All published articles of this journal are available on ScienceDirect.
Sleep Apnea throughout the First Two Years of Life: Assessment of the Effect of Pacifiers in Patients with ALTE
Abstract
Background:
Apparent Life-Threatening Event (ALTE), Obstructive Sleep Apnea syndrome (OSAs) and Sudden Infant Death Syndrome (SIDS) are strongly correlated and few studies analyze the role that pacifiers play in such conditions.
Objective:
The aim of the study is to evaluate how pacifiers affect sleep apnea among children with a history of ALTE.
Methods:
10 subjects between 1 month and 2 years of age with a history of idiopathic ALTE were non-selectively recruited. Patients were subjected to cardio-respiratory monitoring at home for two consecutive nights; during the first night, the pacifier was not used, whereas the second night, the pacifier was used by the child for at least four hours sleep. Parents were given an assessment questionnaire to evaluate and report any irritation due to the pacifier use. All obtained traces were blindly analyzed by a pediatric specialist in sleep disorders, more specifically: Peripheral oxygen saturation (SpO2), heart rate (FC), Electrocardiogram (ECG) and the presence of apneas and/or hypopneas were assessed. In order to evaluate the differences between the average values collected from the two groups, a t-test was performed.
Results:
The use of the pacifier resulted in a statistically significant reduction in the number of pathological apneas/night (-1.7%; p = 0,0024), an improvement in the average SpO2 (+ 0.8%; p = 0.3328) and an increase in the value of the minimum SpO2 detected (+ 2%; p = 0.2571).
Conclusion:
The results show that the use of pacifiers improves the respiratory capacity of children that suffer from nocturnal apneas at night.
1. INTRODUCTION
SIDS (Sudden Infant Death Syndrome), commonly known as ‘cot death’ is the name given to the sudden and unexpected death of an apparently healthy infant, which remains unexplained even after performing a complete post-mortem investigation. Safeguarding behaviors appear to be the supine position and the use of pacifiers [1]. It also seems that children who use pacifiers have a lower awakening threshold, and a greater probability of arousal (higher probability/chance of arousal) in the event of hypoxia. Furthermore, the use of pacifiers modifies air spaces by bringing the tongue in an anterior position, a benefit that is maintained even if the baby loses the pacifier in his sleep [2, 3]. In 2006, the American Academy of Pediatrics (AAP) issued some guidelines regarding the use of pacifiers and established that the latter represents a safeguard element in the incidence/impact of SIDS [4]. Pacifiers also have the following benefits: They do not reduce the period of breastfeeding for premature babies and children after the 1st month of life; they help premature infants throughout neurological maturation. There are also some possible risks: They may cause dental misalignments if the use is extended beyond the 2 years of age; they may disadvantage breastfeeding if used during the first month of life, before starting breastfeeding; they may determine the risk of otitis media, especially after the first year of age [5]. Very similar to SIDS, the ALTE, a term introduced in 1986 during the National Institute of Health Consensus Conference on Infantile Apnea, is the acronym for ‘Apparent Life-Threatening Event’ or ‘Apparently Risky Event for Life’ (National Institutes of Health Consensus Development on Infantile Apnea and Home Monitoring, 1986) [6]. ALTE implies an episode that has frightened the observer, characterized by a varying association of apnea (central or occasionally obstructive), a variation of skin color (cyanosis, pallor or erythrosis), marked alteration of muscle tone (hypo or hypertonic), apparent suffocation. This episode, which usually lasts a few seconds, may occur during sleep, in waking hours or during a meal. In some cases, it resolves spontaneously, in other cases, it can be tackled thanks to the immediate intervention of a witness. In the case of more serious episodes, rescuers’ reanimation maneuvers are necessary and can escalate up to cardiopulmonary resuscitation [7]. In literature, it is estimated that about 5% of infants who have presented an episode of ALTE will die of SIDS: therefore, this data should act as an alert and not be underestimated. ALTE is not a diagnosis but represents an idiopathic clinical manifestation (IALTE, about 50%) or secondary to multiple morbid conditions. The most common causes are gastrointestinal (about 50%), neurological (30%), respiratory (20%), cardiovascular (3%), metabolic and endocrine (< 5%) or it might include other reasons, such as abuse episodes [8]. The real incidence of episodes is difficult to define since the values reported in literature swing dramatically between 0.6 and 10 among 1000 live births [9, 10]. Most episodes occur in children under the age of one and the average age is of 8 weeks [11, 12]. It is not known whether the use of pacifiers in sleep can reduce the risk of ALTE or sleep apnea in the first two years of life. Obstructive Sleep Apnea Syndrome (OSAS) in children is defined as breathing disorder during sleep characterized by extended partial upper airway obstruction and/or intermittent complete obstruction (obstructive apnea) that disrupts ventilation during sleep and fragments sleep patterns. It is estimated that 3% to 26% of children are usual snorers and 1.2% to 5.7% of the general pediatric population presents OSAS. The peak incidence was found between 2 and 8 years of age and is widely identified among the causes of a discrepancy between the size of the lymphoid tissue and airway size. OSAS takes place with the collapse of the upper airway during sleep as a result of an unbalance between the structure of the upper airway due to several factors such as adenotonsillar hypertrophy, craniofacial anomalies, the tone of the upper airway neuromuscular and obesity. The consequences that OSAS can generate are neuropsychological and cognitive impairment, systemic and pulmonary hypertension and endothelial dysfunction which all occur quite frequently. Most articles demonstrate that protruding children’s jaw reduces obstruction during sleep [13]. These considerations lead us to hypothesize that pacifiers can be a useful tool for better nighttime breathing. For this reason, the aim of the study is to evaluate how pacifiers impact sleep apnea during the first two years of life in children with a history of ALTE.
A positive response for a severe respiratory condition may also turn out as beneficial for simple conditions of difficult nasal breathing.
2. MATERIALS AND METHODS
For this study, 10 children between 1 month and 2 years of age with a history of idiopathic ALTE, were non-selectively gathered at the Respiratory Disorders of Sleep Department of the University of Insubria (Varese) as an experimental group. The study was brought to the attention of the provincial Ethics Committee of Varese, which expressed a favorable opinion and authorised the study (number 110-2017). The sample consisted of 5 males and 5 females, all of them born in 2016. Informed consent was signed by the parents of the patients who were eligible for the study. During the visit, all the information regarding the study was provided, together with the Getemed Vitaguard VG3100 monitor, which allows the monitoring of the peripheral arterial Oxygen Saturation (SpO2), the heart rate and the detection of apneas and/or hypopneas. All procedures linked to the nocturnal cardiorespiratory recording (placing of the cardiac electrodes and of the SpO2 sensor) were explained. Patients were subjected to cardiorespiratory monitoring at home for two consecutive nights; during the first night, the pacifier was not used, whereas the second night, the pacifier was used for at least four hours of sleep. The next morning, parents returned to the monitor with a journal, in which they noted sleep and waking hours, timelapse of the pacifier usage, the quality of sleep and possible problems during the night. Parents were given an assessment questionnaire to evaluate and report any irritation due to the use of the pacifier. The pacifier used by the sample group showed the following features: The recess, the inclined profile and the reduced vertical dimension of the body which allowed the natural (front and upper) positioning of the tongue inside the oral cavity, supporting its natural movement. This condition brings the tongue forward and as a result, the upper airway is more open, this is useful for the nocturnal period in order to ensure a physiological respiration and a correct craniofacial growth; the simulation of the palatine rugae at the base of the pacifier body stimulates the tongue and the tactile stimulus thanks to the contact with the palate. These raised sections of the pacifier reproduce the natural rugae present on the palate, crucial to position the tongue up and therefore stimulate the growth of the maxilla without negatively affecting the mandible growth. All the traces obtained were blindly analysed by a pediatric specialist in Sleep Disorders. The recorded parameters were the following: peripheral Oxygen Saturation (SpO2), heart rate (FC), Electrocardiogram (ECG) and the presence of apneas and/or hypopneas. To evaluate the differences between the average values collected from the two groups, a t-test was performed (P<0.05 was considered as significant).
3. RESULTS
Within the experimental group, the basal recording without pacifiers showed an average of 3.2 pathological apnea/night, whereas with the use of pacifiers, the average result was 1.5 pathological apnea/night: in all cases, there was a significant reduction in nocturnal apnea events (p-value 0,0024). Only when mothers reported the impossibility of using the pacifier, there was no positive impact. Therefore, the use of the pacifier resulted in a reduction of pathological apnea cases/night (-1.7%), a nonstatistical improvement in the average SpO2 (+ 0.8%) and an increase in the value of the minimum SpO2 (+ 2%) (Table 1). In most cases (90%), there was a linear and constant progress of the respiratory capacity, with a reduction in the number of apneas and improvement of SpO2 (Figs. 1-3). No questionnaire reported problems or irritations to the skin caused by pacifiers.
| - | Control | With Pacifier | Difference | P Value |
|---|---|---|---|---|
| AHI average | 3,2 | 1,5 | - 1,7 | 0,0024 |
| SpO2 average | 96,9 | 97,7 | + 0,8 | 0.3328 |
| SpO2 minimum | 87,7 | 89,7 | + 2 | 0.2571 |
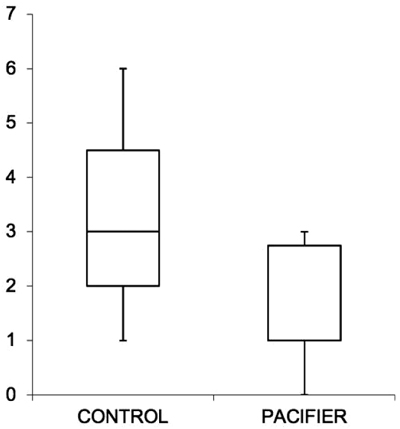
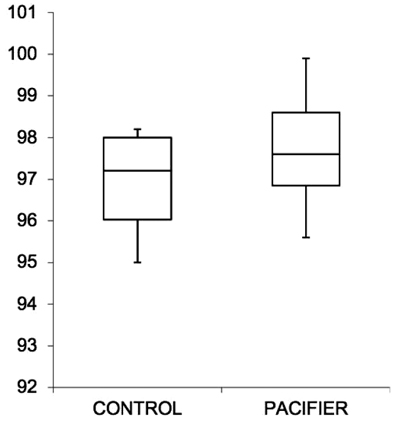
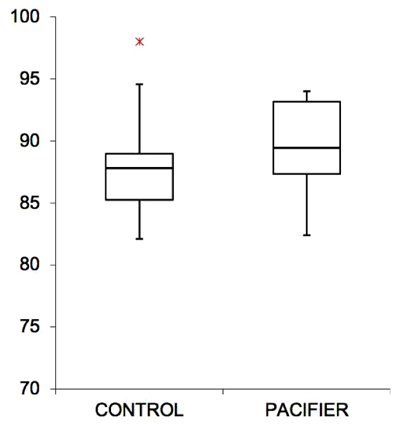
4. DISCUSSION
The results of this analysis show that the use of the pacifier improves the respiratory capacity of children with nocturnal apneas at night. This effect is particularly reassuring as it enables the protection/treatment of children affected by OSAS, although it does not solve the root causes of the problem. Practically, it could be considered as an effective conservative therapy before the age of 2 in pediatric patients suffering from OSAS. Referring to the literature, the effectiveness of the pacifier in resolving minor respiratory disorders, such as nocturnal oral breathing or oral breathing during wakefulness, could be argued. Cozzi in 2002 showed how pacifiers are able to increase the breath flow in patients with oral respiration, in particular by analyzing oxygen saturation at the arterial level. The rationale of this efficacy is probably due to the jaw advancement induced by the pacifier [14]. To confirm this rationale, Levrini and Nosetti in 2018 conducted a study on OSAS patients, applying silicone preformed oral devices with the purpose of advancing the patients’ jaw and a reduction in the hypomea apnea index was highlighted [15]. On this subject, in 2007, Tonkin showed that pacifiers in preterm births cause a significant yet small advancement of the jaw, which is stable once the pacifiers get removed. Tonkin himself has published a case of patients suffering from severe retrognathia in which the use of pacifiers produced an increase of 7 mm in the pharyngeal gold space. However, the application of a jaw advancement device such as a pacifier does not exclusively provoke a forward repositioning of the jaw but also a downwards movement; for this reason, it is better to identify it as ‘mandibular repositioning’ [16, 17]. The mandibular repositioning devices can, therefore, contribute at improving the airflow during sleep, due to the reduction of the upper airway collapse and the improvement of the muscle tone, as suggested by Villa in 2012 [18]. The rationale of these phenomena is to advance the jaw and tongue in order to have a positive impact on the caliber and function of the upper airway [19]. This may be due to the fact that children’s upper airways reflect the concepts of the Starling resistance model, with a complex multi-system involvement to regulate patency. The Starling resistance model can be described as a tube with a rigid proximal and distal component but with a flexible central portion. This flexible region is exposed to the surrounding tissue pressures and collapses if the pressure of surrounding structures increases. The term used to describe the moment in which the pressure of the pharynx decreases is Critical Pressure (PCRIT). In order for the flexible portion to remain previous, the pressure in the proximal and distal segment must be greater than the PCRIT. To create a complete occlusion, the upstream and downstream pressure of the flexible segment must be lower than the PCRIT. However, limited respiratory flows (hypopnea) take place if a certain flow of air is maintained during inspiration, with the upstream pressure remaining higher than PCRIT. This pattern occurs during severe snoring episodes, by which the airflow fluctuates during closure and reopening of the upper airway [20]. According to this theory, we can assume that pacifiers could increase the PCRIT in the upper segment, but not enough to bring the flow back to the highest PCRIT values. Nevertheless, pacifier use should not be underestimated as the jaw advancement in growing subjects determines a positive stimulus to the growth of the mandibular body. Cranio-facial morphology plays a key role in the pathophysiology of Pediatric Obstructive Sleep Apnea Syndrome (OSAS). Upper airway obstruction, as a result of hypertrophy of lymphoid tissue or skeletal deficiency in a growing child, can prompt oral breathing, endangering normal craniofacial development, resulting in a reduced upper airway thickness and promoting breathing disorders during sleep. Therefore, it is reasonable to affirm that pacifiers generate an immediate relief but can also be a treatment for the stimulation of the mandibular growth and, as a consequence, for a stable opening of airway spaces, useful for correct breathing behind the tongue. However, in accordance with other studies [21], it is theoretically possible that snoring itself is a more serious respiratory phenomenon in young children than apnea or hypopneas. If snoring causes significant physiological changes (eg, inflammation) to an equal or greater extent of apneas and hypopneas, children with Primary Snoring may not actually be affected by a ‘mild’ form like children with OSAS. Obviously, the protective and preventive role that pacifiers play with SIDS should not be underestimated, especially in studies that describe the mechanisms and underlying this specific benefit [22]. Based on our results, it has not been demonstrated that the etiology of the problem has been resolved [23].
CONCLUSION
The study results may suggest that using pacifiers during night-time is useful to improve the respiratory capacity of children. The benefits could be due to the healing of the oral muscle balance and the forward and lower repositioning of the jaw with an immediate increase of the upper airway patency. These consi- derations place the pacifier as an innovative and necessary tool, not only for a correct growth of the jaws and a development of the oral muscular system but also for the aspiration of determining correct nocturnal breathing, essential for systemic health. In this regard, it is necessary to underline that these results are associated with the use of a specific pacifier with an anatomical and functional geometry; it is not possible to attribute such benefits to general pacifiers, because the geometry of each pacifier could result in better or worse effects. The achieved data is interesting and promising, but it is necessary to expand the study applying it to more cases. Moreover, it could be useful to practice this research through the use of a polysomnograph with a greater number of channels to be able to detect the effects of pacifiers on sleep efficiency and identify the various types of apneas (central, obstructive, mixed), but also to analyze subjects that suffer from simple snoring.
LIST OF ABBREVIATIONS
| ALTE | = Apparent Life-Threatening Event |
| OSAS | = Obstructive Sleep Apnea Syndrome |
| SIDS | = Sudden Infant Death Syndrome |
| SpO2 | = Oxygen Saturation |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was approved by the institutional ethical review board Ospedale di Circolo, Fondazione Macchi in Varese (Italy) (Study number: 2017-110).
HUMAN AND ANIMAL RIGHTS
All research procedures followed were in accordance with the Helsinki Declaration of 1975, as revised in 2013. Written informed consent for participation was obtained from each patient.
CONSENT FOR PUBLICATION
Informed consent was signed by the parents of the patients who were eligible for the study.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


