All published articles of this journal are available on ScienceDirect.
Reliability of Gingival Crevicular Fluid Biomarkers for Detecting the Pubertal Growth Spurt: A Critical Review
Abstract
Aim
In recent years, components of gingival crevicular fluid (GCF), including alkaline phosphatase (ALP), serotransferrin (TF), and vitamin D-binding protein (DBP), have attracted attention as biomarkers for determining stages of pubertal growth in orthodontic treatments. In this review article, we critically evaluated the reliability of biomarkers present in GCF for the accurate identification of the pubertal growth spurt.
Materials and methods
A systematic search of scientific sources was conducted in the PubMed, Web of Science, and Scopus databases. In this search, the relationship between biomarkers present in gingival crevicular fluid and key stages of skeletal development, particularly the mid-pubertal stages of jaw growth (CS3–CS4 stages in the CVM index), was examined. The main focus of this article was to determine the association of biomarkers such as ALP with specific maturation milestones in the cervical vertebrae.
Results
The results obtained from the literature search in this article showed that there is a significant association between the concentration of some GCF biomarkers and the peak growth velocity.
Discussion
If the obtained data are confirmed, it can be expected that the diagnostic accuracy and treatment timing in growth-modification-based orthodontic interventions will improve considerably.
Conclusion
This non-invasive diagnostic approach appears to have promising clinical potential. However, its current clinical application is limited due to heterogeneity in study methodologies. Standardized protocols need to be developed as a fundamental prerequisite for the reliable use of these biomarkers in routine orthodontic practice.
1. INTRODUCTION
One of the determining factors in the success of dentofacial orthopedic treatments is the accurate identification of the onset of the pubertal growth spurt. In conventional methods, skeletal maturation is evaluated using radiographic techniques such as the cervical vertebral maturation (CVM) index or hand-wrist radiography [1]. Despite their widespread use, these methods face challenges, including patient exposure to ionizing radiation and variability in image interpretation due to their subjective nature. These challenges can lead to diagnostic errors in estimating skeletal age [2].
The existence of such limitations has increased interest in non-invasive and biochemical approaches. In this context, gingival crevicular fluid (GCF) appears to be a promising source. Its collection is simple and painless, and it contains a variety of metabolic biomarkers that reflect biological changes in the tissues surrounding the teeth during different stages of growth [3-5]. Recent studies have shown that specific biomarkers present in GCF, such as alkaline phosphatase (ALP), vitamin D-binding protein (DBP), and transferrin (TF), are of particular importance [5].
Studies have demonstrated that the levels of these proteins in GCF are associated with key stages of skeletal maturation. These indicators may be used to identify the onset or peak of the pubertal growth spurt [6-8]. It should be noted that transferring this method from the laboratory to the clinical setting is not straightforward. Establishing a consistent and reliable approach is difficult because of differences in sampling methods, the types of biomarkers measured, and the analytical techniques used [9].
Skeletal maturation during growth is a complex process. It is influenced by genes, hormones, and environmental factors [10]. Conventional radiographic methods only provide a general picture of bone changes and cannot show the real biological and cellular activities that drive bone growth [11]. Recent advances in molecular biology have helped identify biomarkers that give a more accurate and real-time view of the body’s physiological status [12]. Gingival crevicular fluid (GCF) is especially valuable because it is in direct contact with periodontal tissues and is rich in proteins and signaling molecules involved in bone remodeling. Using these biomarkers in orthodontics can change the way growth is assessed. This allows clinicians to choose the timing of treatment more precisely and in a more personalized way, ultimately leading to better outcomes for patients [13, 14]. However, it should be noted that scientific reports regarding the accuracy and reproducibility of these biomarkers have yielded conflicting results. This has limited their widespread acceptance in clinical settings.
The aim of this article is to provide a critical review of the available evidence regarding the reliability of GCF biomarkers in determining stages of pubertal growth. The results of this study clarify whether these tools can serve as reliable alternatives to conventional radiographic methods. The process is illustrated schematically in Fig. (1).
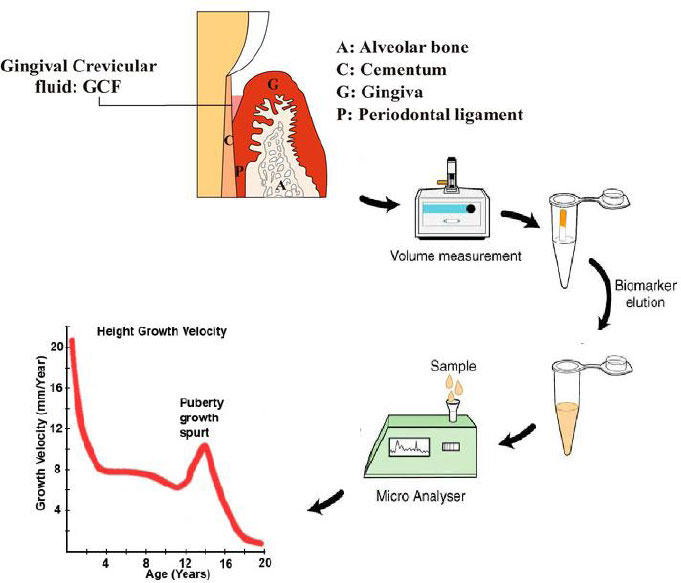
A schematic process of the gingival crevicular fluid biomarkers measurement for detecting the pubertal growth spurt.
2. MATERIALS AND METHODS
This study was designed as a critical review with the aim of evaluating the available evidence regarding the reliability of gingival crevicular fluid (GCF) biomarkers in identifying the pubertal growth spurt. A systematic search was conducted in the PubMed, Web of Science, and Scopus databases to identify and review relevant studies. The search strategy included combinations of keywords related to “gingival crevicular fluid,” “biomarkers,” “pubertal growth spurt,” “cervical vertebral maturation,” and “orthodontic growth assessment.” In addition, the reference lists of selected articles were examined to identify additional relevant studies that might not have been captured in the initial search. Inclusion criteria consisted of studies that directly investigated the relationship between biomarkers present in GCF and skeletal maturation indicators, particularly stages of cervical vertebral maturation. Studies focusing on human populations and reporting quantitative data on biomarker levels at different stages of growth were included in the final analysis. Studies that lacked clear data regarding skeletal maturation stages, as well as those that evaluated only salivary or serum biomarkers without assessing GCF, were excluded from this review. In addition, case reports, review articles, and studies with insufficient methodological information were also excluded.
After selecting the eligible articles, relevant data, including the type of biomarker studied, the GCF sampling method, laboratory techniques used for measuring biomarker concentrations, and the skeletal maturation stage of the participants, were extracted and compared. The aim of this step was to identify common patterns as well as methodological differences among studies that could influence the interpretation and generalizability of the results.
3. RESULTS AND DISCUSSION
Although the biochemical composition of gingival crevicular fluid (GCF) is related to skeletal maturation, this relationship is complex, and establishing a definite link is still difficult. This section examines this relationship. The main focus is on identifying important biomarkers. The methodological limitations that prevent their consistent validation are also discussed.
3.1. Biological Basis of GCF Biomarkers
Gingival Crevicular Fluid (GCF) is a naturally occurring fluid that comes from the gingival sulcus and the surrounding epithelial layers. This fluid serves as an important source of biochemical markers-markers that reflect both changes in periodontal tissues and the overall physiological condition of the body. The composition of GCF is constantly and dynamically influenced by factors such as blood vessel permeability, the presence and activity of immune cells, and hormonal signals. This complex nature makes GCF a highly valuable diagnostic medium for assessing skeletal maturation and orthodontic-related changes [15]. At the same time, although GCF is strongly affected by local oral conditions, it also responds to systemic changes in the body. These include hormonal fluctuations and bone metabolic changes that occur during growth. For example, signaling pathways involving Growth Hormone (GH) and Insulin-like Growth Factor 1 (IGF-1)-which play key roles in stimulating osteoblasts and forming bone matrix-can influence the enzymatic and cellular profile of GCF by altering tissue blood flow and vascular reactivity [16].
Although the numerical data in the meta-analysis came only from blood tests, Kapoor’s team also noticed interesting patterns in the GCF results. They observed that the levels of some markers in the gingival fluid, such as ALP, DBP, and serotransferrin, increase noticeably during puberty [17-19].A major problem in current studies is that each research group uses a different method to measure GCF markers. These differences make it impossible to combine the data and perform a strong statistical analysis. Because of this, even though GCF has high potential, the existing evidence is mostly observational rather than solid statistical proof. This gap shows that we urgently need standardized, long-term studies that focus specifically on GCF changes over time [19].
During the pubertal growth spurt, bone formation and bone remodeling increase significantly. Since the tissues around the teeth are closely connected to this process, the biochemical components of GCF provide a local and precise picture of the body’s biological state. For this reason, many believe that if these signals are correctly interpreted, GCF can become an important tool for identifying the best biological timing for orthodontic treatment [20].
The site-specific nature of GCF also creates a major clinical advantage in orthodontics. Alveolar bone remodeling directly affects tooth movement, tooth stability, and craniofacial development [20, 21]. This close biological connection has increased interest in using GCF biomarkers for personalized orthodontic treatments and better clinical outcomes.
3.2. Key GCF Biomarkers Related to Growth Spurts
This section of the review focuses on three key biomarkers found in gingival crevicular fluid (GCF): Alkaline Phosphatase (ALP), Transferrin (TF), and Vitamin D-Binding Protein (DBP). These molecules were selected because they have important roles in bone metabolism and have been associated with skeletal growth and maturation. ALP reflects the activity of bone-forming cells, TF is involved in iron transport and cell growth, and DBP regulates vitamin D transport and may influence bone remodeling.
3.2.1. Alkaline Phosphatase (ALP)
Among GCF biomarkers, alkaline phosphatase (ALP) has received the most research attention due to its well-established role in bone mineralization. Most studies, including those by Perinetti et al., consistently report a peak in GCF ALP activity during the cervical stages CS3–CS4 [17]. Although this pattern is promising, it is important to note that many of these studies are cross-sectional and use different methods for measuring ALP activity, which makes direct comparison difficult [17-19].
Sonwane and Bhad systematically reviewed the diagnostic potential of GCF ALP activity for assessing skeletal maturity. Across the six included studies, a consistent pattern emerged: ALP levels showed a statistically significant increase precisely during the pubertal growth period, strongly correlating with CVM stages 3 and 4. This review confirmed the central biological role of ALP in bone remodeling and strengthened its position as a viable, non-invasive indicator for identifying growth spurts. However, the original authors cautioned that limitations such as small sample sizes, reliance on cross-sectional designs, and variation in assay protocols require larger, longitudinal studies to confirm these preliminary findings [22]. Perinetti and Contardo et al. (2011, n=50) reported a significant peak in total GCF ALP activity at pubertal stages CS3–CS4, independent of dentition phase or jaw location (PubMed). However, when ALP was normalized to protein content, its association with growth stage became weaker, highlighting the importance of reporting both absolute and relative enzyme activity [23].
3.2.2. Vitamin D Binding Protein (DBP) and Serotransferrin (TF)
Recent research using proteomic methods has shown that serotransferrin (TF) and vitamin D–binding protein (DBP) are present in gingival crevicular fluid (GCF) and can serve as useful markers for detecting puberty. DBP is important because it carries vitamin D metabolites and affects osteoclast activity. TF is important because it transports iron and supports cell proliferation, making it involved in bone metabolism and skeletal growth [24-26].
To improve skeletal maturity assessment, Wen and Gu examined the ability of TF and DBP to identify the pubertal growth peak. This study included 66 orthodontic patients (32 Class I and 34 Class II). Patients were divided into prepubertal, pubertal, and postpubertal stages based on CVM. TF and DBP concentrations in GCF were measured using ELISA. The results showed that TF concentration increases clearly during the pubertal stage, and this pattern appeared in both malocclusion groups. But the relationship of DBP with the pubertal stage was less clear. In this study, TF alone was the most accurate diagnostic marker, performing better than DBP and chronological age. Combining TF level with chronological age further increased predictive power. Threshold analysis identified 4.20% in the maxilla and 4.09% in the mandible as the best cut-off values to distinguish pubertal from non-pubertal stages [26]. These percentages indicate the ratio of TF concentration to total GCF protein, determined through ROC analysis. These findings show that TF in GCF can be a sensitive and precise marker of the pubertal stage. Its accuracy becomes much higher when combined with chronological age. This is very important for orthodontic treatment planning, especially in Class II cases where timing is critical. Finally, the AUC of TF alone was 0.82, and when age was added, it increased to 0.91, showing the value of combining a biomarker with demographic data [27].
3.2.3. Other Potential Biomarkers
Researchers haven’t limited themselves to the well-known factors. For example, consider insulin-like growth factor-1 (IGF-1). This molecule plays an important role in overall growth, and its link to skeletal maturation in blood and saliva is well established. But detecting it in GCF and understanding what its levels really mean is still unclear [28].
Enzymes like MMP-8 and MMP-9 also take part in tissue breakdown and rebuilding. In theory, their levels in GCF should rise during growth spurts. But study results vary a lot. This is probably because different research teams collect and process their samples in very different ways [29].
Inflammatory markers such as IL-1β, TNF-α, and OPG have also been studied. They play roles in bone remodeling. But the problem is that their increase might be due to normal growth or just gum inflammation. It’s hard to tell the difference, and that’s a major challenge [30-31].
Molecules like RANKL, osteocalcin, and TGF-β also show good potential. They might provide a clearer picture of bone metabolism through GCF. Including them in future studies could help us better understand the molecular pathways involved in puberty [32, 33].
3.3. Methodological Challenges
The use of gingival crevicular fluid biomarkers for identifying the pubertal growth spurt has produced promising results. However, several methodological challenges still prevent the widespread acceptance of this approach in clinical settings. One of the most important challenges is the lack of standardized protocols for collecting and analyzing GCF samples. Various methods, including absorbent paper strips, micropipettes, and other collection tools, have been used across different studies, each of which can influence the volume and composition of the samples.
3.3.1. Sampling Site Variability
Another factor contributing to variation in results is the sampling site. In some studies, samples were collected from anterior maxillary teeth, whereas in others they were obtained from posterior teeth or from both dental arches. Local periodontal tissue conditions vary in different regions of the oral cavity; therefore, these differences may affect the concentration of biomarkers measured in GCF [34, 35]. In another study, only the central incisors were examined to keep the conditions consistent. However, differences between the upper and lower teeth were still observed [34].
3.3.2. Collection Technique
The method used for collecting gingival crevicular fluid (GCF) is of great importance. One of the most common methods is the use of small paper strips or paper points that are placed inside the gingival crevice. However, how deep the strip is inserted, how long it remains in place, or whether it becomes contaminated with saliva or blood can affect the volume and composition of the sample [36]. In a review article, it was examined that studies commonly use these paper strips, but some studies have also used other methods, such as microcapillaries [36]. If a standard and uniform protocol for sample collection does not exist, it makes it difficult to determine whether the differences observed between studies are real or simply caused by differences in the collection method [36].
3.3.3. Normalization and Quantification Methods
Quantification and data normalization methods represent another major challenge. [37]. The volume of collected GCF is usually very small and can be affected by factors such as gingival inflammation, oral hygiene status, and mechanical pressure during sampling. [38]. As a result, different normalization approaches may be used across studies, which complicates the comparison of results [39].
3.3.4. Analytical Techniques
Our review also showed that laboratory techniques used to measure biomarkers differed among studies. Many studies used immunoassay methods such as Enzyme-Linked Immunosorbent Assay (ELISA), whereas others employed more advanced proteomic techniques. Differences in the sensitivity, specificity, and accuracy of these methods may contribute to discrepancies in reported results [40]. Many factors are involved in the inconsistency of test performance. These factors include the type of antibody clones used, the test calibration protocol, and the inherent purity of the chemical reagents. Each of these factors may introduce significant bias into the obtained measurements. It is also possible that before the test is performed, the way samples are stored, handled, or repeatedly frozen and thawed may damage sensitive biomarkers and make the data unreliable [40]. ALP assays used in one study showed that rapid maxillary expansion (RME) studies differed in kit calibration and sample preparation methods, which resulted in reporting different levels of ALP. Pre-analytical factors such as sample storage methods and freeze–thaw cycles can also greatly damage sample integrity [41].
3.3.5. Study Design Limitations
A large portion of the existing research has been conducted as cross-sectional studies with a small number of participants. These types of studies only show the situation at a single point in time and cannot properly follow the changes of biomarkers throughout different stages of growth [42]. For this reason, there is not enough information about the temporal pattern of these changes. Therefore, conducting longitudinal studies is very important. In such studies, researchers follow the same individuals over a longer period of time, which allows them to better determine the relationship between changes in GCF biomarkers and the timing of peak growth velocity and to evaluate their predictive value more accurately.
Most of the conducted studies are also cross-sectional and have limited sample sizes. In some of these studies, the number of participants was small, and the study design did not include blinding or control groups, which can reduce the statistical strength of the results and limit understanding of changes over time [43]. The longest study investigating ALP in GCF during RME treatment followed 23 prepubertal patients for six months during the retention period and showed that the level of this biomarker remained stable during this time. This finding highlights the importance of using longitudinal designs in research [44].
3.3.6. Standardization Imperative
To advance this field, the most important step is to define clear and unified standard protocols [5, 45]. Researchers need to reach an agreement on the best sampling sites, the proper method for collecting the fluid without contamination, and also the way the data should be normalized and reported. In addition, conducting large and long-term studies in multiple centers is necessary to confirm the reliability of these biomarkers and to provide sufficient evidence for their use in clinical settings [46]. Table 1 summarizes these key methodological challenges and sources of variability.
| Category | Challenges | Examples / Implications | Proposed Solutions | Ref. |
|---|---|---|---|---|
| Sampling Site Variability | Differences in GCF biomarker levels between maxilla vs. mandible; anterior vs. posterior teeth | Inconsistent ALP levels depending on collection site | Standardize anatomical sampling sites, e.g., mandibular incisors | [47] |
| Collection Technique | Variations in insertion depth, duration, contamination by saliva or blood | Blood contamination skewing biomarker levels | Strict protocol for collection time and contamination control | [48] |
| Normalization and Quantification | Reporting per volume, protein content, or enzyme activity; variable GCF volume | Different units limit comparability | Adopt uniform normalization metrics (e.g., per total protein) | [49] |
| Analytical Techniques | ELISA assay variability due to antibody specificity, calibration, storage conditions | Sample degradation with freeze-thaw cycles | Use standardized reagents and sample handling protocols | [50] |
| Study Design Limitations | Mostly cross-sectional, small samples, lack of longitudinal data | Limited insight into biomarker changes across growth stages | Conduct larger, longitudinal multicenter studies | [51] |
| Standardization Imperative | Lack of consensus on protocols and reporting | Hinders reproducibility and clinical adoption | Develop international guidelines for GCF biomarker studies | [52] |
4. COMPARATIVE RELIABILITY WITH STANDARD SKELETAL MATURITY INDICES
The conversion of gingival crevicular fluid (GCF) markers from preliminary findings into reliable indicators for predicting skeletal growth is possible when it is shown that the changes in these markers are temporally aligned with established radiographic indicators, particularly the Cervical Vertebral Maturation (CVM) stages and Fishman’s Skeletal Maturity Indicators (SMI) [53]. Alkaline phosphatase is one of the most extensively studied biomarkers in gingival crevicular fluid in relation to the pubertal growth spurt. Several studies have reported increased ALP activity during the mid-stages of skeletal maturation, particularly at the CS3 and CS4 stages of the cervical vertebral maturation index [17, 54-56]. This increase in enzymatic activity is consistent with active bone formation processes that occur during periods of rapid growth.
Despite some promising findings from studies on gingival crevicular fluid (GCF), the available evidence is still moderate in both quality and quantity. One of the main problems is the variation in methods among studies. Differences in participant characteristics, especially age, sampling methods, and biomarker measurement techniques, make comparisons difficult. Many studies also have small sample sizes. This reduces statistical power and limits how widely the results can be applied. Another limitation is the lack of proper research controls in some studies. In several cases, adequate blinding was not used. Important confounding factors were also not always controlled. Factors such as periodontal status, systemic diseases, and nutritional status can influence the expression of GCF biomarkers.
One of the most important gaps is the lack of longitudinal studies. In many studies, biomarker changes were not followed over time in the same individuals. Such studies are necessary to track how biomarkers change during different stages of growth. They also help distinguish normal physiological fluctuations from real biochemical changes related to growth.
Considering these limitations, current evidence mainly supports a supportive role for GCF biomarkers such as ALP rather than replacing standard methods. At present, these biomarkers cannot replace conventional radiographic techniques. However, they show considerable potential as complementary diagnostic tools. Combining biochemical data from GCF with routine imaging may help clinicians assess skeletal maturation more accurately. This combined approach may also reduce radiation exposure by allowing longer intervals between radiographs, while providing additional insight into local bone metabolism [57]. One study clearly demonstrates this potential. Perinetti and colleagues examined 72 healthy individuals. In this study, GCF samples were collected from the anterior teeth of both the maxilla and the mandible. The results were then compared with skeletal maturity stages determined by radiography using the cervical vertebral maturation (CVM) method. The researchers observed a clear pattern. ALP activity increased about twofold during the critical skeletal stages CS3 to CS4. This pattern was observed in both the maxilla and the mandible. While radiographs often indicate the onset of the pubertal growth spurt, the marked increase in ALP activity provides important biochemical confirmation of these findings. In clinical practice, such additional evidence can increase the clinician’s confidence when determining the optimal timing for orthopedic treatments, such as rapid maxillary expansion. This study shows that GCF analysis can add valuable objective information to radiographic assessment and support more informed clinical decisions [58].
5. CLINICAL RELEVANCE AND FEASIBILITY
When gingival crevicular fluid is used for the assessment of skeletal maturity, it has clear advantages. First, this method is completely non-invasive. This issue reduces the concern associated with working with children and adolescents [17, 18]. Second, exposure to ionizing radiation is eliminated in this method, which is an advantage compared with traditional radiographic methods [55]. Also, because of the simplicity of this method, it is possible to collect samples at several different time points. This allows specialists to monitor the growth process dynamically over time [3, 20].
Before the GCF test can move from research environments into routine orthodontic clinics, its major problems must be resolved. The absence of rapid chairside diagnostic tools is one of the most important of these challenges. It should be noted that GCF analysis requires laboratory procedures, including centrifugation, ELISA kits, and special storage conditions. In most orthodontic clinical settings, these conditions are not feasible. Studies on point-of-care diagnostic devices are ongoing, but they are not yet commercially available [59].
Since the volume of collected GCF is very small, it is highly susceptible to contamination. Even minor gingival bleeding, which may occur due to inflammation or the sampling method, can change the levels of biomarkers such as ALP or IGF-1 and distort the results [3]. Therefore, developing and following precise and standardized sampling protocols appears necessary. In addition, clinicians should be trained in uniform and proper methods of sample collection [60].
CONCLUSION AND FUTURE OUTLOOK
In recent years, gingival crevicular fluid biomarkers have been investigated for identifying the timing of the pubertal growth spurt. According to the available evidence, several biomarkers, including alkaline phosphatase, transferrin, and vitamin D-binding protein (DBP), may be associated with different stages of skeletal maturation. Among these biomarkers, ALP has been one of the most extensively studied due to its association with bone formation activity, and increased activity has been reported near the peak growth period. Additionally, according to recent studies, TF may show considerable diagnostic accuracy for identifying the pubertal growth spurt stage. Evidence regarding DBP, however, still appears to be more limited. It should be noted that significant limitations remain in the current body of research. Differences in sampling methods, laboratory techniques, and study designs have made direct comparison of results across studies difficult. Most of the available studies have used cross-sectional designs, and only a limited number of longitudinal studies have examined biomarker changes over time. There is still insufficient evidence to support the independent use of GCF biomarkers for biological growth assessment. Conducting longitudinal studies with larger sample sizes, together with the standardization of sampling protocols and analytical methods, may clarify the true role of these biomarkers in the assessment of skeletal maturation.
AUTHORS’ CONTRIBUTIONS
The authors confirm contribution to the paper as follows: A.M., A.G., A.H.H.: Study conception and design; A.M., A.G., A.H.H.: Data collection; A.M., A.G., A.H.H.: Analysis and interpretation of results; A.M., A.G., A.H.H.: Draft manuscript. All authors reviewed the results and approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| CVM | = Cervical vertebral maturation |
| ALP | = Alkaline phosphatase |
| GCF | = Gingival crevicular fluid |
| SMI | = Skeletal maturity indicators |
AVAILABILITY OF DATA AND MATERIALS
Upon request to the corresponding author following publication, the raw and processed data necessary to replicate these findings will be made available.
CONFLICT OF INTEREST
No potential conflicts of interest, whether financial or non-financial, were declared by the authors.
ACKNOWLEDGEMENTS
Declared none.


