All published articles of this journal are available on ScienceDirect.
Comparative In-Vitro Evaluation of Probiotic Mouthwash Formulations with Cannabis Extracts and Propolis on TNF-α Reduction
Abstract
Introduction
Tumor necrosis factor-alpha (TNF-α) is a pro-inflammatory cytokine involved in the wound healing process. Elevated TNF-α levels contribute to chronic inflammation and impaired healing. Probiotics have demonstrated potential in reducing TNF-α, and both cannabis extract (CBD) and propolis possess anti-inflammatory properties. This study aimed to evaluate the effects of probiotic mouthwash formulations containing CBD, propolis, or both, on TNF-α secretion in vitro, with potential applications in managing oral inflammation.
Methods
Four probiotic mouthwash formulations were prepared: (1) probiotics with 1% propolis, (2) probiotics with 5% propolis, (3) probiotics with 1% CBD and 1% propolis, and (4) probiotics with 1% CBD and 5% propolis. Additional controls included propolis extracts at 1% and 5%, as well as a previously studied optimal formulation containing 10% probiotics and 1% CBD. TNF-α production was induced in LPS-stimulated THP-1 cells, and secretion levels were quantified using sandwich ELISA.
Results
All tested formulations significantly inhibited TNF-α secretion (>90%), with the highest inhibition (92.96%) observed in the formulation containing probiotics and 5% propolis. In contrast, the 10% probiotic + 1% CBD formulation and 1% CBD extract alone showed lower inhibition rates of 66.54% and 13.25%, respectively. Statistical analysis confirmed the superior efficacy of propolis-containing formulations (p < 0.05). No cytotoxic effects were observed.
Discussion
The findings confirmed that propolis enhanced the anti-inflammatory efficacy of probiotic mouthwashes, likely through synergistic mechanisms involving flavonoids and polyphenols that suppress TNF-α by inhibiting the NF-κB pathway. In contrast, adding CBD did not yield additional benefit when combined with propolis. A concentration-dependent effect was observed, as 5% propolis yielded greater inhibition than 1%. Despite assay limitations and in vitro constraints, these results support the potential of probiotic–propolis mouthwashes as adjunctive therapies for inflammatory oral conditions.
Conclusion
Probiotic mouthwash formulations containing propolis, either alone or in combination with CBD, significantly suppressed TNF-α secretion more effectively than probiotic or CBD formulations alone. The formulation with 5% propolis demonstrated a significant anti-inflammatory effect, suggesting a promising role for propolis in oral healthcare applications.
1. INTRODUCTION
In oral surgery treatment, one of the primary concerns is achieving proper healing following the procedure. Wound healing is a complex process that occurs in a sequential yet overlapping manner, encompassing four distinct phases: hemostasis, inflammation, proliferation, and remodeling, each requiring an appropriate duration for optimal recovery [1-3]. Disruptions in any of these phases, whether abnormal progression, absence, or prolongation, can lead to delayed healing or the development of chronic wounds, most commonly associated with a prolonged inflammatory phase [1-3]. Tumor necrosis factor-alpha (TNF-α), a key pro-inflammatory cytokine released during the inflammatory phase, plays a pivotal role in the wound healing process. However, elevated levels of TNF-α have been linked to impaired healing and chronic wound formation. Consequently, therapeutic strategies aimed at managing abnormal wound inflammation often target the reduction of TNF-α levels [1]. Recently, probiotics have garnered attention for their potential therapeutic benefits, particularly their anti-inflammatory properties, which include the suppression of various pro-inflammatory mediators [4-6].
A previous study by Ladda et al. [7] demonstrated that the supernatant derived from probiotics, Lactobacillus paracasei -MSMC39, exhibited strain-specific activity in reducing the pro-inflammatory cytokine tumor necrosis factor-alpha (TNF-α). Building on this, Banjonjit et al. [8] found that Lactobacillus paracasei MSMC39-1 significantly reduced TNF-α levels in wounds following third molar (wisdom tooth) extractions. In their study, the probiotic supernatant was used as a wound rinse for post-operative care, and results showed that the TNF-α levels in the gingival crevicular fluid of the experimental group were significantly lower than those in the control group, indicating a higher percentage of TNF-α inhibition. These findings led to the development of a mouthwash formulation for more practical applications. Subsequently, a study by Nisapar et al. [9] developed a probiotic mouthwash formulation by incorporating cannabis extract to further enhance its anti-inflammatory properties. They found that the mouthwash formula containing 10% probiotics with 1% cannabis extract was the most effective, not only enhancing TNF-α inhibition compared to the probiotic alone, but also confirmed to be non-toxic to cells.
Cannabis contains a primary active compound called Cannabidiol (CBD). Numerous studies have confirmed that cannabis possesses various properties, including anti-inflammatory, antioxidant, and antimicrobial effects [10, 11]. In dentistry, research has explored the use of cannabis in treatments as well, given its pain-relieving, anti-inflammatory, and antibacterial properties. It has been used to treat pain from teeth and gums, prevent tooth decay, and reduce gum inflammation, serving as an antimicrobial and antiseptic to promote patients' oral health [12]. In addition to cannabis, several other extracts with anti-inflammatory properties can promote wound healing. Some extracts may enhance the effects of probiotics or potentially be more effective than cannabis or even work synergistically with both cannabis and probiotics to amplify their effects. Propolis has been found to possess a wide range of therapeutic properties, including antibacterial, antifungal, antiviral, anticancer, antioxidant, and anti-inflammatory activities [13-15]. Numerous studies have investigated the use of propolis in dental treatments, demonstrating its ability to promote healing within the oral cavity [16, 17]. Propolis has also shown benefits in the management of periodontal diseases. Furthermore, mouthwashes containing propolis have been reported to effectively reduce the incidence and severity of oral mucositis in patients undergoing radiation therapy and chemotherapy [17-19].
This study aims to compare the effectiveness of a probiotic mouthwash containing cannabis extract, a probiotic mouthwash containing propolis, and a formulation combining both cannabis extract and propolis. The objective is to evaluate and compare the anti-inflammatory effects of these formulations, specifically their ability to inhibit TNF-α secretion, relative to a mouthwash containing 10% probiotics alone and the optimal formulation from a previous study (10% probiotics with 1% CBD) [9]. Building on our preliminary findings reported in a conference proceeding, which first demonstrated that a probiotic mouthwash containing cannabis extract and propolis could significantly reduce TNF-α levels in vitro [20], we hypothesized that: (1) the probiotic mouthwash formulation combined with propolis extract would reduce TNF-α levels more effectively than the formulation combined with cannabis extract; and (2) the probiotic mouthwash formulation containing both cannabis extract and propolis extract would be more effective in reducing TNF-α levels than formulations containing either cannabis extract or propolis extract alone. If the formulations developed in this research prove effective, they may offer significant benefits in surgical fields and could also be applicable in the treatment of oral diseases associated with tissue inflammation.
2. MATERIALS AND METHODS
2.1. Study Design and Type of Study
This in vitro study was conducted using the human monocytic cell line THP-1, which was stimulated with lipopolysaccharide (LPS) to induce the production of TNF-α. Experimental and control treatments were applied to evaluate their effects on TNF-α inhibition. The levels of TNF-α were quantified using the Sandwich ELISA technique. The methodology is described as follows:
2.1.1. Probiotic Cell-free Culture Supernatant Preparation
Lactobacillus paracasei MSMC39-1 was cultured in liquid medium under anaerobic conditions at 37°C for 48 hours, then diluted in liquid food to achieve a concentration of 10^8 cells/milliliter. It was then incubated under the same conditions for another 48 hours. After that, the culture supernatant was separated by filtering it through a sterile 0.22 μm filter and stored at -20°C until needed.
2.1.2. Preparation for Test Substances
2.1.2.1. Probiotic Supernatant
The probiotic supernatant was thawed at room temperature and diluted with 0.9% saline to achieve a concentration of 10% v/v.
2.1.2.2. Propolis Solutions
Propolis extract powder containing 30% flavonoids was dissolved in saline solution to prepare final concentrations of 1% and 5% w/v.
2.1.2.3. Cannabidiol (CBD) Solutions
The solution was prepared following the method described by Nisapa et al. [9], in which cannabis oil containing 5% CBD was diluted in saline to achieve a final concentration of 1% v/v.
2.1.3. Preparation of Individual Mouthwash Formulations
The substances were combined to prepare four mouthwash formulations, as follows:
Formula 1: 10% Probiotic supernatant mixed with 1% propolis.
Formula 2: 10% Probiotic supernatant mixed with 5% propolis.
Formula 3: 10% Probiotic supernatant mixed with 1% CBD and 1% propolis.
Formula 4: 10% Probiotic supernatant mixed with 1% CBD and 5% propolis.
All prepared substances were divided into experimental and control groups for testing with the human monocytic cell line THP-1, as follows:
2.1.4. Preparation of Human Monocytic Cell Line THP-1
The human monocytic cell (THP-1 monocytic cell line [ATCC TIB-202]) was cultured using RPMI 1640 supplemented with 10% heat-inactivated fetal bovine serum and incubated at 37°C with 5% CO2.
2.1.5. Cytotoxic Testing
The cytotoxicity of all variable substances was tested using the MTT assay. The tests were conducted in a 96-well plate with 50,000 THP-1 human monocytic cells per well in 100 μl of culture medium. Then, 100 μl of each mouthwash formulation was added, including positive and negative control groups, with three wells for each formulation. They were incubated at 37°C with 5% CO2 for 3 hours and 30 minutes, then centrifuged at 4,000 rpm for 10 minutes. The supernatant was removed, and 100 μl of MTT solution (0.5 mg/ml) was added to each well, followed by incubation at 37 °C with 5% CO2 for 2–4 hours. After incubation, the plates were centrifuged at 4,000 rpm for 10 minutes. The supernatant was then discarded, and 100 μL of DMSO was added to each well. The absorbance was then measured at 595 nm, and the resulting values were analyzed to determine cell viability for each treatment.
2.2. Measurement of TNF-α inhibition with ELISA
The ability of each mouthwash formulation to inhibit TNF-α secretion was evaluated by first stimulating TNF-α production with 10 μl of purified lipopolysaccharide (LPS) from Escherichia coli O127:B8, achieving a final concentration of 100 ng/ml. Each mouthwash formulation, along with positive and negative control groups, was then added to the wells, with three replicate wells per formulation. The plates were incubated at 37 °C with 5% CO2 for 3 hours and 30 minutes. Following incubation, the supernatant was collected by centrifugation at 3,000 rpm for 5 minutes, and TNF-α levels were measured using the Sandwich ELISA method (R&D Systems, USA). The obtained values were used to generate a standard curve, which allowed calculation of the sample concentrations and determination of the percentage of TNF-α secretion inhibition using the following formula:
TNF-α inhibition percentage = 100 x (1-amount of TNF-α from the supernatant containing each mouthwash formulation (pg/ml) ÷ amount of TNF-α from the supernatant obtained from the negative control group (pg/ml)).
The experiment was performed in triplicate using the same methodology and control conditions to ensure reproducibility.
2.3. Statistical Analysis
Data were analyzed using GraphPad Prism software, version 9.3.1 (GraphPad Software, San Diego, CA, USA). The Kruskal-Wallis test was employed to assess differences among experimental groups at a 95% confidence level. When statistically significant differences were identified, post-hoc comparisons were made using Dunn’s multiple comparisons test to identify pairwise differences between groups.
3. RESULTS
3.1. Cell Toxicity Test Results for the Mouthwash Formula containing the Probiotic Lactobacillus paracasei MSMC39-1, Propolis Extract, and Cannabis Extract Using the MTT Assay
From the test of cell viability, the results showed that the four mouthwash formulas had the following cell survival percentages: Formula 1 (10% probiotic solution, 1% propolis) at 94.74%, Formula 2 (10% probiotic solution, 5% propolis) at 103.36%, Formula 3 (10% probiotic solution, 1% cannabis extract, and 1% propolis) at 168.05%, and Formula 4 (10% probiotic solution, 1% cannabis extract, and 5% propolis) at 212.63%. Meanwhile, the mouthwash formula from previous studies (10% probiotic solution and 1% cannabis extract) yielded a survival percentage of 196.17%. The propolis extracts at 1% and 5% resulted in survival percentages of 127.58% and 124.02%, respectively. Additionally, other tested substances, including 1% cannabis extract, 10% probiotic solution, and 0.9% saline, had cell survival percentages of 240.23%, 101.97%, and 91.07%, respectively, when compared to the cells in the cell culture medium (Fig. 1).
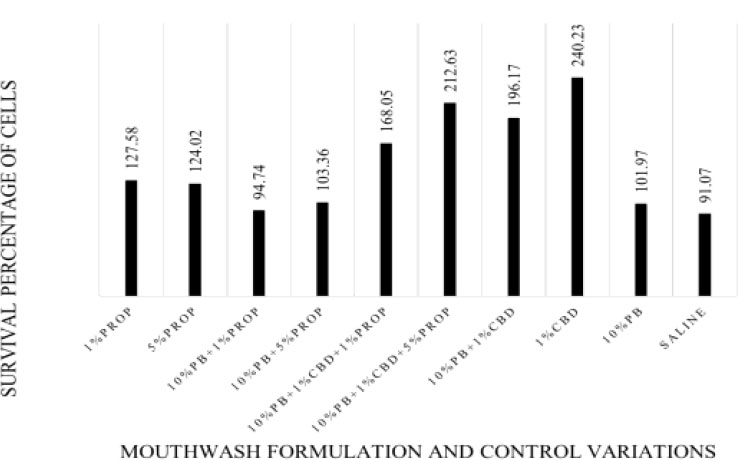
Cell survival percentage after exposure to various mouthwash formulations and control treatments for 3 hours and 30 minutes. The X-axis represents different mouthwash formulations and control conditions, while the Y-axis indicates the percentage of viable cells.
3.2. The Inhibition of TNF-α Release from the Mouthwash containing the Probiotic Lactobacillus paracasei MSMC39-1, Propolis Extract, and Cannabis Extract
In the experiment involving the addition of mouthwash formulations and various test substances to LPS-stimulated THP-1 cells for 3 hours and 30 minutes, the TNF-α levels were extremely low. When compared with the standard curve, some absorbance values fell below the lowest standard point, resulting in negative calculated concentrations that do not accurately represent TNF-α levels. As a result, the data are reported in terms of absorbance values rather than absolute concentration.
The absorbance values and percentage of TNF-α release inhibition of each mouthwash formulation compared to liquid cell culture medium are presented in Table 1.
A Kruskal–Wallis test at a 95% confidence level was conducted to determine if there were statistically significant differences in TNF-α levels, as measured by absorbance values, among the experimental and control groups (H = 80.72, df = 10, p < 0.000001). Post-hoc pairwise comparisons using Dunn’s multiple comparisons test demonstrated that both concentrations of propolis extract (1% and 5%), as well as all four mouthwash formulations, exhibited significantly lower TNF-α levels compared to the negative control (cell culture medium) and the positive control (10% probiotic solution). Furthermore, 5% propolis extract and mouthwash formula 2 (comprising 10% probiotic solution and 5% propolis) showed significantly lower TNF-α levels compared to the 1% cannabis extract group (Fig. 2). No statistically significant differences were observed among the remaining groups (Table 2).
| Tested Group | Ingredients | Absorbance Value | Percentage of TNF-α Release Inhibition compared to Liquid Cell Culture Medium |
|---|---|---|---|
| Formulation 1 | Probiotics 10%, Propolis 1% | 0.0856 | 92.05% |
| Formulation 2 | Probiotics 10%, Propolis 5% | 0.0758 | 92.96% |
| Formulation 3 | Probiotics 10%, Cannabis 1% and Propolis 1% | 0.0841 | 92.18% |
| Formulation 4 | Probiotics 10%, Cannabis 1% and Propolis 5% | 0.0823 | 92.35% |
| Pure extraction | Propolis 1% | 0.0835 | 92.24% |
| Pure extraction | Propolis 5% | 0.0783 | 92.73% |
| Optimal formulation from Nisapa et al. [9] | Probiotics 10%, Cannabis 1% | 0.3602 | 66.54% |
| Positive control | Cannabis 1% | 0.5337 | 50.42% |
| Positive control | Probiotics 10% | 0.9338 | 13.25% |
| Negative control | Liquid cell culture medium | 1.0765 | - |
| - | 1% Propolis | 5% Propolis | 10% Probiotics + 1% Propolis | 10% Probiotics + 5% Propolis | 10% Probiotics + 1% cbd + 1% Propolis | 10% Probiotics + 1% cbd + 5% Propolis |
|---|---|---|---|---|---|---|
| 1% propolis | - | >0.99 | >0.99 | >0.99 | >0.99 | >0.99 |
| 5% propolis | >0.99 | - | >0.99 | >0.99 | >0.99 | >0.99 |
| 10% probiotics + 1% propolis | >0.99 | >0.99 | - | >0.99 | >0.99 | >0.99 |
| 10% probiotics + 5% propolis | >0.99 | >0.99 | >0.99 | - | >0.99 | >0.99 |
| 10% probiotics + 1% cbd + 1% propolis | >0.99 | >0.99 | >0.99 | >0.99 | - | >0.99 |
| 10% probiotics + 1% cbd + 5% propolis | >0.99 | >0.99 | >0.99 | >0.99 | >0.99 | - |
| 10% probiotics + 1% cbd | 0.911 | 0.227 | >0.99 | 0.1197 | 0.852 | 0.617 |
| 10% probiotics | 0.000975*** | 0.000105*** | 0.001876*** | 0.000039*** | 0.000872*** | 0.000515*** |
| culture media | 0.000278*** | 0.000026*** | 0.000556*** | 0.000009*** | 0.000247*** | 0.000142*** |
| 1% cbd | 0.1 | 0.018174* | 0.1634 | 0.008382** | 0.092 | 0.062 |
| - | 1% Propolis | 5% Propolis | 10% Probiotics + 1% Propolis | 10% Probiotics + 5% Propolis | 10% Probiotics + 1% cbd + 1% Propolis | 10% Probiotics + 1% cbd + 5% Propolis |
|---|---|---|---|---|---|---|
| 1% propolis | - | >0.99 | >0.99 | >0.99 | >0.99 | >0.99 |
| 5% propolis | >0.99 | - | >0.99 | >0.99 | >0.99 | >0.99 |
| 10% probiotics + 1% propolis | >0.99 | >0.99 | - | >0.99 | >0.99 | >0.99 |
| 10% probiotics + 5% propolis | >0.99 | >0.99 | >0.99 | - | >0.99 | >0.99 |
| 10% probiotics + 1% cbd + 1% propolis | >0.99 | >0.99 | >0.99 | >0.99 | - | >0.99 |
| 10% probiotics + 1% cbd + 5% propolis | >0.99 | >0.99 | >0.99 | >0.99 | >0.99 | - |
| 10% probiotics | 0.0013** | <0.0001*** | 0.0027** | <0.0001*** | 0.0014** | 0.0008*** |
| 1% cbd | 0.0353* | 0.0020** | 0.063 | 0.0005*** | 0.0366* | 0.0241* |
| 10% probiotics + 1% cbd | 0.2690 | 0.0233* | 0.435 | 0.0071** | 0.277 | 0.196 |
Analysis of the percentage of TNF-α inhibition, calculated relative to the negative control, revealed statistically significant differences among groups (H = 57.85, df = 8, p < 0.0001). Post hoc analysis using Dunn’s multiple comparisons test indicated that both 1% and 5% propolis extracts, as well as all four mouthwash formulations, demonstrated significantly higher TNF-α inhibition compared to the 10% probiotic solution alone. Moreover, 1% and 5% propolis extracts, along with mouthwash formulas 2, 3, and 4, exhibited significantly greater inhibition than the 1% cannabis extract group. When compared to the mouthwash formulation from the previous study (probiotic combined with 1% cannabis extract), both 5% propolis extract and mouthwash formula 2 (10% probiotic solution with 5% propolis) showed significantly enhanced TNF-α inhibition (Fig. 3). No statistically significant differences were observed among the remaining groups (Table 3).
4. DISCUSSION
This study aimed to compare the effectiveness of probiotic mouthwash formulations containing cannabis and propolis in reducing TNF-α secretion from the human monocytic cell line THP-1. Previous research by Ladda et al. [7] demonstrated that the supernatant of Lactobacillus paracasei MSMC39-1 significantly inhibited TNF-α secretion in THP-1 cells. Building on this finding, Banjonjit et al. [8] and Nisapa et al. [9] further investigated its applications in dentistry, reporting that the probiotic supernatant reduced post-surgical inflammation [8]. Notably, a formulation containing 10% probiotic and 1% cannabis extract yielded the highest level of TNF-α inhibition [9]. In the present study, propolis extract was incorporated into the mouthwash formulations, and the anti-inflammatory properties were subsequently tested and compared.
The cytotoxicity of each mouthwash formulation was evaluated prior to testing anti-inflammatory effects to ensure that observed reductions in TNF-α levels were due to inhibition of cytokine secretion rather than a consequence of reduced cell viability or cell death [21]. Cytotoxicity was assessed using the MTT assay, a widely accepted method for determining cell viability, in accordance with ISO 10993-5 guidelines for biological evaluation of medical devices. According to MTT guidelines, a test substance is considered cytotoxic if cell viability falls below 70% [22]. The results indicated that all tested mouthwash formulations, including those containing propolis extract and the positive control, demonstrated cell viability above 70%. These findings confirmed that none of the tested formulations exhibited cytotoxic effects under the experimental conditions.
In the evaluation of TNF-α secretion inhibition by mouthwash formulations, results were reported in terms of absorbance values rather than absolute TNF-α concentrations. This approach was taken because, although the standard curves generated for each experiment were accurate, the detection threshold was suboptimal, possibly due to the use of excessively high standard concentrations, making it impossible to reliably calculate TNF-α concentrations in absolute terms (e.g., picograms per milliliter). Nonetheless, as the absorbance values are directly proportional to TNF-α concentrations, it can be reasonably inferred that higher absorbance readings reflect higher levels of TNF-α.
The results demonstrated that the probiotic mouthwash containing 10% supernatant from Lactobacillus paracasei MSMC39-1, used as the positive control, exhibited notable anti-inflammatory activity by inhibiting TNF-α secretion. This effect is attributed to the presence of immunomodulatory proteins, referred to as immunomodulin, with molecular weights below 30 kDa, found in the probiotic supernatant. Supporting this mechanism, prior studies have reported that treatment with proteinase K significantly reduces the anti-inflammatory effect of the supernatant, indicating that these immunomodulatory proteins are susceptible to enzymatic degradation [7].
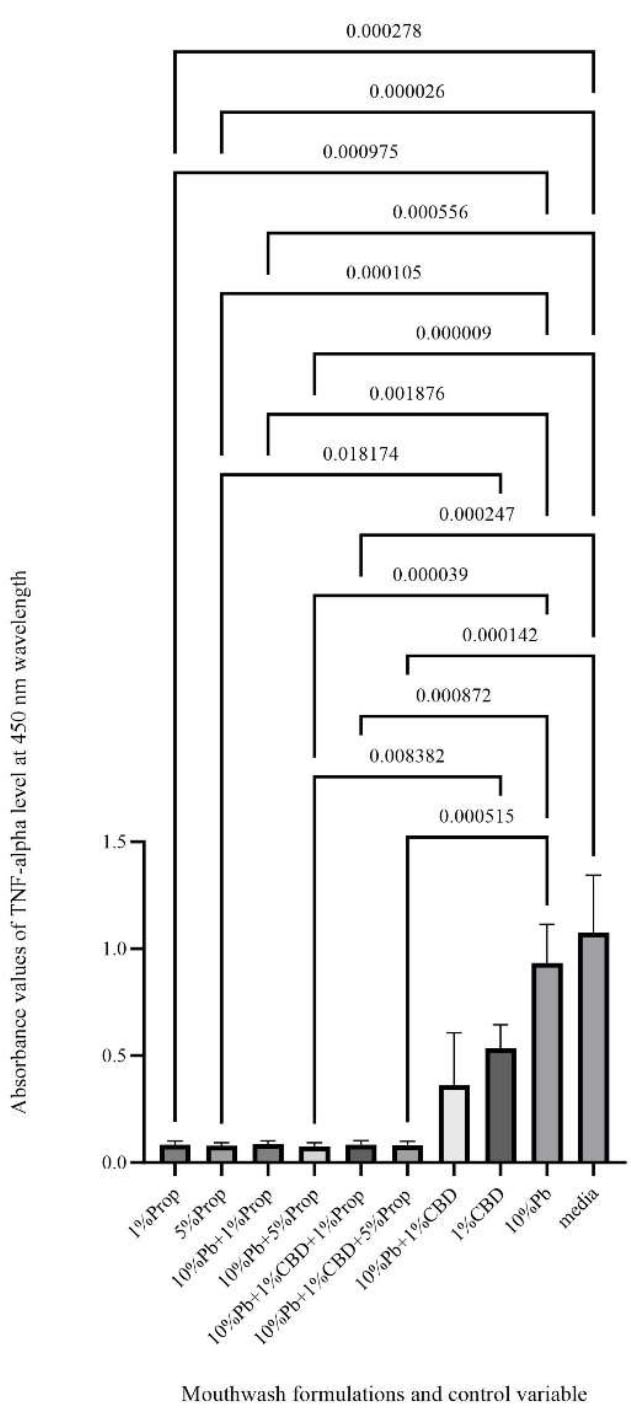
Absorbance values at 450 nm, representing TNF-α levels in response to various mouthwash formulations, are compared across all groups, including negative and positive controls. The X-axis represents the different mouthwash formulations and control conditions, while the Y-axis shows the absorbance values measured at 450 nm. p-values are presented only for pairwise comparisons that reached statistical significance (p < 0.05).

Percentage inhibition of TNF-α secretion by various mouthwash formulations. Percent inhibition values were calculated relative to the liquid cell culture medium (negative control), and statistical comparisons were made across all groups, including positive controls. The X-axis represents different mouthwash formulations and control conditions, while the Y-axis shows the percentage inhibition of TNF-α secretion. p-values are shown only for comparisons that reached statistical significance (p < 0.05).
In the study by Nisapa et al. [9], the most effective formulation for inhibiting TNF-α secretion was identified as a probiotic mouthwash combined with 1% cannabis extract. This finding suggests that cannabis not only enhances the anti-inflammatory effects of probiotics but also possesses intrinsic anti-inflammatory properties. Therefore, in the present study, the same concentration of cannabis extract (1%) was used as a positive control. In addition, to investigate the potential synergistic or additive effects of propolis, various concentrations of propolis extract were incorporated into newly developed formulations. The previously identified optimal formulation, as described by Nisapa et al., was included as part of the experimental groups for comparison with the new formulations developed in this study.
In this study, the inhibition of TNF-α secretion observed in the mouthwash formulation containing probiotics and 1% cannabis extract was consistent with the findings of a previous study [9]. Both investigations demonstrated that the addition of cannabis extract enhanced the anti-inflammatory efficacy of the probiotic mouthwash by further suppressing TNF-α secretion. The anti-inflammatory properties of cannabis are supported by findings from a study by Gugliandolo et al. [23], who reported that cannabidiol (CBD), a major component of cannabis, reduces inflammatory responses by modulating NF-κB activity, a key transcriptional regulator involved in the expression of pro-inflammatory cytokines, including TNF-α. Furthermore, Wang et al. [24] demonstrated that CBD exhibits an anti-inflammatory mechanism comparable to that of dexamethasone, acting through modulation of the MAPK pathway via JNK and ERK signaling, thereby suppressing NF-κB activation and reducing TNF-α secretion.
The results of this study also demonstrated that the addition of propolis to mouthwash formulations resulted in TNF-α secretion inhibition rates exceeding 90% across all tested formulations. In comparison, the probiotic supernatant alone and the mouthwash containing the probiotic supernatant combined with 1% cannabis extract showed inhibition rates of only 13.25% and 66.54%, respectively.
This finding is consistent with the study by Furukawa et al. [25], which demonstrated that propolis reduces levels of pro-inflammatory cytokines, such as TNF-α and IL-1β, while promoting wound healing by upregulating the expression of keratin 1 and keratin 5. Furthermore, Jajali et al. [26] reported that propolis exerts immunomodulatory effects by influencing components of the innate immune system and downregulating the expression of several inflammatory genes, including key mediators, such as TNF-α and IL-6. Supporting these findings, a systematic review by Zulhendri et al. [27] concluded that propolis significantly suppresses inflammatory mediators. One of the primary active compounds identified was caffeic acid phenethyl ester (CAPE), which functions by inhibiting the NF-κB signaling pathway. This effect is also attributed to the antioxidant properties of flavonoids and polyphenols present in propolis.
However, as the inhibition percentage of the propolis-containing formulations exceeded 90%, this value was calculated by comparing the TNF-α levels in each formulation to those of the negative control. However, it is important to consider the natural variability inherent in TNF-α assays, which can be influenced by multiple factors, including intra- and inter-assay variation, individual biological variability, and methodological differences between studies [28]. Notably, variations in ELISA kits or assay protocols can yield different absolute TNF-α concentrations for the same sample [29]. The inter-assay coefficient of variation for TNF-α has been reported to be as high as 7.8% [28]. Despite this, the data clearly demonstrated that mouthwash formulations containing propolis consistently exhibited higher TNF-α inhibition compared to formulations without propolis, suggesting greater efficacy in reducing TNF-α secretion.
Furthermore, our findings revealed that both the propolis extract at 5% concentration and the mouthwash formulations containing 5% propolis demonstrated higher TNF-α inhibition compared to those with 1% concentration. This suggests a concentration-dependent enhancement in the anti-inflammatory activity of propolis. These results are consistent with the study by Sahlan et al. [30], which reported that the anti-inflammatory effects of propolis vary depending on its concentration. At lower concentrations, propolis may stimulate TNF-α secretion; however, as the concentration increases, its inhibitory effect on TNF-α secretion becomes more pronounced.
Despite these findings, the study has certain limitations. One limitation is that the ELISA-based TNF-α concentrations were reported in terms of absorbance values due to issues with the detection threshold. We recommend that future studies adjust the dilution range of standards and samples to improve quantification accuracy, ensure appropriate assay sensitivity, and enhance the interpretability and comparability of the data.
This study also has several other limitations that should be considered when interpreting the results. First, the experiments were conducted in vitro, which does not fully replicate the complex environment of the human oral cavity. As such, the biological responses observed in cell culture may not directly translate to clinical outcomes. Second, no formal sample size calculation was performed, as this is a preliminary in vitro study; although triplicate measurements and repeated experiments were conducted, the statistical power remains limited. Third, the stability and shelf-life of the tested formulations were not evaluated, which may impact their real-world applicability and long-term effectiveness. Additionally, the study did not assess long-term cytotoxicity or effects on oral microbiota, which are important for comprehensive safety and efficacy assessments. Finally, the specific mechanisms of TNF-α inhibition by the combined ingredients (probiotic, propolis, and cannabis extract) were not explored in detail and require further molecular investigation.
Above all, it is important to note that TNF-α is not the sole parameter to consider in the context of oral wound healing. Numerous other cytokines also play crucial roles in the inflammatory and wound healing processes through various signaling pathways. Therefore, future studies should explore additional inflammatory markers and molecular mechanisms to provide a more comprehensive understanding of the therapeutic potential of these formulations.
CONCLUSION
Probiotic mouthwashes containing propolis, as well as those formulated with both cannabis extract and propolis, demonstrated statistically significant inhibition of TNF-α secretion compared to the probiotic culture alone, with a confidence level of 95%. In addition, these formulations exhibited a higher percentage of TNF-α inhibition than the probiotic mouthwash containing 1% cannabis extract, highlighting the enhanced anti-inflammatory potential when propolis was incorporated into the formulation.
Among all tested formulations, mouthwash formula 2, comprising 10% probiotic culture and 5% propolis, emerged as the most effective, with a TNF-α inhibition rate of 92.96%. This value was significantly higher than that observed for the probiotic mouthwash containing 1% cannabis extract, reinforcing the potent synergistic effect of probiotics and propolis in suppressing inflammatory mediator secretion.
These results suggest that combining probiotics with propolis may provide a superior strategy for developing anti-inflammatory mouthwash formulations with potential clinical applications, particularly in the management of oral inflammatory conditions and post-surgical wound healing. Given the promising outcomes, further research should be conducted to validate these findings in more physiologically relevant models, including animal studies or clinical trials.
Moreover, further investigations into the stability, storage conditions, and shelf life of these formulations are warranted to ensure the long-term maintenance of their biological activity and therapeutic efficacy. Stability studies will be particularly important to determine whether the active components remain effective over time under various environmental conditions, which is critical for product development and potential commercialization.
Furthermore, exploring the molecular mechanisms underlying the synergistic effects observed could provide valuable insights into how probiotics and propolis interact to modulate inflammatory pathways. Investigating additional inflammatory markers beyond TNF-α would also offer a more comprehensive understanding of the therapeutic potential of these formulations.
In summary, the incorporation of propolis into probiotic mouthwashes represents a promising approach to enhance anti-inflammatory efficacy. This study lays important groundwork for future development of effective oral care products aimed at improving oral health and supporting wound healing processes.
AUTHORS’ CONTRIBUTIONS
The authors confirm their contribution to the paper as follows: M.T.: Study conception and design; S.R.: Conceptualization; N.S.: Data collection. All authors reviewed the results and approved the final version of the manuscript.
ABBREVIATION
| TNF-Α | = Tumor Necrosis Factor-Alpha |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was approved by the Ethics Committee for the Review of Human Research Projects at Srinakharinwirot University, Thailand (Approval number: SWUEC/X-100/2565). This study did not involve human or animal subjects; only commercially available cell lines were used.
HUMAN AND ANIMAL RIGHTS
All procedures performed in studies involving human participants were in accordance with the ethical standards of institutional and/or research committee and with the 1975 Declaration of Helsinki, as revised in 2013.
AVAILABILITY OF DATA AND MATERIALS
All data generated or analyzed during this study are included in this published article.
FUNDING
This research project was financially supported by the Faculty Research Grant Budget for 2021, Faculty of Dentistry (Grant number 290/2564), and the Center of Excellence in Probiotics, Strategic Wisdom and Research Institute, Srinakharinwirot University.
ACKNOWLEDGEMENTS
Declared none.
DISCLOSURE
Part of this article has previously been published in the thesis, titled “The Effectiveness Comparison of Probiotic Mouthwash Formulation with Cannabis Extracts and Propolis on the Reduction of Tumor Necrosis Factor-Alpha,” available at: https://swu-ir.prosys.plus/items/ f8df5633-ca84-4d0d-ac88-44b737827b9e.


