All published articles of this journal are available on ScienceDirect.
Manifestations in the Oral Mucosa of Erythromelalgia. A Case Report
Abstract
Introduction:
Erythromelalgia or erythermalgia, is a peripheral paroxysmal vascular disease of the distal extremities, although it can also affect the face, ears and knees. It is characterized by increased skin temperature, erythema and intermittent episodes of burning pain. Likewise, it can also be characterized by lesions of the oral mucosa.
Case report:
Here we present the case of a 6-year old female with EM presenting ulcerative and scaly lesions in the oral cavity and whitish linear branching ulcerative lesions on the tongue. Fabry disease and rheumatoid conditions were ruled out and there were no mutations in SCN9A gene. Treatment with voltage-gated sodium channel blockers (oxcarbacepine-Trileptal®) and anticonvulsants (Perampanel-Fycompa®) did not improve pain symptoms, however, performing moderate physical activity, use of comfortable footwear, and local application of cold resulted in the restoration of patient´s quality of life
Conclusion:
To our knowledge, this report described for the first time, the successful treatment of ulcerative lesions of the oral mucosa in Erythromelalgia. We believe that the treatments described here (exercise, comfortable footwear and local application of cold) could improve the quality of life of Erythromelalgia patients.
1. INTRODUCTION
Considered by National Organization for Rare Disorders (NORD) as a rare disease, Erythromelalgia or Erythelmalgia (EM) is a rare peripheral paroxysmal vascular disease that can appear in two forms: i) primary, associated with autosomal dominant inheritance (involving the SCN9A gene, which encodes the subunit protein of Nav 1.7 voltage-gated sodium channels) with no genetic correlation and of unknown cause, and ii) secondary, associated with different disorders such as autoimmune and neurological diseases, myeloproliferative medullary disorders, hypertension, venous insufficiency, diabetes mellitus, systemic lupus erythematosus, lichen planus, arthritis, monosodium urate crystal accumulation, or as a side-effect of certain drugs (bromocriptine or nifedipine) [1-3].
Its reported prevalence rate is of 0.36 individual per 100,000 inhabitants, with no differences between its primary and secondary forms [4], although being more frequent in women [5].
The early symptoms of primary erythromelalgia include increased skin temperature, erythema and intermittent symmetrical bilateral burning pain in the extremities, which can be relieved by immersion in cold water. The paroxysmal symptoms can be triggered by temperatures above 30ºC, vigorous exercise or general exhaustion [6-8]. In its secondary form, the onset of symptoms takes place in childhood or adulthood, and in this case, EM can be associated with chronic inflammatory oral mucosa diseases, compatible with forms of oral lichen planus [5, 9]. Its clinical features are commonly associated to nightly rest, although in the most severe cases, symptoms may persist during the whole day, which can affect patients’ normal activities to the point of partially or completely impairing them. For these reasons, it is necessary to establish a differential diagnosis of post-traumatic processes, peripheral neuropathies, Fabry disease (lipid storage disorder) or bacterial cellulitis.
Laboratory diagnosis tests focus on the analysis of SCN9A gene sequences, located in chromosome region 2q24.3 (size ±167.3 and 27 exons, 26 of which encode for a voltage-gated sodium channel, playing a relevant role in the nociception of inflammatory pain) [7, 10]. To date, 85 mutations of this gene have been described as related to the development of EM. Likewise, mutations in additional genes (SCN10A and SCN11A) can be associated with the pathogenesis of this disease, which is clearly related to patients’ family medical history [10, 11]. Nevertheless, certain authors reported the absence of SCN9A mutation and additional genes in the diagnosis of EM, concluding that the origin of the causative mutation is unknown, registering false negatives in 30% of cases [10, 12].
Treatment of primary erythromelalgia is focused on control and avoidance of trigger factors. In case of its secondary form, treatment is mainly based on the patient's record of pre-existing conditions, and individualizing the recommended drug regime (ASA, tricyclic antidepressant or sodium channel blockers, among others) [4, 5]. A clinical trial has been conducted on EM treatment using molecule inhibitors of SCN9A gene [13]. In this line, alternative therapies to deal with oral and mucosa disease have been explored. Thus, the successful use of herbal extract composed of baicalin (190 mg), bromelain (50 mg) and escin (30 mg) to mitigate the pain in oral mucosa after surgical intervention [14], the use of photocoagulation to treat vascular malformation of less than 3 cm in the oral and perioral area [15] and diode laser therapy to treat aggressive periodontitis that successfully reduced the levels of bacteria of orange complex and interleukin 1 have been reported [16].
Even EM is considered a rare disease, during the last 5 years, from 2013 to 2018, we have observed 8 young patients,, to be treated in the Stomatology Area of the Odontology Clinic of the University of Salamanca (Spain), with an age range of 4 to 11 years old, referred from the Internal Medicine Service of the University Clinic Hospital of Salamanca, diagnosed with EM and with lesions in the mucous membranes of the oral cavity, mainly during periods of exacerbation, in the zones of labial transition and tongue, in the form of whitish, ulcerative and desquamative lesions of linear character and arboriform disposition, very similar to the reticular lichen planus (Table 1).
Given that there is not an effective treatment for EM, our aim was to test if different adjuvants such us physical exercise, comfortable footwear and local application of cold in the mucosa’s lesions were able to reduce pain and restore the quality of life of the patient.
2. CASE REPORT
We present the case of a 6-year old female with no relevant medical family history, affected by symptoms of atypical febrile seizures since she was eight months old. These episodes were more frequent in hot weather and were associated with intense foot pain at night that can be relieved by applying cold poultices or by the ingestion of systemic analgesics. The clinical status worsened with age, an increase of physical activity and use of certain footwear. Physical examination of the feet revealed thinning of the epidermis, which presents an oedematous consistency and bilateral plantar erythema.
The patient showed a physiological process of dental replacement according to the order of time and sequence within normality, having replaced the deciduous teeth: 51, 61, 71 and 81 for permanent teeth. The permanent teeth were found in an eruptive period, and therefore, an open bite was observed in the anterior maxillo-mandibular area, as previously reported [17, 18], associated with poor occlusion and aesthetics, but without association with the skeletal characteristics typical of the skeletal open-bite pattern described by Ricketts [19]. The periodontal biotype found, according to visual parameters, was classified as thick, with a voluminous and bulging gum, associated with a predominance of width over the coronary length.
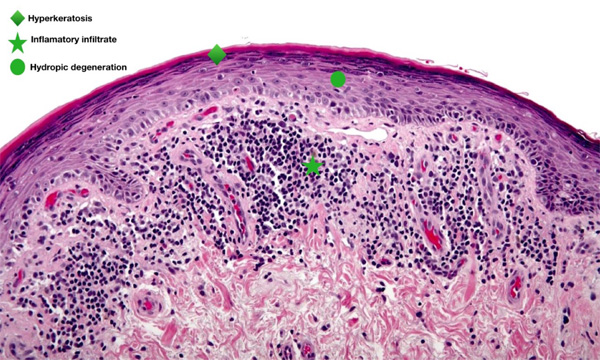
In periods of exacerbation, she presented ulcerative and scaly lesions in the oral cavity on the labial transition and lining mucosa (Fig. 1), and whitish linear branching ulcerative lesions on the tongue (Figs. 2, 3) similar to those described in the clinical manifestations of reticular lichen planus. After the treatment, there was a remission in these lesions (Fig. 4) whereas histological examination was compatible with lichen planus (Fig. 5).
These lesions usually heal within 3 to 5 weeks using a topical antiseptic agent (0.12% chlorhexidine) and a bioadhesive hyaluronic acid oral gel, because of their properties of repairing and regeneration of oral tissue. Analgesics (paracetamol) and nonsteroidal anti-inflammatory drugs (NSAIDs) i.e. ibuprofen are orally administered in episodes of intensification of the burning pain.
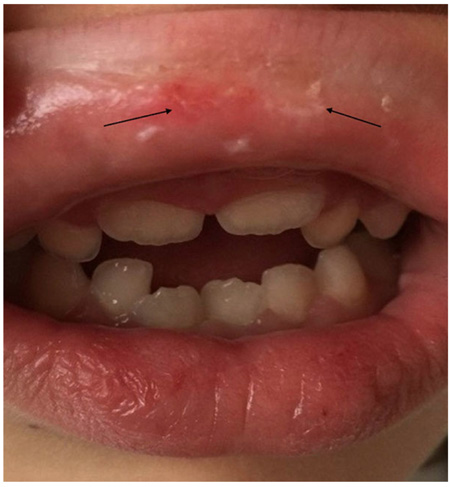
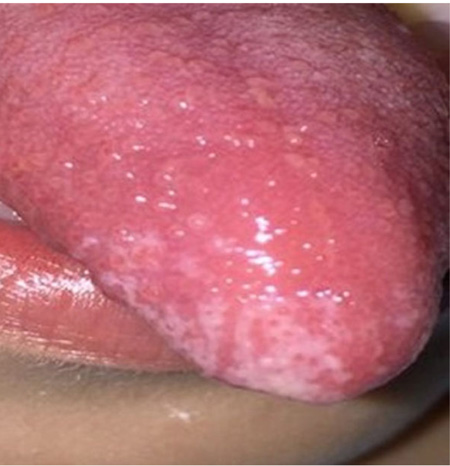
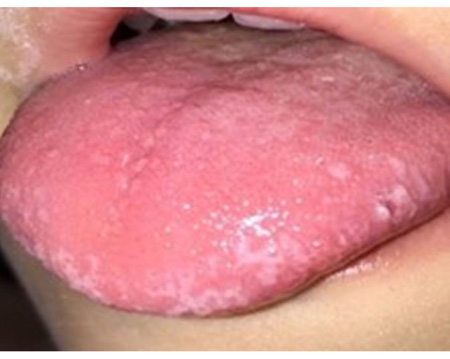
A complete blood count was performed, ruling out Fabry disease [20], secondary causes and rheumatoid conditions, and the results showed low serum iron values. Likewise, neurological tests and electromyography yielded normal results. A polysomnographic study revealed restless legs syndrome, and SCN9A gene sequencing showed no mutations in the area of study. Accordingly, pathognomonic findings and test results suggested a diagnosis of primary EM, in spite of the signs present in the oral mucosa, which are consistent with secondary EM.
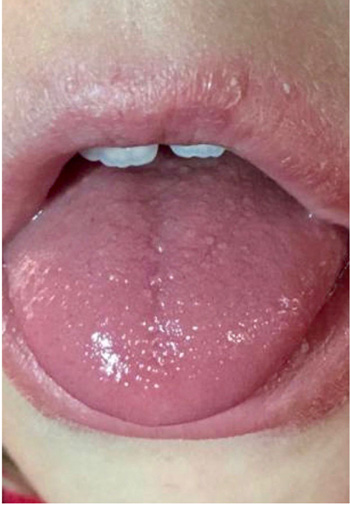
A treatment to raise iron levels was initiated, leading to an improvement in the sleep routine. Likewise, voltage-gated sodium channel blockers (oxcarbacepine-Trileptal®) and anticonvulsants (Perampanel-Fycompa®) were administered as previously reported [21], which did not improve pain symptoms. However, the use of common trigger prevention measures, such as avoiding exposure to temperatures above 30ºC, use of comfortable footwear, performing moderate physical activity and local application of cold proved to more effective and the patient enjoyed a practically normal quality of life.
| Patients (age) | Lesions in Labial Mucosa | Lesions in Tongue | Treatment | Duration of Treatment (days) |
Remission of Injuries (days) |
|---|---|---|---|---|---|
| 1 (4 years) | * | *** | Chlorhexidine 0,12+BHA | 21 | 18 |
| 2 (6 years) | ** | * | Chlorhexidine 0,12+BHA | 12 | 10 |
| 3 (11 years) | 0 | *** | Chlorhexidine 0,12+BHA | 21 | 16 |
| 4 (5 years) | * | ** | Chlorhexidine 0,12+BHA | 21 | 18 |
| 5 (11 years) | *** | *** | Chlorhexidine 0,12+BHA | 34 | 29 |
| 6 (5 years) | * | ** | Chlorhexidine 0,12+BHA | 21 | 16 |
| 7 (12 years) | *** | ** | Chlorhexidine 0,12+BHA | 28 | 26 |
3. DISCUSSION
Primary erythromelalgia is associated with episodes of erythema and bilateral symmetric and paroxysmal burning pain in the extremities, the treatment of which involves management of triggering factors [5, 13]. Secondary EM can be linked to a chronic inflammatory disorder of the oral mucosa, consistent with lichen planus [1]. Here, we have treated an EM case with adjuvant therapy based on exercise, application of cold in the lesion and wearing comfortable footwear combined with the use of analgesic. As a result, the patient returned to their regular activities and improved her quality of life.
The case report presented and diagnosed here, in principle as primary EM displays a mixed clinical pattern, with a combination of symptoms from both the primary and secondary forms of the disease, with evidence of ulcerative, scaly and reticular lesions of the oral mucosa that improved with the use of antiseptics and topical repairing gel. Four of the cases treated in our service (Table 1), were within a similar age range to that of the case described, and the other three were in 11 to 12 age range. The characteristics were similar to those reported by Oaklander and Klein, based on their analysis of 41 subjects with painful pediatric syndromes associated with small fibre polyneuropathies, compatible with erythromelalgia, in a population group with an average age of 12.3 years [22].
The signs observed in the clinical case here described, as well as in the series analysed in our service, included painful disabling lesions in periods of exacerbation, that affected patients’ quality of life. These episodic symptoms and their association with quality of life have been described by several authors.
Thus, Latessa states that in both its primary and secondary forms, associated with autoimmune diseases and neurological or haematic disorders, the episodic symptoms are related to severe disability in patients, concluding that early detection of the signs and symptoms is required to provide suitable treatment to improve patients’ quality of life [23].
Parker et al. [5], based on the analysis of a sample of 46 subjects, reported that the symptoms’ impact on quality of life in the patients studied, ranged between 15 and 58%, describing pain (87%) as the second most common symptom and concluding that regularly used drugs did not meet the required degree of effectiveness, this being the evidence of the need for further research on how to treat this rare pathology.
The presence of desquamative and ulcerative lesions manifested in the oral cavity and mucosa of labial transition during periods of exacerbation of the disease, could be associated with narrowing of the precapillary sphincters of the arteriovenous network, which would favor an increase in perfusion blood associated with a nutrient deficit of the blood with a low concentration of oxygen [24]; likewise, Low et al., described the case of a 41-year-old patient with primary EM, in whom the analysis of transcutaneous oxygen pressure by using a non-invasive electrode showed abnormal peripheral hemodynamics [25]. It is well known from the study of Fife et al., [26] that the deficit of oxygen partial pressure is associated with a higher prevalence of ulcerations. Thus, Parker et al., reported that 13% of the 46 patients analyzed in their cohort study (89% of women) had ulcers, fissures, and whitish linear branching ulcerative lesions on the tongue similar to those described in the clinical manifestations of reticular lichen planus, an inflammatory disease of unknown cause that can be classified as autoimmune and that can affect the inside of the cheeks, gums and tongue [5]. Even literature does not describe an association between this rare peripheral vascular disease and oral lichen planus, it does so with myeloproliferative diseases and connective tissue diseases. Thus, the cohort study conducted by Davis et al., [27] on 168 patients with EM (73% of women) reported a frequency of 10% of subjects with myeloproliferative diseases and Parker et al., already mentioned an association of 18% with connective tissue diseases [5].
In the case described, episodes of intensified burning pain improved with the combined use of analgesics and nonsteroidal anti-inflammatory drugs, although there was no response to either voltage-gated sodium channel blockers or to anticonvulsants. This effect may be explained because of the association of this pathology with the occlusion of the arterial micro articulation, probably a trigger point, associated with activation and platelet aggregation, so the use of high doses of aspirin could be beneficial [4]. In contrast, Michiels et al., concluded that the use of salicylates is ineffective in the treatment of neuropathic pain related to EM, recommending only the use of salicylates to reduce the prevalence of arterial thrombosis in subjects with thrombocytopenia and polycythemia vera [28]. The consulted literature also recommends treating painful episodes of ME with drugs that target the voltage-dependent sodium channels, a family composed of SCN9A genes that codified for Nav 1.1 to Nav 1.9 proteins associated with inflammatory pain. Thus, Geha et al. recommend the use of carbamazepine, an anticonvulsant (ef-fective in the treatment of neuropathic pain) that, in doses of 200 to 400 mg/day, would reduce subjects’ most painful episodes by almost 50% [12]. Similarly, Natkunarajah et al., recommended the combination of this anticonvulsant with gabapentin, since both drugs are effective in the treatment of peripheral neuropathic pain associated with mutations of the SCN9A gene, and act as a selective blocker of the sodium channels Nav 1.7, showing a reduction of the painful symptoms [29].
Likewise, the pharmacodynamics of systemic corticosteroids (prednisone 40 mg/day for 5 days) seems to be effective in EM cases associated with infections and lesions [4]. All the cases registered in our service were treated with the same types of drugs described in the case reported here, together with antibacterial and antifungal antiseptic oral rinses (0.12% chlorhexidine). Several authors [30-32], have recommended the combined use of anti-inflammatories and analgesics (capsaicin, lidocaine, ketamine, amitriptyline and phenytoin), alongside antiseptics in topical form, obtaining adequate responses.
CONCLUSION
EM is a rare peripheral skin disease, with a low prevalence and a complex etiopathogenesis, associated with neural and vascular complications. Since there is no effective drug treatment, here we demonstrate that controlling the factors that trigger the pain episodes and together with performing moderate physical activity, comfortable footwear and local application of cold would result in the remission or attenuation of patients’ symptoms, thereby improving their quality of life.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Ethical approval for the study, was obtained from the ethical Committee of University Clinic Hospital of Salamanca reference number is ER AZ 001 COD HU 13/1027.
HUMAN AND ANIMAL RIGHTS
No animals were used in this research. All research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2008.
CONSENT FOR PUBLICATION
Informed consent was obtained from the parents of the patients, before enrolled in the study.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
The authors thank the Odontology Clinic of the University of Salamanca.


