2.73±1.25α | 1.91±1.00α | 2.42±1.17α |
| KT | 1.96±1.80 | 2.04±0.95 | 2.23±1.15 | 2.58±1.47 | 2.55±1.62 | 2.50±1.58 |
| PHM | 3.36±1.05 | 3.46±0.83 | 2.86±0.67 | 3.23±0.86 | 2.77±0.47 | 3.27±0.78 |
| PHD | 3.32±1.03 | 3.58±0.93 | 2.82±0.75 | 3.35±0.75 | 2.86±0.64 | 3.31±0.72 |
| PWM | 3.68±0.84 | 3.23±0.86 | 3.23±0.75α | 2.69±0.72α | 3.00±0.71α | 2.85±0.59α |
| PWD | 3.36±0.71 | 3.23±0.83 | 3.05±0.79 | 3.00±1.00 | 2.82±0.75 | 2.96±0.78 |
| α: indicates statistical difference between baseline and 3 or 6 month for either CTG or OrACELL™ All measurements are in millimeters and statistical significance was set at α ≤ 0.05 | ||||||
| - | Baseline | 3 Months | 6 Months | |||
|---|---|---|---|---|---|---|
| Material | CTG | OrACELL | CTG | OrACELL | CTG | OrACELL |
| % Root Coverage | 75.98% | 74.32% | 95.33% | 90.12% | 95.66% | 91.25% |
| - | 3 Months | 6 Months | ||
|---|---|---|---|---|
| Material | CTG | OrACELL | CTG | OrACELL |
| % Defect Coverage | 80.56% | 61.54% | 81.94% | 65.94% |
3.1. Vertical Recession
In general, both CTG and OrACELL™ had significant improvement in VR at 3 months and 6 months when compared to baseline (p=0.001). There was no difference between CTG and OrACELL for each time point (p=0.650).
3.2. Horizontal Recession
Similarly, both CTG and OrACELL™ had significant improvement in HR at 3 months and 6 months when compared to baseline (p=0.001). There was no difference between CTG and OrACELL for each time point (p=0.758).
3.3. Probing Depth
At baseline, the PD for CTG and OrACELL™ were 1.59±0.58 mm and 1.23±0.39 mm, respectively. At 6 months, PD remained relatively unchanged at 1.23±0.52 mm for CTG and decreased to 1.31±0.43 mm for OrACELL™. All intergroup differences did not reach statistical significance.
3.4. Clinical Attachment Level
Similarly, both CTG and OrACELL™ had significant improvement in CAL at 3 months and 6 months when compared to baseline (p=0.001). There was no difference between CTG and OrACELL for each time point (p=0.234).
3.5. Keratinized Tissue
Both groups had a greater amount of KT gain as a result of the procedure (approximately 0.50 mm). However, changes in KT over time were not statistically significant (p=0.426) nor were intergroup differences over time (p=0.891).
3.6. Papillary Measurements
From all the papillary measurements only the decrease in PWM was found to be statistically significant over time (p=0.028). However, the difference between groups was not significant at any time point for PHM, PHD, PWD, and PWM.
Bleeding on Probing BOP was recorded with ordinal data (yes or no) and was therefore evaluated with a Chi-Square Test (α=0.05). For the OrACELL group, 85% of patients experienced no BOP at baseline, 92% at 3 months and 85% at 6 months. There were no significant differences (p=0.795). The CTG group showed 73% patients with no BOP at baseline, 82% at 3 months and 91% at 6 months. There were no significant differences (p=0.543). No differences were detected comparing the two groups at baseline (p=0.475), at 3 months (p=0.439) and at 6 months (p=0.642).
4. DISCUSSION
This study compared the use of decellularized dermis in providing root coverage to that of the gold standard, CTG. Decellularized dermis/OrACELL™, unlike ADM and ECM, has little current literature regarding its efficacy as a CTG substitute. The goal of therapy for many clinicians to find a material that decreases the post-operative morbidity of a palatal harvest site, yet maintains a similar effectiveness for root coverage and esthetics as CTG. Decellularized dermis, like Acellular Dermal Matrices (ADM) and Extracellular Matrices (ECM), reduces post-operative morbidity and surgical time since a palatal surgical site is not required. Decellularized dermis also allows for significantly more sites to be treated in one surgery, as there is a limitless supply of graft material.
The current reporting trends of root coverage can be misleading and deceiving, according to Greenwell et al., because only the amount of soft tissue covering the original defect tends to be calculated [9]. For example, a tooth with 4 mm of VR is treated and the result is the VR is decreased to 2 mm. This finding would be reported as 50% of root coverage being achieved. According to Greenwell et al., this method doesn’t appropriately explain the measurement of root coverage and should rather be referred to as “defect coverage calculated” [9]. Greenwell goes on to suggest it be calculated as follows:
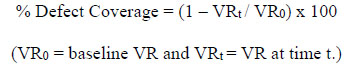 |
Greenwell et al., recommends labeling root coverage as a function of VR at baseline and after treatment over root length, i.e. 5 mm of VR on a 15 mm length root would have 66% root coverage. Subsequently, the actual root length of every tooth that is treated is often unknown. Therefore, Greenwell et al., recommends a generally accepted universal root length of 13.63 mm for standardization of calculation [10]:
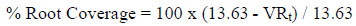 |
Table 2 provides a summary of percent root coverage in test and control groups over time according to criteria set by Greenwell et al. See Table 3 for a summary of percent defect coverage according to criteria set by Greenwell et al.
Greenwell et al., proposes that defect elimination is successfully achieved when 95 to 100% mean root coverage is obtained [9]. At 6 months, 95.66% mean root coverage was achieved for CTG and 91.25% mean root coverage for OrACELL™ Table 2. At 6 months the defect coverage was 81.94% and 65.94% for CTG and OrACELL™, respectively, which is illustrated in Table 3. These findings are likely explained by the inclusion of Miller Class III defects in 23 of the 24 subjects. Miller explained that complete root coverage wasn’t realistic in Class III defects due to interproximal bone loss, extrusion, or malpositioning [3]. The surrounding blood supply for a graft in a Miller Class III defect is often apical to the CEJ and therefore both graft and overall tissue height cannot be maintained at the CEJ.
It was noted at sites included in this study, teeth that were misaligned outside the alveolus. The baseline average distance from the CEJ-to-bone was 5.87 mm, and it’s plausible that malpositioning lead to an absence of buccal alveolar housing (hidden recesssion), which subsequently initiated gingival recession at the site. The vasculature to the site from the periodontal ligament, alveolar bone, and supraperiosteal vessels is often compromised in these malpositioned Miller Class III defects. Marini et al., evaluated gingival recession in 380 adult subjects and found that 3,526 teeth had a total 6,123 surfaces with recession. This ultimately equated to 89% of subjects exhibiting at least one tooth surface with gingival recession [11]. Furthermore, Marini et al found that the majority of these defects were 59% Miller Class I and 33% Class III defects [11]. Twenty-three of the 24 sites analyzed in this study were Miller Class III recession defects due to interproximal bone loss and tooth malpositioning. The authors included Miller Class III defects because of the significant prevalence of these defects among individuals with gingival recession. The authors also found in this study that Miller Class III defects treated with OrACELL™ failed to demonstrate a significant difference in remaining VR and CAL when compared to defects treated with CTG over a 6-month follow-up. There was a statistically significant difference in CAL at 3 months, but the difference was no longer found to be statistically significant at 6 months. Gingival recession studies commonly limit recession defect inclusion to only Miller Class I and II defects because of their increased treatment predictability. With Miller Class III defects being less predictable, they may perhaps serve as a better gauge of donor graft tissue efficacy. Historically, CTG would serve as the material of choice for treatment of Miller Class III defects due to its predictability and status as the gold standard. However, this study demonstrates that OrACELL™ was as effective as CTG in the treatment of Miller Class III defects. A recent review by Miller revisited the concepts of predictable root coverage for Class III defects [12]. He stated that newer surgical techniques that preserve the intact papilla allow for more root coverage than earlier anticipated. He also found that the width and height of the papilla makes a significant determinant in root coverage outcome. This study would confirm that statement and the use of OrACELL™ for treatment of gingival recession.
Similar findings to this current study have been reported in other studies that included Miller Class III recession defects. Few studies have reported on the use of decellularized dermis for root coverage but numerous investigators have reported on ADM. Barker compared the differences in root coverage of Miller Class I and III defects, using two different ADM products (AlloDerm® and Puros® Dermis). This study reported mean defect coverage of 81.4% and 83.4% using the two products [13]. Mean root coverage was 95.2% for AlloDerm® and 95.1% for Puros® Dermis when using the Greenwell et al., criteria for root coverage. Another study by Shin treating 42 Miller Class I and 40 Miller Class III defects in 14 patients reported mean defect coverage of 73.4% for ADM and 79.4% for ADM plus enamel matrix derivative (EMD) [14]. When using the Greenwell et al., criteria for root coverage, the mean was 93.1% for ADM and 94.1% for ADM plus EMD. Miller Class I, II, and III defects were examined in a study by Carney using ADM with and without growth factors (GEM21™). Carney reported average defect coverage of 76.7% in the group without addition of growth factors and 69% for the group with growth factors [15]. When analyzed separately, Miller Class III defects demonstrated mean defect coverage of 60.8% and 51.5% for ADM and ADM plus rhPDGF, respectively. When using the Greenwell et al., criteria for root coverage, mean root coverage was 94.4% for ADM and 95.2% for ADM plus rhPDGF.
An interesting finding from the current study was the increase in keratinized tissue for both the CTG and OrACELL™ groups. There was no statistically significant difference between groups at baseline, 3 months, or 6 months but there was a gradual incremental increase from baseline to 6 months. KT for CTG improved from an average of 1.96 ± 1.80 mm at baseline to 2.55 ± 1.62 mm at 6 months, while OrACELL™ enhanced from 2.04 ± 0.95 mm to 2.50 ± 1.58 mm. While neither of these findings is statistically significant, the authors believe they may be clinically significant. This investigation is not the first to report an improvement in KT after a root coverage procedure. Barros reported a gain of KT of 1 mm at 6 months after ADM procedures for root coverage in Miller Class I and II defects [16]. Also looking at Class I and II defects with ADM for root coverage, Aichelmann-Reidy reported a gain in KT of 1.2 mm at 6 months [17]. According to Karring et al., the genotype of underlying connective tissue determines the character of the overlying epithelium [18]. The use of deceullarized dermis produced small but similar increases in KT compared to CTG, which undermines the mechanism described by Karring et al. This leads the authors to speculate that the increase in KT from decellularized dermis is potentially a result of measuring error or perhaps a signaling process that is not currently well understood.
The proceedings of the 2014 AAP Regeneration Workshop noted the lack of evidence to support the use of alternative grafting materials for root coverage procedures. They stated that the subepithelial connective tissue graft was still the gold standard and provided the best root coverage outcomes. They also called for additional research needed to support the treatment outcomes of Miller Class III and IV defects [19]. The present study gives additional evidence in support of alternative grafting materials, specifically for the Miller Class III defects and addresses the advantage of increasing the zone of keratinized tissue with both materials [20].
The latest revisions to the classification of periodontal disease and conditions also mentions the value of basing diagnosis and treatment options on additional criteria, such as periodontal biotype, recession severity, and root surface conditions [21] and recommends a modern classification based on the interdental clinical attachment level measurement as proposed by Cairo et al., The Cairo classification also helps distinguish the differences among Miller Class III defects as to their predictability for complete root coverage based on the level of the interdental CAL in relation to the buccal CAL. It specifies the difference between the recession type 2 (RT2) and recession type 3 in evaluating the outcome of treatment and again lends credibility to the possibility for complete root coverage in RT2 defects (previously classified as Miller Class III) [22].
CONCLUSION
This randomized clinical trial failed to find any statistical differences in VR between groups at any time point in the treatment of gingival recession defects. While there was a small but significant difference in CAL between groups at 3 months, no statistically significant difference remained regarding CAL after 6 months of healing. Evidence from this study supports the use of OrACELL™ in root coverage procedures in Miller Class I, II, and III defects. In Miller Class III recession defects, OrACELL™ provided a viable alternative to CTG with similar results. Conclusions drawn from this study pertain solely to OrACELL™ as it was the only decellularized dermis product tested. Future studies should include long-term follow-up and larger sample populations. Additionally, histologic comparison of CTG and OrACELL™ tissue biopsies should be evaluated for differences in root attachment and keratinization.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was approved by the institutional review board TAMU College of Dentistry (Study number: 2014-0833-BCD-FB), and registered under Clinicaltrials.gov (ID: NCT03226600).
HUMAN AND ANIMAL RIGHTS
No animals were used in this research. All research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013 (http://www.wma.net/en/20activities/10ethics/10helsinki/).
CONSENT FOR PUBLICATION
Written informed consent for participation was obtained from each patient.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
All OrACELL™ material was donated to Texas A&M University College of Dentistry (Dallas, TX) from LifeNet Health (Virginia Beach, VA).


