All published articles of this journal are available on ScienceDirect.
Time-dependent Tooth Color Changes Following Conventional, Silver-based, and Photodynamic Root Canal Irrigants: An In Vitro Study
Abstract
Introduction
Discoloration of endodontically treated teeth is an aesthetic concern. The primary causes include persistent pulpal tissue breakdown and the use of various irrigants. This study compared the effects of different endodontic irrigants on tooth color stability over time.
Materials and Methods
Eighty single-rooted human central incisors were allocated into eight groups (G): G1, G2, G3, G6, and G7 used sodium hypochlorite and silver-based compounds; G4 received Photodynamic Therapy (PDT); G5 received combined PDT and NaOCl; and G8 was treated with 0.9% normal saline. Tooth shade was assessed at baseline (T0), after 1 (T1), 7 (T2), 14 (T3), and 28 days (T4) using a spectrophotometer.
Results
At T1, G5 showed the least color change (ΔE), while G2 had the most (p <0.05). By T2, G1 had lower ΔE values than G3 and G7 (p <0.05). At T3, G5 showed the least ΔE changes compared to G2, G3, G6, and G7 (p <0.05). Conversely, G7 had the highest ΔE compared to G1 and G4 (p <0.05). At T4, G7, G6, G2, and G3 exhibited higher ΔE than G5, G4, G1, and G8 (p <0.05).
Discussion
Silver-based irrigants caused significant darkening due to silver deposits in dentinal tubules and silver-protein precipitates. Sodium hypochlorite caused fewer changes due to its crystallization potential. Photodynamic therapy led to color changes above clinical thresholds, but its combination with NaOCl reduced discoloration to imperceptible levels. Silver-based irrigants should be used cautiously in anterior teeth due to their high staining potential.
Conclusion
Root canal irrigants induced varying degrees of tooth color changes. Among them, silver fluoride caused the most significant early darkening, while the combination of photodynamic therapy with sodium hypochlorite resulted in minimal darkening over time.
1. INTRODUCTION
Discoloration of teeth following endodontic treatment is a significant concern for patients [1]. Changes in tooth color are a primary contributor to patient dissatisfaction. The clinical perception of detectable color changes is crucial in addressing the growing aesthetic concerns of patients of all ages, especially among parents of pediatric patients and adolescents [2]. The main causes of discoloration include the persistence of remaining pulpal tissues and internal pulpal hemorrhage. Additionally, materials and chemicals used during various treatment steps can also contribute to the discoloration of endodontically treated teeth. Some obturation materials, intracanal medications, temporary fillings, and irrigants can lead to internal dentin discoloration, influencing the choice of irrigants based on their aesthetic outcomes [3].
Sodium hypochlorite (NaOCl 0.5% - 5.25%) is widely regarded as the standard irrigant and is the most commonly used irrigant in dentistry [4]. It possesses strong antimicrobial properties and unique tissue-dissolving capabilities [5]. However, due to its limited ability to remove the dentin smear layer, there is a growing need for additional irrigants or conjugation regimens [4]. Silver diamine fluoride (SDF) has been investigated as a root canal irrigant for its potent antibacterial properties and ability to enhance root canal disinfection [6]. Silver nanoparticles have various biomedical applications due to their antimicrobial effects, particularly against E. faecalis, and their ability to penetrate dentinal tubules [7]. Photodynamic therapy (PDT) for root canal disinfection relies on activating a photosensitizer with specific wavelengths of light to generate reactive oxygen species that rupture and kill bacterial cells. The method of light delivery can affect the efficiency of root canal disinfection. For instance, optical diffuser fibers inserted into the root canal have shown effective microbial reduction compared to laser tips placed at the cavity access, making them suitable for endodontic disinfection [8, 9].
Various methods can assess color stability, including visual inspection to compare tooth color before and after treatment, spectrophotometry to measure light absorption and reflection at specific wavelengths, and colorimetry to quantify color using standardized color spaces. A staining test can expose the tooth to staining agents and assess color changes [10]. The spectrophotometer measures spectral reflection based on color wavelengths, while the CIELAB color space serves as a precise tool for assessing color changes in dentistry [11].
Limited studies have evaluated the effect of various root canal irrigants on changes in tooth color over time. Therefore, the aim of this study is to evaluate the effect of various endodontic irrigants on the color stability of teeth over time. The null hypothesis states that no significant differences in color change will be observed among the irrigants.
2. MATERIALS AND METHODS
2.1. Ethical Statement
This study was conducted at the Faculty of Dentistry, Umm Al-Qura University, Saudi Arabia. Ethical approval was granted by the local Biomedical Research Ethics Committee at Umm Al-Qura University (HAPO-02-K-012-2023-01-1394), in accordance with the Declaration of Helsinki regarding the use of human teeth in research.
2.2. Sample Size Calculation
The ClinCalc tool (ClinCalc LLC, Chicago, Illinois, USA) was used for sample size calculation, determining a minimum of ten teeth per group to achieve a study power of 80% with a two-sided α level of 0.05. The mean color change ΔE in the control (saline) group was 5.4 ± 0.78, with an anticipated 20% increase in ΔE expected with the use of the SDF irrigant [12].
2.3. Teeth Preparation
Eighty sound single-rooted human maxillary central incisors with fully developed root apices, extracted for periodontal reasons, were selected for the study. Teeth with caries, restorations, coronal staining, cracks, fractures, previous endodontic therapy, or root resorption were excluded. The teeth were anonymously donated and collected from the Dental Hospital at Umm Al-Qura University.
All teeth were ultrasonically cleaned to remove calculus and soft tissue debris, then polished with a rubber cup and pumice to eliminate any remaining debris and stains. The teeth were preserved in 0.9% normal saline until instrumentation to prevent dehydration. An access cavity was prepared for each tooth, and a glide path was established for the root canal using stainless-steel K-files up to size #25 (Dentsply Maillefer, Ballaigues, Switzerland). Canal preparation was performed using the ProTaper Next rotary system (Tulsa Dentsply, Tulsa, OK, USA) with files X1, X2, X3, and X4. During instrumentation, 5 ml of normal saline was used for irrigation, administered via a disposable syringe fitted with a 23-gauge side-vented needle tip (Endo EZ, Ultradent™ Products Inc., South Jordan, Utah, USA), which was passively positioned in the canal. Each tooth was vertically inserted to the level of the cementoenamel junction in a plastic cylindrical mold, 1.5 cm in diameter and 2.00 cm high, filled with dental stone and left to harden at room temperature.
2.4. Specimen Grouping
Samples were randomly assigned to eight groups (n=10/group) based on the type of irrigant used. Table 1 lists the irrigant agents used in this study. The irrigation routine for each group was as follows: G1: 2.5% NaOCl, G2: 3.8% Silver diamine fluoride (SDF), G3: Silver nanoparticles irrigation solution (AgNP), G4: Photodynamic therapy (PDT) with methylene blue, G5: Combined PDT and 2.5% NaOCl (PTNaOCl), G6: Combined 3.8% SDF and 2.5% NaOCl (SFNaOCl), G7: Combined silver nanoparticles and 2.5% NaOCl (NSNaOCl), and G8: 0.9% Normal saline (control group).
| Group | Code | Irrigating Agent | Manufacturer |
|---|---|---|---|
| G1 | NaOCl | 2.5% Sodium Hypochlorite | Chlorox, Cleanser company, Egypt |
| G2 | SDF | 3.8% Silver Diamine Fluoride | FAgamin, Tdequim, Industria Argentina |
| G3 | AgNP | 100 ppm Nano Silver Fluoride | Nanogate company, Cairo, Egypt |
| G4 | PDT | 25 μg/mL Methylene blue crystalline powder | Oxford Lab Fine Chem LLP, Maharashtra, India |
| G5 | PTNaOCl | 25 μg/mL Methylene blue crystalline powder and 2.5% Sodium Hypochlorite | Oxford Lab Fine Chem LLP, Maharashtra, India & Chlorox, Cleanser company, Egypt |
| G6 | SFNaOCl | 3.8% Silver Diamine Fluoride and 2.5% Sodium Hypochlorite | FAgamin, Tdequim, Industria Argentina & Chlorox, Cleanser company, Egypt |
| G7 | NSNaOCl | 100 ppm Nano Silver Fluoride and 2.5% Sodium Hypochlorite | Nanogate company, Cairo, Egypt & Chlorox, Cleanser company, Egypt |
| G8 | Control | 0.9% Normal Saline | FIPCO, Borg Al-Arab, Alexandria, Egypt |
For each specimen, 5 ml of irrigant was agitated in the canal for 10 minutes, followed by a rinse with 3 ml of sterile physiological saline. For photodynamic therapy with methylene blue, 100 μL at a concentration of 25 μg/mL was injected into the radicular canal, followed by diode laser emission directed within the canal. The intra-radicular tip of a diode laser device (Laser-HF, H. Hager & Werken GMBH & Co.) with a wavelength of 660 nm operated at 100 mW for 60 seconds, delivering nine joules of energy. The device was connected to an optical fiber with a diameter of 200 μm, initially positioned 1 mm short of the working length. A spiral movement from apical to cervical ensured proper light distribution. Each exposure lasted 60 seconds (20 seconds of exposure followed by a 10-second pause) and was repeated three times, followed by a rinse with 3 ml of sterile physiological saline [13]. The samples were blot-dried with a suitable paper point, and a sterile cotton pellet was placed in the access cavity, followed by a final seal with a 2 mm thick light-cured composite resin restoration (Shade A1, Filtek, 3M ESPE, USA) applied according to the manufacturer’s instructions. Each specimen was stored at room temperature in an individual airtight pouch under moist conditions to minimize the risk of dehydration-induced color changes [14].
2.5. Spectrophotometry Assessment
A clinical spectrophotometer, the VITA Easyshade Advance 4.0 (VITA Zahnfabrik, Bad Säckingen, Germany), was used to evaluate the color change of each specimen. The device was calibrated according to the manufacturer's instructions before each measurement to assess the baseline shade of the tooth before treatment and at various intervals during treatment. The instrument probe tip was positioned at a right angle on the middle third of the labial surface of each sample crown.
A custom-made silicone key fitted around the spectrophotometer’s mouthpiece standardized color measurements by controlling both the angulation and distance from the light source. This silicone key not only standardized the color measurements but also blocked the effect of external light during the evaluations [15]. All measurements were performed by a single, experienced operator who was blinded to the test groups. Specimens were blot-dried, and the “Tooth Single” program was selected for recording different shade parameters under constant natural daylight conditions.
Data were collected as a Commission Internationale de l’Eclairage (CIE) system using CIE L*a*b* coordinates, and the color change ΔE was calculated for each tooth at the selected time intervals; a baseline measurement was recorded right after canal preparation before irrigant regimen application (T0), followed by measurements taken after one day (T1), seven (T2), 14 (T3), and 28 days (T4), respectively, from the baseline record and after root canal irrigation procedure.
The L*, a*, and b* values were recorded three times for each sample, and an average value was calculated. The extent of color changes of each specimen was calculated using the following formula [11, 15]:
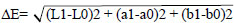 |
The L* axis reflects lightness, ranging from black (0) to white (100), the a* axis ranges from red (+a*) to green (-a*), and the b* axis ranges from yellow (+b*) to blue (-b*). The perceptibility threshold for detecting color changes was set above 3.3 ∆E units. Differences exceeding this threshold were considered clinically detectable [16].
2.6. Statistical Analysis
The mean (±SD) ΔE values were derived and statistically analyzed. The Shapiro-Wilk test was conducted to determine whether the data followed a normal distribution. Color changes (ΔE) were analyzed using one-way ANOVA to compare differences between test groups and repeated-measures ANOVA to assess changes within each group over time; Tukey’s post-hoc test was applied for multiple comparisons in both cases. Statistical analysis was performed using the Statistical Package for Social Sciences (SPSS) software version 23 (IBM, Armonk, USA), with an alpha error of 5% (p ≤ 0.05).
3. RESULTS
Color changes in all test groups over the study intervals are shown in Fig. (1). The mean ±SD values of ΔE for the test groups are presented in Table 2. The results revealed no statistically significant differences in baseline (T0) CIELAB values among the experimental groups (p > 0.05). The NaOCl, SDF, PDT, PTNaOCl, SFNaOCl, and control groups exhibited no significant differences in the rate of color changes at different assessment intervals within each group (p > 0.05). The AgNP and NSNaOCl groups showed significantly lower ΔE mean values at T1 (one-day interval) compared to T2 (7 days), T3 (14 days), and T4 (28 days) following the irrigant regimen (p < 0.01).
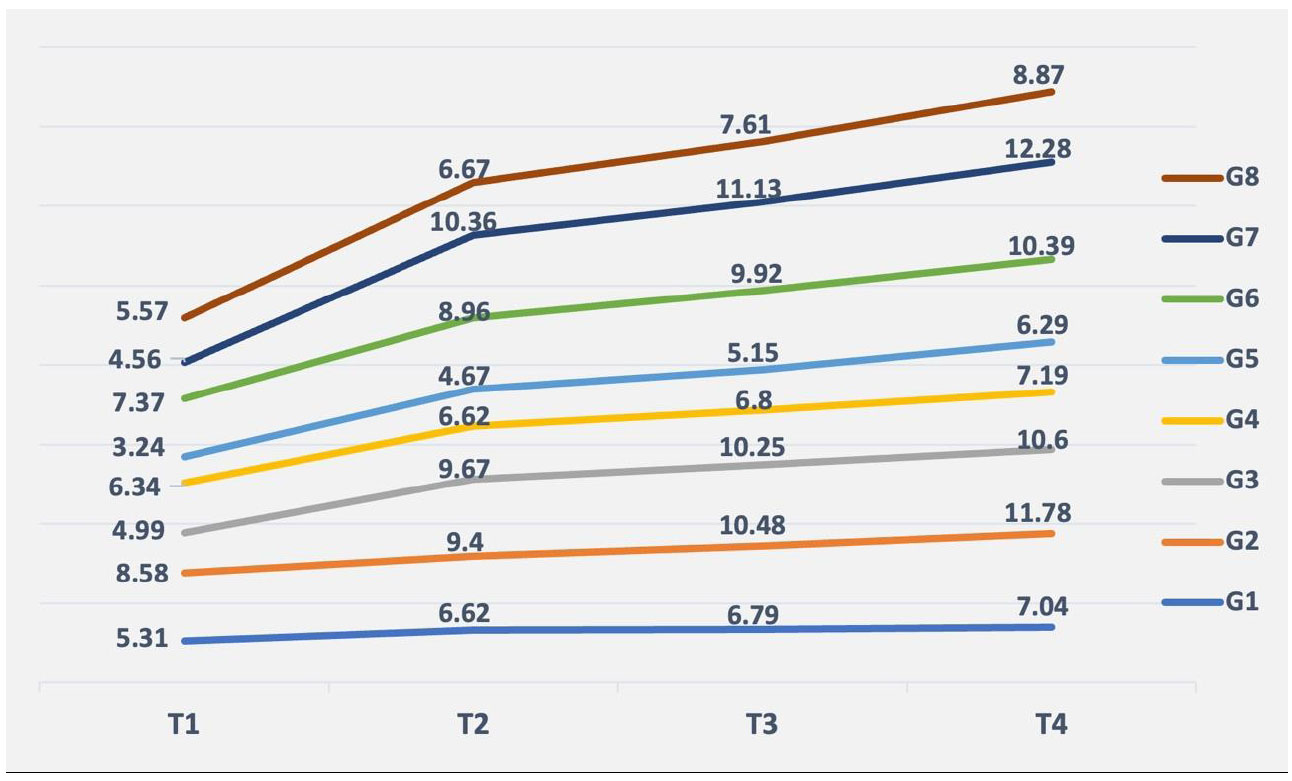
Color change (∆E) values (y-axis) of different test groups: G1: Sodium hypochlorite, G2: Silver diamine fluoride, G3: Silver nano-particles, G4: Photodynamic therapy, G5: Combined photodynamic therapy and sodium hypochlorite, G6: Combined silver diamine fluoride and sodium hypochlorite, G7: Combined silver nanoparticles and sodium hypochlorite, and G8: Normal saline at different time intervals (x-axis); T1:after one day, T2:after seven days, T3:after 14 days and T4:after 28 days.
| Group | Time Intervals | P-value | |||
|---|---|---|---|---|---|
| T1 | T2 | T3 | T4 | ||
| G1(NaOCl) | 5.3195±2.1800a,A | 6.6291±2.5096a,A,C | 6.7906±3.1332a,A,D | 7.0448±3.4678a,A | <0.05* |
| G2(SDF) | 8.5855±3.5371a,B,C | 9.4026±3.8059a,A | 10.4858±4.3431a,A,C | 11.7809±2.0672a,B | |
| G3(AgNP) | 4.9938±1.7465a,A | 9.6776±2.0968b,A | 10.2579±2.5477b,A,C | 10.6002±2.7962b,B | |
| G4(PDT) | 6.3466±1.9455a,A,C | 6.6201±2.8159a,A,C | 6.8003±2.2917a,A | 7.1999±2.8220a,A | |
| G5(PTNaOCl) | 3.2422±1.0969a,A | 4.6734±2.0442a,B,C | 5.1591±2.2113a,D | 6.2970±2.0086a,A | |
| G6(SFNaOCl) | 7.3746±3.2381a,C | 8.9698±3.4963a,A | 9.9280±2.2956a,A | 10.3902±2.4264a,B | |
| G7(NSNaOCl) | 4.5655±1.7700a,A | 10.3685±2.9505b,A | 11.1310±3.2154b,B,C | 12.2844±2.2143b,B | |
| G8(Control) | 5.5704±1.8017a,A,C | 6.6775±1.9854a,A,C | 7.6151±2.5257a,A,C | 8.8728±3.7501a,A | |
| P-value | <0.05* | - | |||
After one day, the SDF group displayed a significantly higher ΔE mean value compared to NaOCl, AgNP, PDNaOCl, and NSNaOCl (p < 0.05). The PDNaOCl group showed clinically undetectable changes, with a ΔE mean value not exceeding the perceptual threshold of 3.3, indicating this regimen is least likely to produce aesthetically concerning changes in the short term. By day seven, PTNaOCl consistently demonstrated the lowest ΔE values, significantly lower than those of the SDF, AgNP, SFNaOCl, and NSNaOCl groups, indicating sustained color stability (p < 0.05).
By day 14, the assessment of color changes showed the least ΔE discoloration in the PTNaOCl group compared to the SDF, AgNP, SFNaOCl, and NSNaOCl groups (p < 0.05). The NSNaOCl group presented the highest ΔE value compared to the NaOCl and PDT groups (p < 0.05). This suggests that the combination of silver nanoparticles and NaOCl may enhance staining effects over time. By day 28, the PTNaOCl and PDT groups exhibited the lowest ΔE values compared to the NSNaOCl, SDF, AgNP, and SFNaOCl groups (p < 0.05).
4. DISCUSSION
Tooth discoloration following endodontic treatment and retreatment is a common aesthetic concern, particularly among children and adolescents, due to the increasing aesthetic demands from patients and their parents. Visible tooth discoloration can negatively affect patients' self-confidence [2, 17].
Extracted human teeth were used to simulate clinical conditions [18, 19]. Prior to the irrigation protocols, all extracted teeth underwent baseline measurements to confirm the homogeneity of the samples’ initial shade and ensure that subsequent color changes were attributable to the impact of root canal irrigants.
The complex color of tooth structure was measured using the VITA Easyshade, which employs the L*, a*, and b* color systems to depict each color in three dimensions [11]. This device is highly accurate and reliable, eliminating subjectivity in color assessment. It provides quantitative values to calculate the ΔE value, indicating overall variations between final and baseline values [20, 21]. The accuracy and reliability of VITA Easyshade surpassed 90% of other shade evaluation methods and offered significantly higher inter-rater and intra-rater reliability than visual methods [22, 23]. The baseline assessment confirmed that all test groups started with comparable tooth shades, ensuring that any subsequent color changes could be attributed solely to the tested irrigant regimens.
Based on the human eye’s ability to perceive color changes, ΔE* < 1 is considered undetectable; 1.0 < ΔE* < 3.3 can be identified only by an expert and is clinically acceptable. However, ΔE* > 3.3 is regarded as a noticeable change in color [16].
The findings of this study supported the rejection of the null hypothesis, demonstrating different impacts of various root canal irrigants on tooth color stability assessed at 1, 7, 14, and 28 days post-baseline. All groups showed a clinically detectable change in tooth color at different intervals, except for the early assessment of combined photodynamic therapy and sodium hypochlorite.
Normal saline does not exhibit antibacterial properties or tissue-dissolving potential [24, 25]. Consequently, residual pulp tissue, microbial byproducts, and necrotic debris within the canal system and dentinal tubules are more likely to persist. These retained substrates can undergo oxidative degradation, bacterial metabolism, and proteolytic breakdown, producing chromogenic compounds that may diffuse through the tooth structure, leading to intrinsic discoloration [24]. Therefore, although normal saline does not chemically stain tooth structure, its limited cleansing efficacy may allow the retention of chromogenic substrates that cause tooth discoloration over time.
Sodium hypochlorite is a widely used root canal irrigant known for its tooth bleaching capacity via hypochlorite ions (OCl-) [26]. Similar to the findings of this study, sodium hypochlorite can cause tooth discoloration due to its potential to crystallize on the root dentin and penetrate up to 300 μm, which complicates its removal from the root canal [4, 27-29]. However, since its effect on tooth discoloration was less pronounced than that induced by normal saline, it may not be considered a significant discoloring agent [30]. The tissue-dissolving effect and antimicrobial activity of sodium hypochlorite are directly proportional to its concentration, which inversely affects its biological compatibility and can lead to periapical tissue inflammation [31]. Additionally, it is ineffective at removing the smear layer and has limited extended antimicrobial activity; thus, the adjunctive use of other irrigation regimens is proposed, as effective removal of hard tissue debris is crucial for successful endodontics [4, 32].
Silver diamine fluoride (SDF) is an anti-cariogenic substance used as a safe and effective caries-arresting material. Due to its antibacterial properties, it has also been suggested as an intracanal medicament and root canal irrigant [33]. However, it can cause unwanted tooth discoloration due to the precipitation of silver oxides and sulfide deposits in the dentinal tubules [34]. Patel et al. found that black staining began within two minutes of SDF application, reaching its maximum effect after 12 hours [35].
In this study, SDF irrigant caused the most pronounced tooth discoloration from the first day post-irrigation, indicating its potential for early tooth darkening due to silver deposits at the orifices of dentinal tubules. The discoloration intensified over time as silver ions interacted with sulfides in the dentin's organic structure. Reports indicated that the darkening effect of SDF becomes clinically detectable by the seventh day and beyond [36]. These findings strongly caution against using SDF in areas of esthetic importance.
Nanomaterials consist of small particles (1-100 nm) that enhance antimicrobial efficiency due to their increased surface-to-volume ratio and high chemical reactivity. Silver nanoparticles can disrupt bacterial cell function and penetrate both gram-negative and gram-positive bacteria [37]. In endodontics, silver nanoparticles are used as root canal irrigants, incorporated into sealers, serve as vehicles for calcium hydroxide, and applied as antibacterial coatings for gutta-percha [38].
While nano-silver particle-containing irrigants showed minimal color change immediately after treatment, their effect was less stable over time. The discoloration likely results from the oxidative aggregation of nanoparticles and surface interactions with protein complexes, followed by gradual penetration into dentinal tubules. Over time, the cumulative deposition and oxidation of silver nanoparticles led to further discoloration [30]. In contrast, combining silver nanoparticles with calcium hydroxide as intracanal medication did not cause significant tooth color changes compared to calcium hydroxide alone [20].
Photodynamic therapy (PDT), originally developed for cancer treatment, has shown promise for root canal disinfection. It involves four components: a LASER or light source, a light delivery method, such as fiber optics, a photosensitizer (PS), and oxygen. The PS absorbs light and generates active oxygen radicals that destroy microorganisms [13]. Methylene blue (MB), a hydrophilic PS, effectively inactivates both Gram-positive and Gram-negative bacteria [39].
The results of PDT in this study align with those of Costa et al., who reported that photodynamic therapy may cause changes in tooth color [40]. The influence of MB on the color stability of intra-radicular dentin has been shown to result in alterations that exceed the clinically noticeable threshold [41]. Methylene blue is a cationic dye that penetrates dentinal tubules and porous structures of dental tissue, exhibiting strong chromogenic properties [19].
Given the anticipated discoloration associated with PDT using MB, a combination of PDT and NaOCl was employed in this study to mitigate this effect. Our findings indicated clinically undetectable color changes after the first day of assessment. Carvalho et al. studied the effectiveness of various chemicals in eliminating MB dye after PDT and concluded that 2.5% NaOCl was the most effective. However, complete removal of MB dye from the root canal system could not be achieved due to its affinity for dentinal tubules and organic matrix components [42]. The MB photosensitizer is used in low concentrations, undergoes photobleaching during activation, and can be flushed away with sodium hypochlorite irrigant. Furthermore, it does not leave behind persistent pigment-forming compounds in dentin, unlike silver-based irrigants.
Although the combination of PDT and NaOCl reduced the discoloration effect of methylene blue (MB), pairing NaOCl with either silver diamine fluoride (SDF) or silver nanoparticles did not mitigate their tooth-staining effects. While SDF with NaOCl is noted for its strong antimicrobial efficacy, some staining persisted. This is attributed to the resistance of silver sulfide and silver-protein complexes to chelation and removal by NaOCl irrigation. Similarly, the combination of nano-silver and NaOCl resulted in noticeable discoloration, due to the deep penetration and agglomeration of nanoparticles, as well as their oxidative interactions with organic structures that resist sodium hypochlorite's action [43].
The strengths of this study lie in the use of an objective color measurement tool, which adds precision to the evaluation of tooth discoloration. The findings emphasize the importance of selecting irrigants for both their antimicrobial efficacy and their impact on the long-term aesthetics of treated teeth.
5. LIMITATIONS
The limitations of this study stem from its in vitro nature, as it does not account for the complex intraoral factors, such as temperature fluctuations, saliva, and dietary habits, that influence tooth discoloration over time. Additionally, the limited observation period may not capture long-term outcomes, and the use of only one type of tooth may restrict generalization to other types with varying morphology and enamel thickness. Therefore, further research is warranted to explore strategies for mitigating the discoloration effects of irrigants while preserving their therapeutic benefits, monitored over an extended period, and including different types of teeth to assess color changes across various surfaces.
CONCLUSION
Within the limitations of this in vitro study, it can be concluded that silver nanoparticles, whether used alone or in combination with sodium hypochlorite, were associated with a noticeable early tooth darkening, particularly evident from the seventh day onward. The combination of photodynamic therapy and sodium hypochlorite resulted in only initial, clinically imperceptible color changes, with no significant progression over time. Silver diamine fluoride produced the highest level of discoloration among all regimens tested, indicating its strong staining potential.
AUTHORS’ CONTRIBUTIONS
The authors confirm contribution to the paper as follows: A.A. and L.K.: Study conception and design; M.F. and K.A.: Data collection; A.A. and M.F.: Analysis and interpretation of results; A.A. and L.K.: Draft manuscript. All authors reviewed the results and approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| AgNP | = Silver nanoparticles |
| ANOVA | = Analysis of Variance |
| CIE | = Commission Internationale de l’Eclairage system |
| MB | = Methylene blue |
| NaOCl | = Sodium hypochlorite |
| OCl- | = Hypochlorite ions |
| PDT | = Photodynamic therapy |
| SDF | = Silver diamine fluoride |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Ethical clarification to conduct this research was approved by the Biomedical Research Ethics Committee, Umm AlQura University, SA (HAPO-02-K-012-2023-01-1394).
HUMAN AND ANIMAL RIGHTS
All procedures performed in studies involving human participants were in accordance with the ethical standards of institutional and/or research committee and with the 1975 Declaration of Helsinki, as revised in 2013.
AVAILABILITY OF DATA AND MATERIALS
The data and supportive information are available within the article.
ACKNOWLEDGEMENTS
Declared none.


