All published articles of this journal are available on ScienceDirect.
Mineralized Plasmatic Matrix for Horizontal Ridge Augmentation in Anterior Maxilla with and without a Covering Collagen Membrane
Abstract
Background:
Mineralized Plasmatic Matrix [MPM] is a unique form of platelet rich fibrin that contains mineralized bone graft particles within a fibrin network.
Aim:
This study was conducted to evaluate horizontal ridge augmentation using MPM with and without a coverage membrane.
Materials and Methods:
Sixteen edentulous spaces were randomly divided into 2 equal groups. MPM was used for horizontal ridge augmentation with and without a covering collagen membrane (group 1 and 2, respectively). Cone Beam CT images were obtained preoperatively as well as 1 week and 4 months postoperatively to evaluate alveolar ridge and the resorption of the grafting material at 3 predetermined points along the site where the future dental implant will be placed.
Student’s t-test (Unpaired) was used for comparing two different groups with quantitative parametric data and student’s t-test (Paired) was used for comparing two related groups with quantitative parametric data while repeated measures ANOVA (Analysis of variance) followed by post-hoc Bonferroni was used for comparing more than two related groups with quantitative parametric data.
Results:
There was no statistically significant difference between the gained bone width in both groups. More but not statistically significant resorption was recorded in group 2.
Conclusion:
MPM can be successfully used for horizontal ridge augmentation without a barrier membrane.
1. INTRODUCTION
Resorption of the alveolar ridge is inevitable following tooth loss. This situation is particularly more pronounced in the anterior maxilla. Up to 33% of the alveolar ridge width and height are reported to extend within the first year after extraction. The resultant insufficient bone quantity and quality is reported to be associated with compromised esthetics, poor long-term prognosis and an increased failure rate of dental implants [1-4].
Despite the wide variety of grafting techniques reported in the literature, there is no consensus regarding which is the best method to be used. The decision is usually tailored by the surgeon, depending on the clinical situation [5-7]. Horizontal ridge augmentation often utilizes a block graft or a particulate bone graft with the barrier membrane [8].
Resorption of bone graft materials is a complicating factor and the stability of the graft is always questioned [5, 8-10]. The resorption rate as high as 46% was reported to occur following horizontal ridge augmentation [10]. Several modalities have been described to aid in graft stability and to ensure predictable bone formation. These include the use of different types of bone substitutes, barrier membranes, platelets rich plasma, bone morphogenic proteins, and Platelets Rich Fibrin (PRF) [6, 8, 11, 12].
The use of barrier membranes has always been an essential and key component of guided bone regeneration procedures [13]. Membranes are used to exclude the fast-growing epithelial and connective tissue cells from invading the graft material, giving time for the slowly growing bone cells to populate the created space and form osseous tissue [14, 15]. Resorbable barriers are more commonly used because there is no need for a second procedure for removal, as with the case of non-resorbable barriers [16].
Platelets rich fibrin has been used and researched extensively in the field of guided bone regeneration. PRF is proved to enhance both soft tissue and bone healing. PRF can be either incorporated into the bone substitute material or used as a membrane to cover bone grafts with or without a coverage collagen membrane [17-19].
Mineralized Plasmatic Matrix (MPM) is a platelet concentrate containing a bone substitute. The unique feature of MPM is that it incorporates platelets and growth factors that are described to offer the osteoinductive property and, at the same time, contains a graft material that will provide osteoconductivity, stability and volume preservation [20].
To the best of our knowledge, whether the physical form of MPM will protect the incorporated graft material from the invasion of the soft tissue cells was not addressed in the literature. This study was designed to investigate whether the use of MPM without a barrier membrane for horizontal ridge augmentation will provide adequate and successful augmentation and also to test if this could result in more graft resorption when compared to the situation in which a collagen membrane is used to cover the MPM.
2. MATERIALS AND METHODS
2.1. Patients Selection
Sixteen patients seeking restoration of a missing maxillary anterior tooth with implant-supported prosthesis were selected from the outpatient clinic of the Oral and Maxillofacial Surgery Department, Faculty of Dentistry, Mansoura University. Inclusion criteria included age of 18 years or older, a missing maxillary anterior tooth, insufficient bone width to accommodate placement of a dental implant, good oral hygiene, satisfactory occlusion, and patient ratification to comply with the follow-up protocol of this study. All patients were given a detailed description of the study protocol and only patients who agreed and signed a written consent form were recruited.
Exclusion criteria included smoking, poor oral hygiene, drug abuse, pregnancy or nursing, parafunctional habits, periodontitis and presence of any systemic disease that could affect the process of soft tissues or bone healing. Patients were randomly assigned to two equal groups.
2.2. Preoperative Evaluation
All patients had a thorough preoperative evaluation regarding the edentulous space and the alveolar ridge. Preoperative panoramic and periapical radiographs were taken to evaluate the neighboring teeth inclination and to rule out the presence of any osseous abnormality. Impression was also taken to fabricate study casts. The mesio-distal space was evaluated to ensure the ability to accommodate an esthetically acceptable future prosthesis. Occlusion was also evaluated to exclude patients with unfavorable occlusion or parafunctional habits. The exact buccolingual dimensions of the alveolar ridge were determined using Cone Beam Computerized Tomography [CBCT]. Patients who had sufficient bone width to accommodate dental implant placement were excluded. The width of the alveolar ridge of the edentulous space was measured at three points; 2mm below the alveolar crest, 6mm from the crest, and 12mm from the crest. Special attention was given to the quality of the oral hygiene of all patients. Only patients with acceptable hygiene were included. Professional hygiene visits were utilized when indicated.
2.3. Preparation of Mineralized Plasmatic Matrix
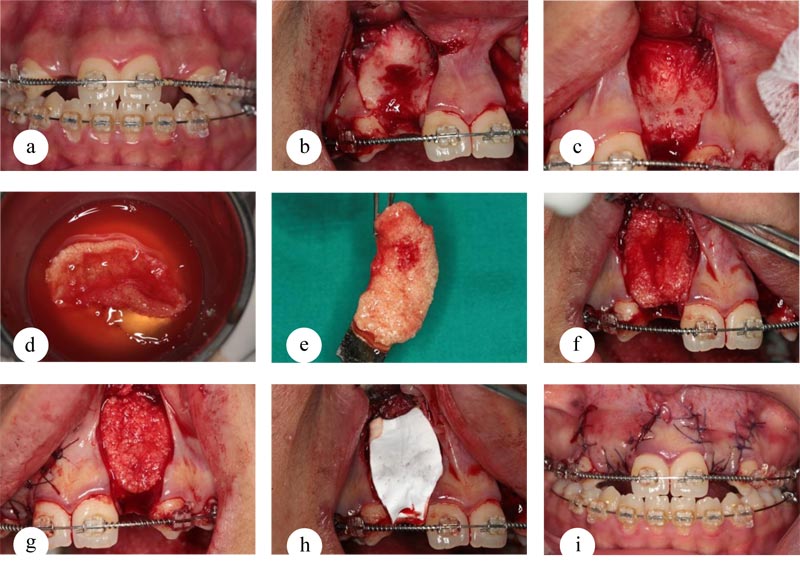
A Xenogenic cancellous bone chip was used in this study as a grafting material (TUTOBONE Microchips, RTI Biologics, TUTOGEN Medical GmbH, Germany). The graft was placed in a sterile mixing bowl and hydrated with saline. Venous blood was then collected from the patient into two 9mL non-coated, non-additive tubes (VACUETTE Tube 9ml, Z No Additive, white cap-black ring, Greiner Bio-One GmbH, Germany) and immediately centrifuged at 2500rpm for 12 minutes (EBA 20 Centrifuge, Andreas Hettich GmbH & Co, Germany). The centrifuge process separates a yellow plasma that is rich in platelets and fibrin concentrate on top of the red blood cell accumulated at the bottom of the tube. The tube cover was gently removed and then the plasma was collected with a syringe and placed over the bone graft material (BGM) and stirred immediately using a mucoperiosteal elevator until a bone-fibrin mass is formed (Fig. 1d and e).
2.4. Surgical Technique
The surgical procedures were performed by an experienced surgeon (Abdelfadil E). All patients were instructed to rinse their mouth with 0.12% chlorhexidine for 1 minute to decrease the intra-oral bacterial load. Anesthesia was obtained through the infiltration of Mepivacaine HCL 2% with levonordefrin 1:20.000 (Scandonest 2%, Septodont, France.). A full 3-lines mucoperiosteal flap was incised and reflected to expose the alveolar ridge. Periosteal releasing incisions were done at this point to ensure tension-free closure at the end of surgery. Small cortical perforations were done using a small round bur. The MPM was adapted to the labial surface of the alveolar bone, followed by flap repositioning and closure using 4-0 Vicryl sutures Assut Medical Sarl, Switzerland]. A collagen membrane (TUTOPATCH Collagen, RTI Biologics, TUTOGEN Medical GmbH, Germany) was used to cover the MPM in group 1, while in group 2, no barrier membranes were used (Fig. 1). A prophylactic antibiotic composed of amoxicillin/clavulanic (Augmentin 1gm, SmithKline Beecham, Egypt) was prescribed twice a day for all patients to prevent graft infection. A non-steroidal anti-inflammatory drug (Brufen 400mg, Abbot Pharmaceuticals, Egypt) was also prescribed for pain control. Patients were recalled one week postoperatively for wound evaluation and sutures removal.
2.5. Radiographic Evaluation
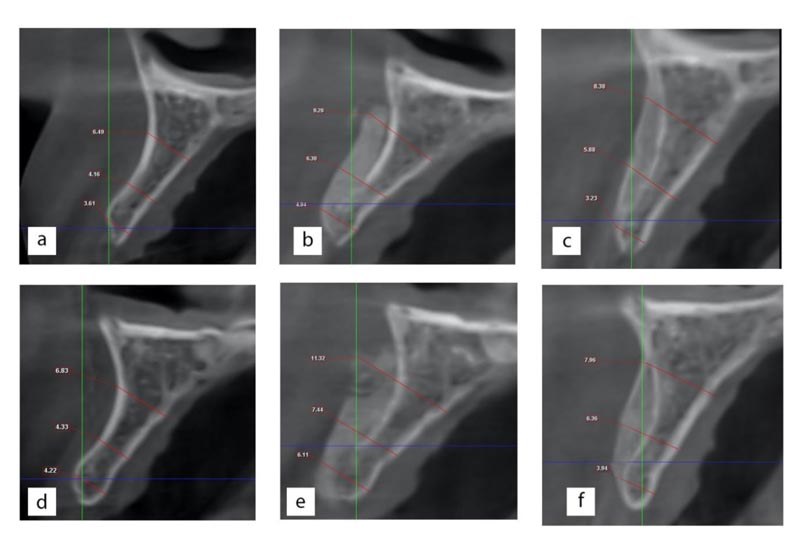
Radiographic evaluation was performed by an experienced radiologist. A postoperative CBCT was obtained one week following the surgery as well as 4 months later. To ensure standardization of images, images were re-oriented using the CBCT software (Planmeca Romexis). A specific cross-section perpendicular to the occlusal plane representing the future implant placement site was selected for evaluation. The same cross-section was generated in the preoperative, one week as well as four months postoperative images to evaluate the bone width. The width of the alveolar ridge of the edentulous space was measured at three points; 2mm subcrestal (point A), 6mm from the crest (point B), and 12mm from the crest (point C) (Fig. 2).
2.6. Temporization
All patients received a composite temporary restoration bonded to the neighboring teeth after sutures removal. No removable partial dentures were used to avoid interference with the graft healing.
2.7. Statistical Methods
Data were analyzed using Statistical Package for Social Science software computer program version 23 [SPSS, Inc., Chicago, IL, USA]. Data were presented in mean and standard deviation. Student’s t-test was used for comparing the age, gender and initial bone width of both groups, as well as the bone width at two different time intervals. ANOVA [Analysis of variance] followed by post hoc Bonferroni test were used for comparing bone width at the three different points at different time intervals. P-value less than 0.05 was considered statistically significant.
3. RESULTS
Sixteen edentulous spaces were grafted to increase the width of the alveolar ridge in 14 patients. The age of the patients recruited in this study ranged from 17 to 27 years. The mean age was 23.3 and 21.5 years for group 1 and 2, respectively. Three males and five females were recruited in group 1, while in group 2, there were six males and two females. There was no statistically significant difference regarding the age or gender between both groups [P= 0.2 and 0.3, respectively]. There were four grafted sites for missing central incisors and four sites of missing lateral incisors in group 1, and five central incisors and three lateral incisors sites in group 2.
3.1. Postoperative Complications
Two patients developed minor wound dehiscence, which was managed conservatively using hygiene instructions and chlorhexidine mouth rinses. No infection was encountered in any of the patients enrolled in this study.
3.2. Radiographic Evaluation of Bone Width
The changes in bone width were evaluated using CBCT. One week postoperatively, the bone width was measured at the three designated pints. In all cases, the graft material was seen in contact with the clearly visible cortex of the alveolar ridge. Four months postoperatively, another CBCT was taken to calculate the final bone width after graft resorption at the same three points. The original cortex was significantly less demarked (Fig. 2).
3.2.1. Initial Increase of Bone Width
The mean initial increase of bone width [postoperative versus preoperative] was 2.33±0.32mm [49.76±4.54%], 3.16±0.81mm [57.2±8.05%] and 3.69±0.82mm [52.22±13.1%] at point [A], [B] and [C], respectively, for group 1. In group 2, the initial increase of bone width at point A, B and C was 2.2±0.39mm [44.68±18.36%], 3.28±0.58mm [67±11.67%] and 4.08±0.94mm [66.57±22.49%], respectively. There was a highly significant difference between the postoperative bone width when compared with the preoperative width at all points in both the groups (Table 1).
3.2.2. Resorption of the Graft Material
Resorption of the graft material was measured as the reduction of bone width, 4 months postoperatively. The mean alveolar bone width of combined points A, B, and C at 4 months was compared to the corresponding bone widths measured one week postoperatively. Group 2 exhibited more graft resorption when compared to group 1. However, the difference was not statistically significant [P=0.08] (Table 2). The overall average resorption of the graft was 14.3±5.0% and 17.16±4.24% in group 1 and 2, respectively.
| Bone Width [mm] | P | |||
|---|---|---|---|---|
| Preoperative | Postoperative | 4 Months | ||
| Group 1 | ||||
| Point [A] | 4.71±0.74 | 7.04±1.03 | 5.65±0.99 | <0.001* |
| Point [B] | 5.49±1 | 8.65±1.73 | 7.61±1.52 | <0.001* |
| Point [C] | 7.23±1.35 | 10.9±1.69 | 9.73±1.51 | <0.001* |
| Group 2 | ||||
| Point [A] | 4.09±0.61 | 6.29±0.72 | 4.97±0.71 | <0.001* |
| Point [B] | 5.0±1.14 | 8.28±1.61 | 7.21±1.38 | <0.001* |
| Point [C] | 6.5±1.55 | 10.57±1.33 | 8.77±1.34 | <0.001* |
SD: standard deviation, P: Probability, *: Significance <0.05.
Test used: ANOVA.
| Point [A] | Point [B] | Point [C] | MEAN [A+B+C] | |||||
|---|---|---|---|---|---|---|---|---|
| [mm] | [%] | [mm] | [%] | [mm] | [%] | [mm] | P | |
| Initial Bone Width Increase | ||||||||
| Group 1 | 2.33±0.32 | 49.67±4.54 | 3.16±0.81 | 57.20±8.05 | 3.69±0.82 | 52.22±13.10 | 3.06±.49 | 0.6 |
| Group 2 | 2.2±0.39 | 44.68±18.36 | 3.28±0.58 | 67±11.67 | 4.08±0.94 | 66.57±22.49 | 3.19±.47 | |
| Bone width Reduction [Resorption] | ||||||||
| Group 1 | 1.39±0.23 | 20.06±3.94 | 1.04±0.26 | 8.28±6.89 | 1.19±0.33 | 10.83±2.42 | 1.05±.23 | 0.08 |
| Group 2 | 1.32±0.15 | 21.22±3.14 | 1.08±0.24 | 9.49±8.66 | 1.80±0.22 | 17.26±3.02 | 1.28±.24 | |
| Final Bone Width Increase | ||||||||
| Group 1 | 0.94±0.33 | 19.66±5.66 | 2.12±0.66 | 38.41±8.45 | 2.5±0.62 | 35.58±10.32 | 1.85±.38 | 0.07 |
| Group 2 | 0.88±0.47 | 22.09±13.60 | 2.20±0.43 | 45.33±11.04 | 2.28±0.91 | 37.66±18.56 | 1.78±.46 | |
SD: standard deviation, P: Probability, *: Significance <0.05.
Test used: ANOVA.
3.2.3. Final Bone width Increase
Bone width after resorption was measured 4 months postoperatively and compared to the preoperative values. Even after the resorption, the resultant final bone width was significantly higher compared to the preoperative widths in both the groups [P<0.001] (Table 2). Group 1 recorded more bone width after 4 months when compared to group 2. However, this was not statistically significant when compared to group 2 [P=0.074].
4. DISCUSSION
Horizontal ridge deficiency is a common procedure utilized for dental implant placement. Gultekin et al. [8] investigated the use of bone blocks versus guided tissue regeneration for horizontal augmentation of the maxillary alveolar ridge and reported adequate results with both techniques. In this study, the use of particulate bone graft was associated with a short recovery time and acceptable and controllable postoperative pain and discomfort. These advantages, in addition to the absence of a second surgical site, make this technique an attractive alternative to block grafts [21, 22].
Block grafts possess three main advantages: simple stabilization, space maintenance, and osteogenicity. Graft stability is a prerequisite factor for predictable bone healing. The movement of the graft disturbs the blood clot and could result in the formation of a fibrous tissue rather than bone. While the stability of block grafts can be easily provided using screws, the situation is more complicated with the use of particulate bone grafts. Different techniques have been described for graft stabilization in such cases, including the use of titanium-reinforced membranes, titanium mesh, screws, tacks or sutures [15, 23-26].
Sterio >et al. [27] reported displacement or resorption of about 50% of the graft added for horizontal ridge augmentation when no method of membrane fixation is used. This can be easily attributed to the displacement of the graft under the tension of soft tissue closure and muscle function. While the rigidity of MPM, by all means, is inferior to block graft, it is not a question of the degree of rigidity, but rather whether it is enough to resist the soft tissue tension and functional movement during the initial healing period to ensure predictable bone formation.
MPM is similar to PRF with the exception of containing mineralized particles within the fibrin network [28]. The fibrin network has a major role in most of the advantageous properties of MPM. In our study, the prepared MPM had a unique physical form through the bonding between the graft particles and the fibrin network. This form always preserved all the graft particles during adaptation to the host bed, which reflects the strength of the bonding between the particles. This unique form allows MPM to preserve the created form and resist displacement and maintain its integrity without the need for any rigid fixation device. Another unique feature of MPM is the ability to adhere to the bone surface after application, which further enhances its stability in the recipient bed [29].
Resorption of the graft material is inevitable. Thus, there is a consensus that over grafting is needed to overcome the anticipated resorption. Graft resorption, despite being part of the normal bone remodeling process, is attributed to multiple factors. Riachi et al. [30] reported that both the physical and chemical properties of the graft material have a significant effect on the rate of resorption after the sinus lift procedure. In our study, average graft resorption of 14-17% was observed. In accordance with this result, Gultekin et al. [8] reported a similar resorption rate of 12.5% associated with the use of a particulate bone and a collagen membrane fixed with bone tacks for horizontal ridge augmentation. In contrast, Mordenfeld et al. [10], who did not use any membrane fixation method, reported a high resorption rate reaching 46%. When a graft stabilization device is used, the reported rate of resorption is markedly lower [8, 31]. We found that MPM has a self-stabilizing property through maintaining the formed structure and the ability to adhere to the host bed without the use of any membrane fixation device. Perhaps this property, in addition to the accelerated bone healing, could have a role in the decreased resorption rate reported in this study.
The healing of a grafted site starts within the first 24 hours, where the blood fills the created space clots and starts to release growth, chemotactic and mitogenic factors. Newly formed capillaries provide nutrients and mesenchymal cells that differentiate to osteoblasts [6, 26, 32]. Resorption of the graft particles then takes place, which becomes replaced by mature bone [33]. Our results suggest that the platelets and growth factor content of MPM further enhances the healing process and accelerates the consolidation of the bone graft.
Exclusion of the fast-growing epithelial and connective tissue cells is essential for predictable bone healing [14, 15]. Membranes are basically used as physical barriers to separate the soft tissue cells from the graft material. The selection of the barrier membrane depends mainly on the operator preference depending on different factors such as the size of the defect, the desired tinging effect, the type of graft material and the procedure itself [6, 7, 33]. Collagen membranes are by far the most commonly used barrier membranes. The resorbability of the membrane is advantageous as there is no need for a second surgery for removal. Turri et al. [34], reported that collagen membranes actually act as a bioactive barrier as it promotes bone healing through multiple molecular events. Collagen induces the expression of a major osteoblastic and bone formation gene and, at the same time, upregulates the expression of BMP-2, FGF-2, TGF-b1 and VEGF at the RNA level throughout the healing period. However, this bioactive role of collagen membranes does not seem to have a pivotal role in the active process of bone formation during GBR, since multiple studies have reported comparable or even better bone formation with the use of non-resorbable barriers when compared with collagen barriers [35-37]. On the other hand, the use of collagen membranes itself has some potential disadvantages, including membrane exposure, infection, technique sensitivity and cost [38]. The results of this study further disprove any important bioactive role of collagen membranes in the process of guided bone regeneration.
The time needed for soft tissue exclusion in GBR procedures is debatable. It is needless to say that there is a minimum time required for any bone graft to consolidate and resist the penetration of soft tissue cells, which is the main function of barrier membranes in most cases. The literature lacks a definitive evidence regarding this particular issue. However, an approximate time can be derived from studies that described the use of non-resorbable membranes. The time of membrane removal is reported to be 3 weeks to 6 months following placement [39, 40]. The rationale of membrane removal after a relatively long time is to avoid the disturbance of the graft material to give time for bone maturation rather than the need for the cell occlusive property. Early membrane removal with predictable bone formation is reported in cases in which the removal can be done atraumatically without a re-entry procedure [40, 41]. Hoffman et al. [42] investigated the use of high-density PTFE membrane in socket preservation. The membrane was removed 4 weeks following the surgery. A bone biopsy was taken from the augmentation site at the time of implant placement and histologically evaluated for bone formation. The bone of normal architecture and good quality was observed. Yamashita et al. [43], through a histological study report that PRF becomes sparse after 21 days, and almost disappears after 28 days. Based on the similarity between MPM and PRF, it is suggested that MPM can maintain its form for more than 3 weeks. During this period, the sustained release of the growth factor can accelerate and promote bone healing. Considering MPM, it is not logical to expect that biological material can preserve its active properties for an extended time period. The release of growth factors and cytokines is expected to last for a maximum of eight days, which is the life span of platelets [44].
MPM is simply a PRF containing bone graft particles. PRF is reported to effectively release and retain a considerable number of cytokines and growth factors in the supportive three-dimensional fibrin that protects growth factors from proteolytic degradation of endogenous fibrogenic factors [45-47]. In our opinion, the stability of the fibrin network probably contributes to the cell occlusive property. Our study supports the results of Montanari et al. [12], who described the use of multiple layers of PRF membrane over graft materials and reported PRF to be an effective barrier against epithelial cell penetration.
A variety of growth factors, including bone morphogenic proteins [BMPs], platelet-derived growth factor [PDGF], and peptides of the parathyroid hormone [PTH], have been tested for local bone regeneration with promising results [21]. These bioactive materials are reported to induce new bone formation, accelerate the process of osteogenesis, and enhance both bone and soft tissue healing. Accelerated bone formation is always desirable in guided bone regeneration. As the process of bone healing advances, the tissue becomes more resistant to displacement, infection and resorption. El Moheb et al. [29] histologically evaluated MPM for grafting a mandibular defect in sheep. After only 14 days, he observed woven bone, osteocytes, osteoblast and fibroblast. When compared to bone graft material alone or in combination with PRF, MPM exhibited higher woven bone formation in the early healing period. Similarly, Cakir et al. [28] histologically compared MPM and PRF mixed with tricalcium phosphate particles and reported more bone formation with MPM. This is probably due to the bone graft being incorporated inside the fibrin network, which aids in accelerated revascularization of the graft by significantly supporting angiogenesis and enhancing the proliferation of osteoblasts [48-51].
It is fair to say that the present study has some limitations, including the small sample size, short observation period and the absence of a histological evaluation of the quality of the formed bone. Limited studies evaluating MPM are reported in the literature and further clinical and experimental studies are needed to augment the result of this study.
CONCLUSION
In conclusion, MPM can be successfully used for horizontal ridge augmentation without a covering barrier membrane.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Ethical Approval was obtained from Ethical Committee of Faculty of Dentistry, Mansoura University, Egypt (Code no:2020-84).
HUMAN AND ANIMAL RIGHTS
Not applicable.
CONSENT FOR PUBLICATION
All patients were given a detailed description of the study protocol and only patients who agreed and signed a written consent form were recruited.
AVAILABILITY OF DATA AND MATERIALS
Not applicable.
FUNDING
Declared none.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


