All published articles of this journal are available on ScienceDirect.
Current Aspects on Oral Squamous Cell Carcinoma
Abstract
Oral squamous cell carcinoma is the most common malignant epithelial neoplasm affecting the oral cavity. This article overviews the essential points of oral squamous cell carcinoma, highlighting its risk and genomic factors, the potential malignant disorders and the therapeutic approaches. It also emphasizes the importance of the early diagnosis.
INTRODUCTION
Worldwide, oral cancer accounts for 2%–4% of all cancer cases. In some regions, the prevalence of oral cancer is higher, reaching the 10% of all cancers in Pakistan, and around 45% in India [1,2]. In 2004-2009 over 300,000 new cases of oral and oropharyngeal cancer were diagnosed worldwide. During the same time period, over 7,000 affected individuals died of these cancers [3].
Oral cancer includes a group of neoplasms affecting any region of the oral cavity, pharyngeal regions and salivary glands. However, this term tends to be used interchangeably with oral squamous cell carcinoma (OSCC), which represents the most frequent of all oral neoplasms. It is estimated that more of 90% of all oral neoplasms are OSCC [4].
Despite the advances of therapeutic approaches, percentages of morbidity and mortality of OSCC have not improved significantly during the last 30 years. Percentages of morbidity and mortality in males are 6.6/100,000 and 3.1/100,000 respectively, while in females the same percentages are 2.9/100,000 and 1.4/100,000 [5]. Additionally, the incidence of OSCC is increasing among young white individuals age 18 to 44 years, particularly among white women [6]. The percentage of 5-year survival for patients with OSCC varies from 40-50%. Regardless of the easy access of oral cavity for clinical examination, OSCC is usually diagnosed in advanced stages. Most common reasons are the initial wrong diagnosis and the ignorance from the patient or from the attending physician [7].
CLINICAL FEATURES
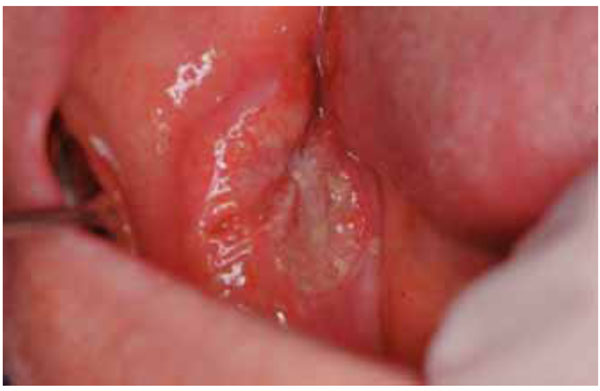
One of the real dangers of this neoplasm, is that in its early stages, it can go unnoticed. Usually at the initial stages it is painless but may develop a burning sensation or pain when it is advanced. Common sites for OSCC to develop are on the tongue, lips and floor of the mouth. Some OSCCs arise in apparently normal mucosa, but others are preceded by clinically obvious premalignant lesions, especially erythroplakia and leukoplakia. Usually, OSCC presents as an ulcer with fissuring or raised exophytic margins (Fig. 1). It may also present as a lump (Fig. 2), as a red lesion (erythroplakia), as a white (Fig. 3) or mixed white and red lesion, as a non-healing extraction socket or as a cervical lymph node enlargement, characterized by hardness or fixation. OSCC should be considered where any of these features persist for more than two weeks.
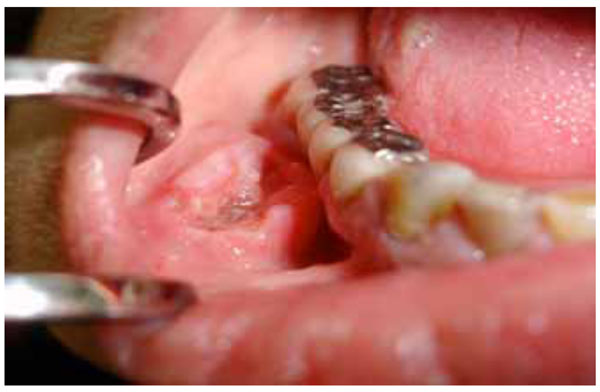
OSCC of the vestibule with raised exophytic margins.
OSCC of the buccal mucosa presenting as an asymptomatic ulcer.

Leukoplakia of the lateral surface of the tongue undergoing malignant transformation.
RISK FACTORS
The greatest risk factor for oral cancer in the western world is the use of tobacco and alcohol [8-12]. Although the risk factors are independent, their action seems to be combined. Tobacco smoking is associated with 75% of all cases of oral cancer. Tobacco smoking carries a six-fold risk of developing oral cancer compared to not smoking. Oral cancer is also six times more likely to develop in alcohol drinkers than in non-drinkers. The combination of tobacco and alcohol use poses a fifteen-fold risk of oral cancer for users compared to non-users [13].
While tobacco and alcohol use are traditionally the greatest risk factors, it is important to consider other known risk factors, such as betel quid chewing, in certain ethnic populations. Betel quid chewing is popular in Indian and Taiwanese populations and is associated with a significantly increased risk of oral cancer [14-16]. Areca nut, narcotics and cannabis use has also been found to be a risk factor for oral cancer [17].
OSCC is most common in older males, in lower socioeconomic groups and in ethnic minority groups.
Other factors also play a role. These include:
- An impaired ability to repair DNA damaged by mutagens
- An impaired ability to metabolize carcinogens
- Deficiencies of vitamins A, E or C or trace elements
- Immune defects.
The inadequate immune response may predispose to cancer development. The most common oral malignancy in individuals with HIV infection is Kaposi’s sarcoma. There is a high prevalence in patients with B cell Hodgkin lymphoma. There is an increased risk of developing OSCC in HIV infected patients and patients submitted to organ transplantations and those who are under immunosuppressive therapy [5, 18].
VIRAL INFECTIONS
Human Papilloma Virus (HPV)
More than 100 types of HPV have been identified and are referred as viruses of low or high danger according to their oncogenetic potential. The predominant viral type, in most studies, was HPV16. HPV was more frequently detected in OSCCs of the oropharynx and tonsil than at other head and neck sites. Research on the participation of HPV in oral carcinogenesis has conflicting results. The percentage of the reported infected neoplastic cells varies from 0-90%.Several research groups proposed that HPV is implicated in the initial stages of carcinogenesis, while others have suggested a short-term (“hit and run”) role of HPV. It is believed that viral protein E6 binds to p53 causing its breakdown, while E7 reacts with retinoblastoma protein (pRb), a tumor suppressor protein, inhibiting its function. The functional deregulation of these oncosuppressive key molecules results in an incontrollable cellular proliferation and to disturbances of apoptosis, leading to oral cancer. It is currently believed that HPV infection of oral mucosal cells per se is not enough for malignant transformation unless cells are exposed to chemical carcinogens, such as benzopyrene [19-23].
Epstein-Barr Virus
Epstein-Barr virus is known to be the infective agent in infectious mononucleosis. While its contribution to malignant transformation of B cells is well documented the participation in pathogenesis of OSCC remains unclear. Some investigators proposed that the dominant oncoprotein of the latent phase (LMP-1) is expressed in oral epithelial malignant cells [23, 24].
Hepatitis C Virus (HCV)
The prevalence of oral squamous cell carcinoma is higher in HCV infected patients. In a Japanese study, it has been shown that HCV infection was strongly associated with the occurrence of multiple primary carcinomas as well as primary OSCC [25].
POTENTIALLY MALIGNANT DISORDERS
In a recently held WHO workshop it was recommended that the distinction between potentially malignant lesions and potentially malignant conditions should be abandoned and rather to use the term potentially malignant disorders instead [26].
Some precancerous disorders can progress to OSCC. These are the following:
- Erythroplakia
- Leukoplakias, particularly:
- Erythroleukoplakia (nodular or verrucous)
- Proliferative verrucous leukoplakia
- Actinic cheilitis
- Lichen planus (mainly the erosive and atrophic type)
- Sideropenic dysphagia (Plummer-Vinson syndrome)
- Submucous fibrosis
- Dyskeratosis congenita
- Discoid lupus erythematosus
MOLECULAR PATHOGENESIS OF OSCC
Several studies have been devoted to the significance of heredity in oral carcinogenesis. The relative danger of the disease development in first degree relatives of patients with oral cancer varies from 1.1-3.8 odds ratios [27]. Several genes are implicated in genetic predisposition of oral cancer. Gene polymorphisms participating in the metabolism of xenobiotic factors, such as cytochrome P450 1A1 (CYPIA 1) and glutathione S-transferase mu 1 (GSTM1) are blamed for the increase of relative danger in the carriers [28, 29]. Individuals with alcohol dehydrogenase 3 genotype are prone to the development of oropharyngeal cancers [30, 31].
However, the relation of oral cancer with an autosomal dominant type of heredity has been rarely recorded, mainly in patients with Fanconi’s anemia.
OSCC arises as a consequence of multiple molecular events that develop from the combined effects of an individual's genetic predisposition and exposure to environmental carcinogens [32], such as, tobacco, alcohol, chemical carcinogens, ultraviolet or ionizing radiation and micro-organisms [33-35]. Chronic exposure to carcinogens may damage individual genes as well as larger portions of the genetic material, such as chromosomes. Genetic damages may activate mutations or amplification of oncogenes that promote cell survival and proliferation. Mutations include DNA general hypomethylation, hyper- or hypomethylation of certain genes such as cyclin D, and alterations of chromatin [36, 37]. Oncogenes are broadly categorized as follows: (i) growth factors or growth factor receptors (hst-1, int-2, EGFR/erbB, c-erbB-2/Her-2, sis), (ii) transcription factors (myc, fos, jun, c-myc), (iii) intracellular signal transducers (ras, raf, stat-3); (iv) inhibitory factors of apoptosis (bcl-2, bax) and (v) cell-cycle regulators (cyclin D1) [38]. Genetic damages may also inactivate tumor suppressor genes involved in the inhibition of cell proliferation. All these events may lead to cell dysregulation to the extent that growth becomes autonomous and invasive mechanisms develop.
As OSCC grows and invades, new blood vessel formation occurs. This angiogenesis is an essential part of tumor formation [39].
Field cancerization is a theory of oral carcinogenesis. According to this theory since the oral epithelium is exposed to carcinogenic factors, the entire area is at increased risk for the development of malignant lesions from the accumulation of genetic alterations of oncogenes and tumor suppressor genes [40].
In cancerization field, multiple oral cancers may develop from independent cell clones. This hypothesis has been supported by data from chromosome X inactivation studies, microsatellite analysis, and p53 mutational analysis [41]. However, more recent genetic studies suggested that multiple cancers can be clonally related and derived from expansion of an original clone [42]. These results gave rise to a modification of cancerization field theory, the patch field carcinoma model [43]. According to this model, a stem cell located in the oral epithelium acquires a genetic alteration and generates daughter cells, all of which share the genetic alteration. This patch of cells expands to a size of several centimeters to the surrounding oral mucosa and macroscopically is often undetectable. In some instances it may appear with distinct morphological characteristics, like leukoplakia or erythroplakia.
STAGING OF OSCC
Staging of OSCC is performed using the TNM system. cTNM is the stage given after the clinical examination of the patient, while pTNM is the stage after the histopathological examination of the surgical specimen. However, this traditional way of staging is often inadequate. Evaluation of other features of the neoplasms, such as degree of differentiation, type of infiltration and degree of recurrence [44] facilitate the exact diagnosis and permit the selection of the appropriate therapeutical approach.
THERAPEUTIC APPROACHES AND PROGNOSIS
Despite advances in surgery and radiotherapy, which remain the standard treatment options, the mortality rate has remained largely unchanged for decades, with a 5-year survival rate of around 50% [45]. In the primary (I and II) stages the treatment of choice is surgery and/or radiotherapy, which usually result in permanent cure. Combination of surgery, radiotherapy or chemotherapy is used for the treatment of the 3rd or 4th stage of OSCC. Oral care is especially important when radiotherapy is to be given, since there is a liability particularly to mucositis, xerostomia and osteonecrosis.
It is generally accepted that prognosis is best in early OSCC, especially those that are well-differentiated and not metastasized: unfortunately, most OSCC are diagnosed at a late stage of the disease. The prognosis of OSCC varies on a number of factors that are related to the tumor, to the treatment, and to the patient [46]. However, five year survival rates in the advanced stages do not exceed 12% of the cases. Most patients with advanced OSCC usually die within the first 30 months of their disease [47, 48].
Metastases from OSCC, when present, will occur in cervical lymph nodes in almost 80% of patients. Cervical lymphadenectomy (radical neck dissection) is traditionally applied in these cases [49]. More recently, selective neck dissection has been developed in order to reduce the morbidity of radical neck dissection [50].
Efforts to increase the efficacy of radiotherapy, especially in local advanced disease, include, altered fractionated radiotherapy or concomitant chemo-radiotherapy (CT-RT) [51].
As for chemotherapy, cisplatin-based chemoradiation remains the standard for locoregionally advanced head and neck SCC [52].
Currently, targeted molecular therapy, like therapy with monoclonal antibodies and gene therapy, has been applied to oral cancer patients. This treatment modality has limited or nonexistent side effects on normal cells of the body, unlike surgery, chemotherapy, and radiotherapy. Targeted molecular therapy can also act as a complement to other existing cancer therapies and has been mainly focused on four molecules; epidermal growth factor receptor (EGFR), cyclooxygenase-2 (COX-2), peroxisome proliferator-activated receptor γ (PPARγ), and progesterone receptor. These molecules are associated with the proliferation and the differentiation of OSCC [53].
Finally, early diagnosis remains the key element for the sufficient therapy of OSCC. Clinicians should be aware that single ulcers, tumors, red or white plaques, particularly if any of these are persisting for more than two weeks, may be manifestations of malignancy. In these cases a biopsy from the suspicious lesion is needed. Finally, new emerging technologies are developing and targeting to early diagnosis of OSCC through molecular analysis of cytologic smears or saliva samples [54].
CONFLICT OF INTEREST
The author confirms that this article content has no conflicts of interest.
ACKNOWLEDGEMENT
Declared none.


