All published articles of this journal are available on ScienceDirect.
Medication-related Osteonecrosis of the Jaw and Dental Implant: A Case Report
Abstract
Introduction/Background
The rising use of anti-resorptive agents, including denosumab, for bone metastases has led to an increased incidence of medication-related osteonecrosis of the jaw. This particular case represents an important, but also often overlooked, potential late sequelae that may be seen in cancer patients, which is the failure of a previously well-functioning dental implant years down the line, which may then become a causative factor for MRONJ.
Case Presentation
A 70-year-old female patient, a chronic user of high-dose denosumab for metastatic breast carcinoma, presented with pain and infection of the mandibular implant, which was functionally osseointegrated for seven years before the onset of symptoms. The patient was evaluated and was found to have a bony sequestrum containing the implant, meaning the diagnosis was a Stage 2 MRONJ. Conservative management was ineffective; thus, surgical intervention involving sequestrectomy and removal of the implant en bloc was performed.
Conclusion
This case of treating the patient also shows us that patients undergoing any treatment with denosumab, who have dental implants, are continuously at risk of late-onset MRONJ and hence need long-term surveillance, and thereby, more emphasis needs to be placed on giving them informed consent for such a lifelong risk of acquiring the disease. Thus, great care must be taken before performing any such surgical procedure. This case also shows that the surgery has been successful in treating the patient, without the need to withdraw any ongoing antiresorptive drugs.
1. INTRODUCTION
There is an increase in the use of anti-resorbing agents worldwide, and it is predicted that this will increase with the rise of the population who require treatment for bone metabolic disorders, for instance, osteoporosis, metastases to bone, and cancer-related skeletal events [1]. On the other hand, dental implant therapy is considered the “standard of care” for oral rehabilitation and has been shown to be more favorable than traditional prostheses, particularly for an aging population.[1] As a result of these developments, it is imperative that dental professionals become familiar with the effect of anti-resorptive agents on dental implant therapy.
Bisphosphonates (BPs) and Denosumab are two of the most commonly prescribed anti-resorptive therapies, but they belong to different pharmacological classes with different mechanisms of action [2]. Nitrogen-containing BPs, for example, have a high affinity for hydroxyapatite, binding to minerals within the bone, which eventually leads to the apoptosis of osteoclasts and reduces bone resorption [2]. Denosumab, on the other hand, is a monoclonal antibody with high affinity for binding to the RANKL, resulting in reduced osteoclast formation, as well as their diminished activities, which leads to the significant interference with bone remodeling, which is a crucial event for osseointegration, with the outcome being the failure of osseointegration, with chances of sequestrations incurred with the use of implants [3-5]. Notably, when applicable to patients treated with high doses of Denosumab (i.e., 120 mg) for cancer treatment, the chance of developing MRONJ is increased, especially since a study on a 2025 population revealed a 11.6% incidence, which is way above the generally quoted 1-2% risk [6, 7].
Denosumab can compromise implant success through multiple mechanisms. The strong antiresorptive effect of denosumab on bone derived from osteoclasts can impair bone's ability to repair microdamage, slow bone remodeling after traumatic injury, and predispose individuals to various infections by virtue of necrotic, avascular bone [8-11]. It is worth noting that the effects of denosumab can be reversed when it is discontinued, unlike BPs, whose components become embedded in bone minerals. Nevertheless, it is the type of high-dose treatment strategy employed in the management of different types of cancers, as illustrated by the use of denosumab (120 mg of active ingredient, monthly), that leads to a high risk of the patient developing MRONJ. Hence, it is in consideration of the aforementioned factors that different organizations, as illustrated by the American Association of Oral and Maxillofacial Surgeons' classification of the drug, classify such drugs as high-risk [8].
The risk of dental implants in patients on or to be started on anti-resorptive or anti-angiogenic therapy must be weighed. “For patients on high-dose treatment for oncologic indications, for example, intravenous bisphosphonates or denosumab (Xgeva), the risk of MRONJ is substantially increased, and dental implant placement is contraindicated [9]. For individuals with osteoporosis taking oral bisphosphonates, there appears to be no increase in the risk of implant failures due to poor osseointegrations, although they are at risk of implant surgery-triggered MRONJ. This is a serious complication, and the risk must be considered during the patient-informed consent process. Clinical guidelines strongly support a dental evaluation, including completion of all invasive dental procedures, such as extractions and dental implants, prior to the onset of anti-resorptive therapy, if the patient’s systemic status permits. This is particularly important for the increasing number of older adults, who are more likely to be receiving polypharmacy, including these drugs, and at the same time require oral rehabilitation with dental implants [9].
Despite extensive research, the level of influence that anti-resorptive medications exert on implant failure as well as marginal bone loss remains a thoroughly controversial topic. It is worth noting that reports on anti-resorptive agents yield inconsistent results, further underscoring the prevalence of controversy. Consequently, considering the level of risk that is associated with denosumab, as well as the unique risks, necessitates a detailed level of reporting, as highlighted below, in an attempt to underscore an essential need, an advanced-stage MRONJ manifesting as a complication around an implant that has previously achieved osseointegration within an individual undergoing high-dose treatment with denosumab.
2. CASE PRESENTATION
A 70-year-old female patient presented to our clinic in December 2021 with a chief complaint of persistent pain and swelling in the region of the mandibular right second premolar (tooth #45) and the crown on the adjacent dental implant (site #46).
Her medical history was significant for metastatic breast cancer. A right mastectomy was performed in 1995. The disease recurred in 2002 with lymph node metastases, prompting treatment with the aromatase inhibitor letrozole (Femara®). In 2015, further progression to bone metastases (spine, ilium, rib) was diagnosed. Treatment with high-dose denosumab (Xgeva® 120 mg subcutaneously) was subsequently initiated on a 28-day interval. In 2016, due to stable disease, the dosing interval was extended to once every 3 months. However, following disease progression in 2020, the regimen was resumed on the original 28-day schedule. Her most recent administration was three months prior to her presentation, on August 28, 2024. The patient had no history of head and neck radiotherapy.
The dental implant at site #46 had been placed in 2014, achieving successful osseointegration and prosthodontic rehabilitation. This was one year prior to the initiation of denosumab therapy in 2015. The postoperative course was uneventful for seven years. In mid-2021, she developed symptoms in the area and was evaluated by a periodontist. Initial treatments included professional oral hygiene, closed curettage of the gingival sulci around teeth #45 and #46, antiseptic rinses with 0.12% chlorhexidine, and a course of amoxicillin-clavulanate. These measures provided only transient relief and failed to resolve her symptoms.
2.1. Clinical and Radiographic Findings at Presentation (December 2021)
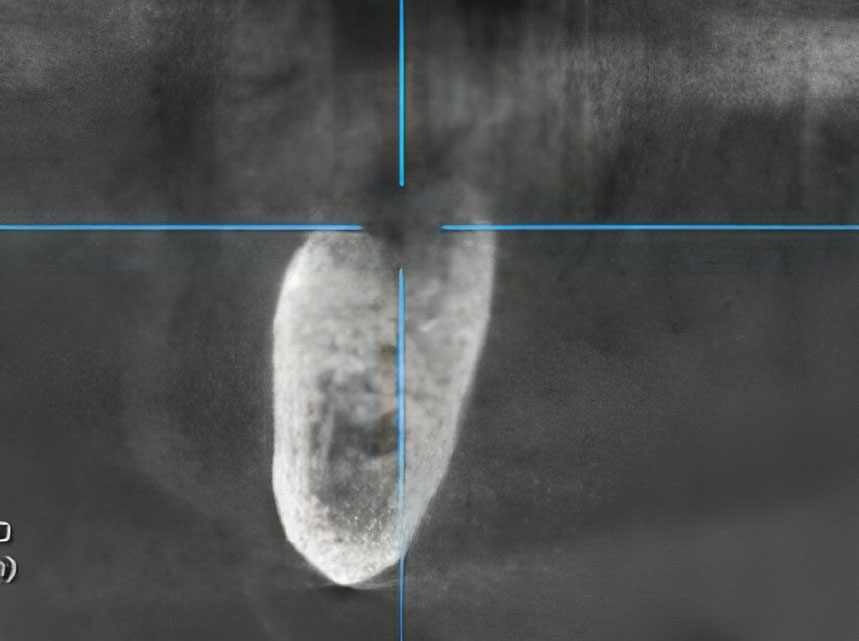
Intraoral examination revealed localized gingival erythema and edema in the area of teeth #45 and #46. Palpation was painful and elicited a purulent discharge from the peri-implant sulcus of #46. Periodontal probing detected deep pockets (>8mm) with palpable, rough, and exposed bone. Notably, the dental implant at site #46 was initially clinically stable. Cone-Beam Computed Tomography (CBCT) imaging revealed a sclerotic bone pattern with increased bone density surrounding the implant, along with a distinct focus of bone destruction adjacent to the implant apex at site #46, consistent with osteonecrosis (Figs. 1, 2). Based on the AAOMS diagnostic criteria, a diagnosis of Stage 2 Medication-Related Osteonecrosis of the Jaw (MRONJ) was established.
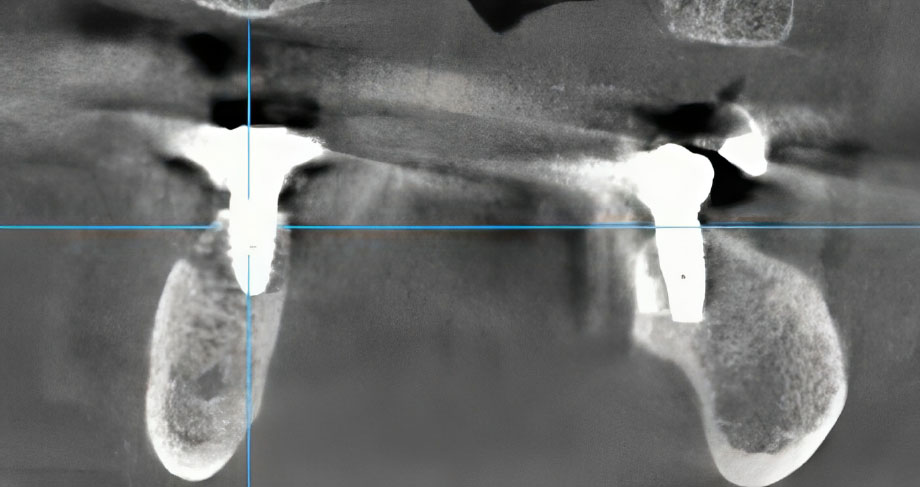
Coronal CT projection showing increased bone density surrounding the implant in the right mandible compared to the contralateral side (preoperative baseline).
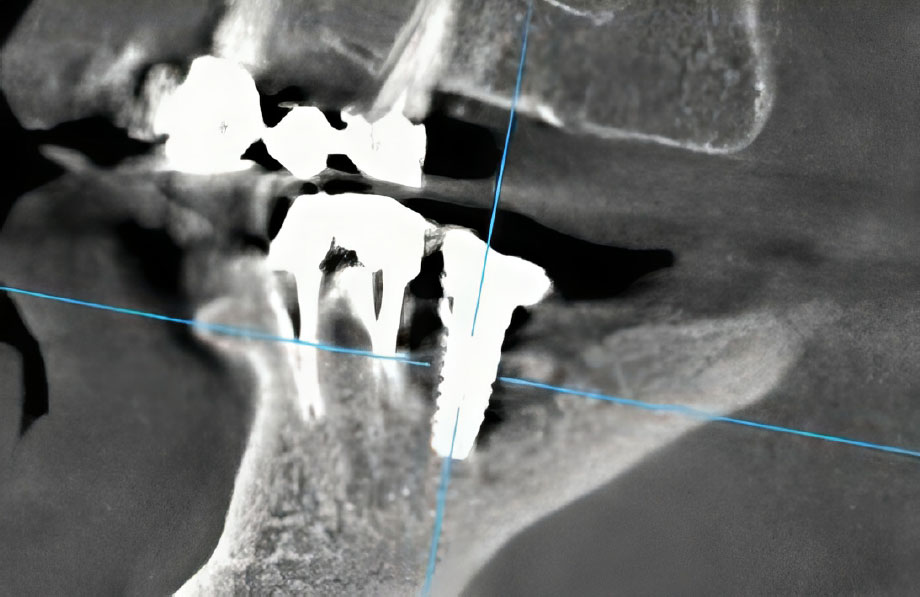
Sagittal CT projection revealing a well-defined focus of bone destruction in the region of tooth #46 with peri-implant involvement.
2.2. Treatment Course and Follow-up
Initial conservative management was instituted, involving local ozone therapy (administered via a periodontal probe for 60 seconds per site), antiseptic treatment with 0.05% chlorhexidine, and courses of systemic antibacterial therapy (amoxicillin-clavulanate 875/125 mg twice daily for 7 days).
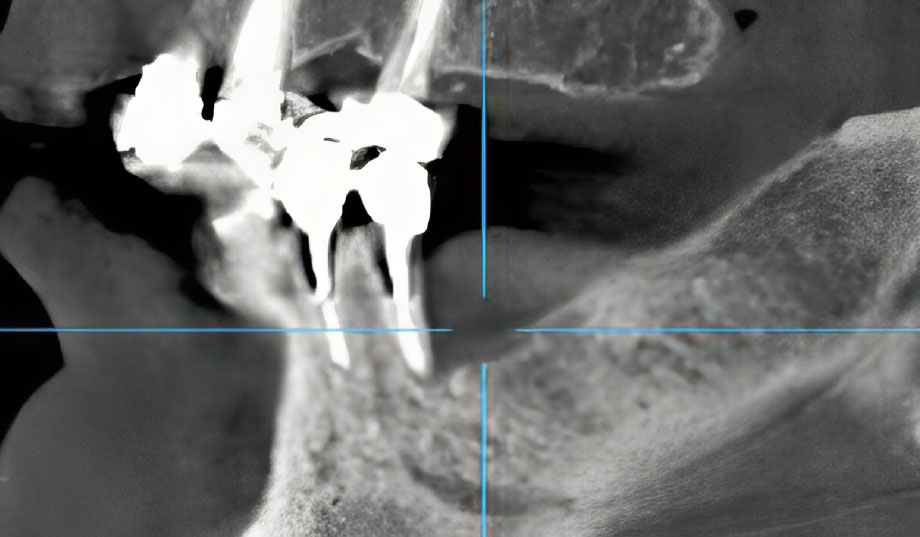
At a follow-up examination in April 2025, a mobile sequestrum was identified, which incorporated the dental implant at site #46. A subsequent CBCT scan confirmed the presence of a fully formed bone sequestrum (Fig. 3).
Surgical intervention was then performed under local anesthesia, consisting of sequestrectomy. The procedure involved the en bloc removal of the necrotic bone, which included the previously osseointegrated dental implant. A postoperative CBCT was obtained (Fig. 4). The surgical site was packed with iodoform gauze, which was left in place for seven days.
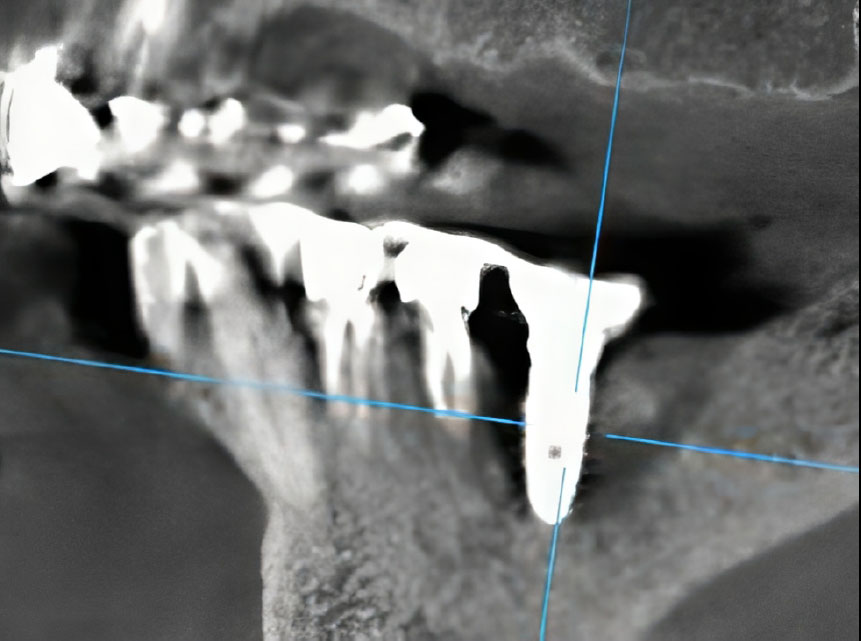
Sagittal CT projection demonstrating a fully formed sequestrum incorporating the dental implant at site #46.
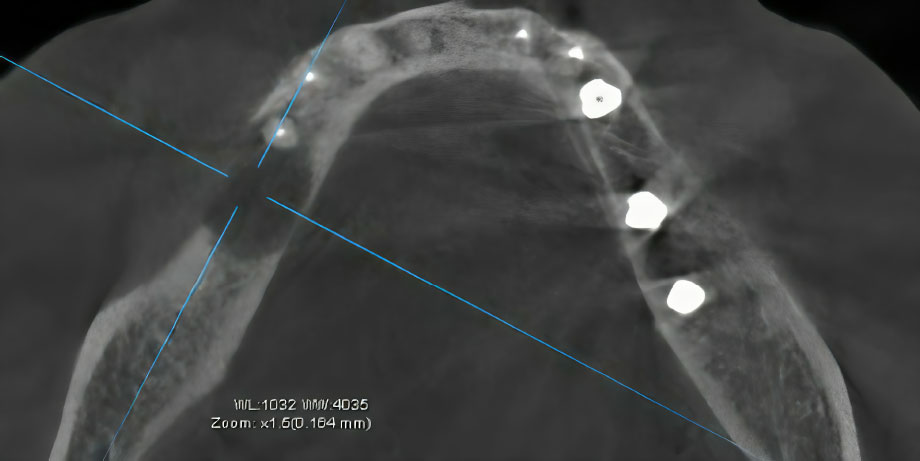
Axial CT projection showing the dimensions of the bone defect immediately following surgical sequestrectomy.
2.3. Outcome
In accordance with AAOMS guidelines for Stage 2 MRONJ, initial conservative management was instituted. This involved superficial debridement, local ozone therapy, daily antiseptic rinses with 0.05% chlorhexidine, and repeated courses of systemic antibiotics (amoxicillin-clavulanate and, subsequently, clindamycin).
The patient was monitored regularly. At a follow-up examination in April 2025, the condition had progressed. The implant was now mobile, and a mobile bony sequestrum incorporating the dental implant body was clinically evident. A subsequent CBCT scan confirmed the presence of a fully formed bone sequestrum (Fig. 3).
Due to the failure of conservative therapy and the presence of a sequestrum with associated symptoms, surgical intervention was indicated. Under local anesthesia, sequestrectomy with en bloc removal of the implant and the surrounding necrotic bone was performed. The surgical site was then packed with iodoform gauze, which was removed after seven days.
Post-treatment follow-up demonstrated successful healing. At the two-month postoperative check, a CBCT scan showed early signs of reparative osteogenesis along the walls of the surgical defect (Fig. 5). Clinically, the mucosa had healed without signs of infection or exposed bone. Five months post-operatively, a control radiograph confirmed complete healing of the bony defect with restoration of the cortical plate (Fig. 6). The patient remained asymptomatic at the site.
Coronal CT projection illustrating active reparative osteogenesis along the walls of the postoperative defect at 3-month follow-up.
Sagittal CT projection showing complete bone regeneration and successful integration of the replacement implant at 12-month follow-up.
3. DISCUSSION
The current case report highlights the occurrence of advanced Medication-Related Osteonecrosis of the Jaw (MRONJ) in a late-stage, advanced metastatic breast carcinoma patient receiving high-dose denosumab (Xgeva) therapy during the course of her treatment. The osseointegrated dental implant in the patient’s right mandibular quadrant had been painlessly functioning for 7 years; however, it now appears to be a nidus of infection and tissue necrosis. The patient now complains of pain and purulent drainage from the affected site. The patient's disease condition was not treated successfully with the initial conservative measures; hence, the successful treatment strategy that was involved in the removal of a nidus of infection and necrosis, an osseointegrated dental implant located within the mandibular right quadrant of the lower jaw of the patient, resulted because the patient healed completely within a range of five months following the procedure itself [11, 12].
The pathogenesis of MRONJ in this setting is multifactorial, with the principal factor being the profound inhibition of osteoclast-mediated bone remodeling induced by denosumab. RANKL antagonistic monoclonal antibody, known as denosumab, is known to inhibit the RANK/RANKL signaling cascade, which is essential for the development, functioning, and survival of osteoclasts. This results in a substantial suppression of bone turnover. Ultimately, this results in impaired healing of physiological microdamage caused by occlusal stresses, reminiscent of the pathogenesis of accumulated microcracks and avascular necrosis [13-15]. Unlike bisphosphonates, which are irreversibly incorporated into the mineralized bone matrix, denosumab's action is reversible upon discontinuation; however, its high potency while active creates a vulnerable bony environment [14, 16]. This risk is further complicated by dental implants, which introduce a permanent foreign body that compromises mucosal integrity and creates a potential nidus for bacterial colonization and biomechanical stress [12, 13].
Recent evidence highlights the critical role of biomechanical factors in MRONJ development, particularly in patients with dental implants. A 2025 observational study identified cantilever extensions in prosthetic designs as a significant risk factor for peri-implant MRONJ (p = 0.002), as non-axial loading patterns may generate excessive mechanical stress on bone already compromised by anti-resorptive therapy [13]. This aligns with our case, where functional occlusal forces likely contributed to microdamage accumulation around the implant. Additionally, factors such as prosthesis misfit, insufficient embrasure space, and emergence angles ≥30° can exacerbate peri-implant inflammation and bacterial infiltration, further increasing MRONJ risk [13]. Since these elements were absent in the patient's prosthesis, it is believed that the drug's bone-remodeling suppression was the major factor; nonetheless, biomechanical stress also played a role.
A notable observation in this patient's case is that, despite the known rapid development of MRONJ following implantation, the latency period is noticeable, with 7 years passing, compared to the more rapid effect seen with dental extractions. This, from a more modern viewpoint and literature, provides credence for the idea that damage is indeed latent and resident at the site of implantation, the RRJMNS presenting as a function of day-to-day trauma and bone infections, though with somewhat delayed onset [13, 16]. This would thus place the patient within a concerning dynamic: one year prior to the implantation of the fixture, receiving denosumab, as the intervention of the implant has undoubtedly exacerbated the impact of anti-resorptive agents on vitality within the subject bone [13]. The rationale with respect to cause, as suspected, lies in the periodontal treatment, resulting from a closed-bone curettage and subsequent microtrauma and bone infections, as part of RRJMNS pathology [15, 17].
The successful resolution of MRONJ in this case through sequestrectomy supports growing evidence that surgical intervention is often necessary for advanced stages. A systematic review of 151 MRONJ cases reported that surgical debridement was the most common procedural treatment, accounting for 32% of cases, and was highly effective when combined with adjunctive therapies [15]. When sequestra are established, conservative measures alone, such as antibiotics, antiseptic rinses, and ozone therapy, often fail, as necrotic bone serves as a persistent nidus for infection [17, 18]. This is similar to our own experience, for example, which mirrors a case series of 2024 in which MRONJ resolution was achieved in 11 patients using procedures such as sequestrectomy and marginal surgery, including the avoidance of segmental jaw surgery [18]. Thus, this is yet another indicator that stages 2-3 MRONJ can be cured with minimally invasive procedures without compromising outcome.
The management of MRONJ in this case did not involve a denosumab holiday, as the patient’s metastatic bone disease required ongoing treatment. This, in turn, underscores the clinical dilemma of finding an appropriate balance between predominantly oncological demands and protection against MRONJ. The current state of evidence on drug holidays is still controversial, especially with regard to denosumab, as withdrawal can cause a rebound phenomenon in bone resorption, thus increasing fracture risk [14, 15]. While caution needs to be exercised for such osteoporotic patients, delaying treatment for RANKL inhibitor therapy will produce a rebound effect, as seen in the case of Denosumab treatment recommended by AAOMS [15]. This is less relevant to oncology patients who will require high doses of these drugs, provided systemic conditions permit [16]. Our case suggests that surgical management without drug interruption can be successful, though close collaboration with oncologists is essential to weigh risks and benefits.
The development of MRONJ around a previously osseointegrated implant, as seen in our case, underscores a critical shift in understanding: dental implants represent not only a surgical risk but also a persistent local risk factor. The pathophysiology is multifactorial, primarily driven by the profound suppression of bone turnover. Denosumab, an anti-RANKL monoclonal antibody, destabilizes osteoclast assembly, function, and survival, effectively arresting bone remodeling [19, 20]. In a normally functioning system, this process constantly repairs the microdamage caused by habitual occlusal stresses. Denosumab-treated systems, however, render the repair mechanism ineffective, culminating in microcracks, ischemia, and avascular necrosis [19].
Our case is consistent with the concept of “implant presence-triggered osteonecrosis,” as described by [21]. The osseointegrated implant serves as a permanent foreign body that violates the mucosal layer, suggesting a potential pathway for bacterial implantation. In addition, the stress concentrated at the implant site is a form of microtrauma, which is difficult for the drug-impaired bone to heal [22]. This can compromise the site to the point where any minor trauma, such as the periodontal curettage, is all that it will take to initiate a chain of infection and osteonecrosis, which can occur years after the initial surgical intervention [23].
Regarding prevention, a proactive and multidisciplinary strategy is paramount. The cornerstone of prevention, as emphasized by the AAOMS, is a comprehensive dental examination and the completion of all invasive dental procedures, including extractions and implant placement, before initiating high-dose antiresorptive therapy [24]. For oncology patients like ours, where drug suspension is often not feasible, the decision to place new implants is generally considered high-risk and relatively contraindicated [24].
Crucially, this case highlights that prevention does not end pre-therapy. For the growing number of patients with existing implants who are subsequently prescribed antiresorptive drugs, prevention must transition to lifelong vigilance. The latter involves complete informed consent, verbal descriptions of the lifelong risk of late-onset MRONJ, meticulous oral hygiene to reduce the incidence of periodontal pathosis, and unlimited oral and medical follow-up care at periodic intervals [21, 23]. The bottom line is a close liaison between oral surgeons and oncologists. In this way, it is possible to strike a balance between the need to deal aggressively with cancer and the need to prevent MRONJ.
The clinical management of this case aligns closely with the recommendations from the American Association of Oral and Maxillofacial Surgeons' 2022 position paper on MRONJ. The diagnosis was based on the established criteria: current treatment with antiresorptive therapy, exposed bone in the mandible for more than eight weeks, and no history of head and neck radiation. The initial conservative approach described for Stage 2 disease, antimicrobial rinse, antibiotic therapy, and superficial debridement, is the approach of first choice. The evolution of disease and the presence of a sequstrum required a surgical approach, and this is indicated for symptomatic non-responsive Stage 2 and all Stage 3 disease. The performed sequestrectomy is a recognized surgical procedure aimed at removing the necrotic nidus, and our successful outcome supports its efficacy. Furthermore, the decision to continue denosumab without a “drug holiday” was made in consultation with the patient's oncologist, reflecting the guideline's emphasis on a multidisciplinary approach and the recognition that interrupting high-dose antiresorptive therapy in oncology patients may not always be feasible due to the risk of skeletal-related events.
Our patient’s profile, an elderly female with metastatic breast cancer, aligns with demographic patterns observed in larger MRONJ cohorts. A systematic review found that 71% of MRONJ cases occur in females, with a mean age of 66.3 years, reflecting the higher prevalence of osteoporosis and breast cancer in this population [14]. Mention is to be made of the fact that the mandible is more affected than the maxilla, as in the here-presented patient, due to its rich vascularization and greater stress compared to the maxilla, accounting for 60% to 75% of MRONJ cases [14,17]. As widely accepted, it is worth noting that comorbid conditions such as diabetes mellitus, hypertension, and various forms of immunosuppression increase the risk of developing MRONJ due to a decreased capacity of natural wound-healing mechanisms [17,25]. The aforementioned leads to a conclusion that various risk assessment tools, considering age and gender, have an important role to play for patients undergoing antiresorptive therapy.
The critical preventive strategy involves risk stratification for patients who may require antiresorptive therapy in the future. For individuals with risk factors for osteoporosis or cancer, a pre-implant assessment should include a discussion about potential future medications. Whenever possible, completing all elective dentoalveolar surgeries, including implant placement, prior to initiating antiresorptive therapy, remains the hallmark of prevention. This approach is taken to ensure optimal oral health with minimal bone-disruptive therapy during treatment and to reduce the lifelong risk of MRONJ as much as possible [19, 26].
This case has critical implications for pre-implant risk assessment and informed consent. The AAOMS clearly categorizes patients receiving high-dose antiresorptive agents for metastatic bone disease as “high-risk,” for whom elective dentoalveolar surgery, including implant placement, is relatively contraindicated. Our patient's implant was placed before denosumab initiation, a scenario that still carries significant long-term risk. This serves to emphasize again the need for adequate informed consent, which involves not merely the usual, as well as potential, implant-related services, but also speaks to a perpetual, lifelong risk of late MRRONJ, even following years of asymptomatic use. For those primarily taking high-dose antiresorptives, there is substantial advice against new implant placement. For those with existing implants, this case mandates indefinite, lifelong monitoring by both dental and medical providers to detect and manage any signs of MRONJ at its earliest, most treatable stage.
This case report provides valuable longitudinal documentation of late-onset MRONJ development around a previously stable implant under denosumab therapy, offering practical insights into surgical management without antiresorptive discontinuation in high-risk oncology patients. However, as a single-case observation, its findings lack generalizability and require validation through larger prospective studies. The inability to provide serial bone turnover markers limits correlative biochemical analysis. The absence of the above adjunctive therapies, such as teriparatide or new biomaterials, within this manuscript presents certain opportunities for further therapeutic exploration in future studies. Established osteonecrosis in high-dose oncology patients is often resistant to conservative management alone. In contrast, surgical intervention, as illustrated in this case, can result in definitive resolution without interruption of antiresorptive therapy. These limitations underscore the need for multidisciplinary collaboration to implement standardized care for this complex patient population.
CONCLUSION
This case underscores that dental implants in patients receiving high-dose antiresorptive therapy represent a lifelong, perpetual risk for MRONJ, even after years of asymptomatic function. The primary clinical takeaway is the critical need for rigorous risk assessment before initiating antiresorptive agents, including a comprehensive dental examination and completion of any invasive procedures. For patients with existing implants, thorough patient education is mandatory; informed consent must explicitly detail the possibility of late-onset MRONJ to ensure shared decision-making and lifelong vigilance. Finally, this case mandates a paradigm of indefinite, multidisciplinary, long-term monitoring involving both dental and oncology teams to enable early detection and intervention. For established cases, surgical sequestrectomy with implant removal proved to be a definitive and successful treatment, achieving complete resolution even while the patient continued denosumab therapy.
AUTHORS’ CONTRIBUTIONS
The authors confirm their contributions to the paper as follows: Z.N.A., D.B., Z.I.A., H.A.H., N.E.T., L.M., A.A.F.W.: Study conception and design; Z.N.A., D.B., Z.I.A., H.A.H., N.E.T.: Data collection; H.A.H., N.E.T., L.M., A.A.F.W.: Analysis and interpretation of results; Z.A.: Draft manuscript. All authors reviewed the results and approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| CBCT | = Cone-Beam Computed Tomography |
| MRONJ | = Medication-Related Osteonecrosis of the Jaw |
| BPs | = Bisphosphonates |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The ethical approval for publication of this case report was provided by the Ethical Review Board of Tishreen University Hospital with Number 402 on 21 June 2025.
HUMAN AND ANIMAL RIGHTS
In accordance with the ethical standards of institutional and/or research committee and with the 1975 Declaration of Helsinki, as revised in 2013.
CONSENT FOR PUBLICATION
Written informed consent was obtained from the patient for publication of this case report and accompanying images.
ACKNOWLEDGEMENTS
Declared none.


