All published articles of this journal are available on ScienceDirect.
Oral Rehabilitation of Patients with Ameloblastoma of the Mandible. Clinical Results in Three Patients with Different Bone Reconstruction Techniques
Abstract
Background:
Ameloblastoma is the second most common odontogenic tumor. It shows a locally aggressive behavior, with a high level of recurrence. Wide resection of the jaw is recommended for treatment of ameloblastoma. However, radical surgery causes an abnormal mandibular movement, facial asymmetry, and masticatory dysfunction.
Methods:
Three cases of different types of ameloblastoma is presented, with different reconstruction techniques including Non-Vascularized Bone Graft (NVBG), Osteocutaneous Fibula Free Flap (OFFF), and Deep Circumflex Iliac Artery flap (DCIA).
Results:
In all three cases the tumor site was successfully reconstructed to obtain very good esthetic results as well as functional oral rehabilitation with implants and fixed prosthetics for optimal masticatory function.
Conclusion:
For reconstruction of the mandible, we prefer bone grafts from the iliac crest. The natural curvature and variable bone height offer a very good reconstruction of the defect.
1. INTRODUCTION
Ameloblastomas represent approximately 1% of all tumours and cysts of the maxilla and mandible, and 10% of all odontogenic mandibular tumours. In the maxillofacial region, the most common site of ameloblastomas is the angle and the body of the mandible. Clinically, these tumours are characterized by slow growth and painless symptoms, which may delay an early diagnosis and cause facial deformity, increased tooth mobility, and ulceration of the oral mucosa [1]. Ameloblastomas have been reported in both sexes and have been diagnosed in patients with a wide range of ages (20-50 years) [2]. The etiology of ameloblastoma is not understood, but according to the World Health Organization (WHO 2005), the ameloblastoma is derived from an odontogenic epithelial origin [3]. Causes may include injury to the mouth or jaw, infections of the teeth or gums, or inflammation of these same areas. Recent studies imply that there may be a host-response factor of periodontal disease that could trigger the neoplasm [4].
It has been realized for some years that ameloblastomas occur rather frequently in Africans [3]. Moreover, in black African countries, these lesions tend to occur in relatively young patients compared to those in white Africans, Europeans, and Asians [4].
Radiographically, ameloblastoma appears either unilocular or multilocular. Although the structure of these lesions can be detected on panoramic radiographs, Computed Tomography (CT) is to be preferred, due to the helpfulness of this imaging technique in determining the contours of the lesion, expansion of buccal and lingual cortical bones, resorption of adjacent teeth, and its extension into soft tissue [4]. Compared to CT, Magnetic Resonance Imaging (MRI) is slightly superior for establishing the exact extent of advanced maxillary ameloblastoma and the invasion of tumor mass into adjacent soft tissue [5].
The recommended treatment for ameloblastoma is surgical resection of tumour with safety margins of 1-2 cm, due to its aggressive recurrence rate [6, 7]. Defect or discontinuity of the alveolar ridge of the mandible can be restored by different reconstruction flap techniques. The aim of this article is to present three cases of ameloblastoma, from diagnosis to rehabilitation, reconstructed with different flap techniques. The rationale of the study is to obtain the natural curvature and height of the mandible in order to rehabilitate a good functional occlusion for the patient.
2. REPORTS OF CASES
2.1. Case 1
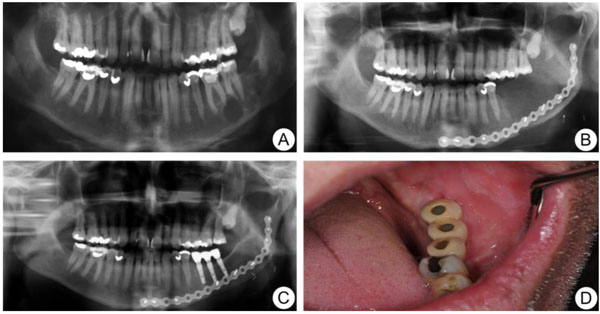
A 54-year-old healthy Caucasian man was referred to the Department of Oral and Maxillofacial Surgery, Linköping University Hospital, in March 2005, due to the finding of a multilobulated cystic lesion in a routine panoramic radiograph at the left side of the mandible, with extension of the lesion from corpus mandible to ramus mandible (Fig. 1a). A biopsy was taken and the histological findings were consistent with ameloblastoma. There were no signs of malignancy. The patient underwent surgical removal of the mandible, including the tumor, with resection margin from tooth 36 to the anterior border of the ramus mandible. The pre-operative height and width of the mandible in region 36 was 26 mm and 13 mm. The defect was immediately reconstructed by Deep Circumflex Iliac Artery flap (DCIA) with a bone piece of 6 × 2.4 cm (Fig. 1b). The post-operative reconstructed site had an increase in both height and width to 29 mm and 15 mm. After a healing period of one year, some resorption of the bone had occurred, but three implants (Microthread® conical; Astra Tech AB, Mölndal, Sweden; 4.5 mm in diameter and 11 mm in length) were inserted at the left side of the mandible in a two-step fashion without complication. Six months later, the bridge was connected by the referring dentist and the patient could start using his new teeth (Figs. 1c & d). No recurrence of the tumour was seen at 5-year follow-up.
2.2. Case 2
A 42-year-old man of central African descent was referred to the Department of Oral and Maxillofacial Surgery, Umeå University Hospital, for a fast-growing mass at the right side of the facial skeleton in 2005 (Fig. 2a). Intraoral biopsy revealed histological features of ameloblastic carcinoma. The patient underwent hemi-mandibulectomy and the mandible was reconstructed with a free vascularized fibular transplant at Umeå University Hospital, Sweden. A postoperative CT-scan 3D-model showed complete healing of the fibula flap (Fig. 2b). In 2006, the patient was referred to the Department of Oral and Maxillofacial Surgery, Linköping University Hospital, Sweden for implant-supported rehabilitation of the right mandible. The bone height of the mandible was around 20 mm and deemed inadequate to receive an implant-supported bridge (Figs. 2b & c), the width however, of circa 12 mm was sufficient. Thus, we decided to extend the bone tissue in a vertical direction with distraction-osteoneogenesis technique. Buccal and lingual plate horizontal corticotomy was performed and the distraction device was placed perpendicular to the corticotomy (Fig. 2d).
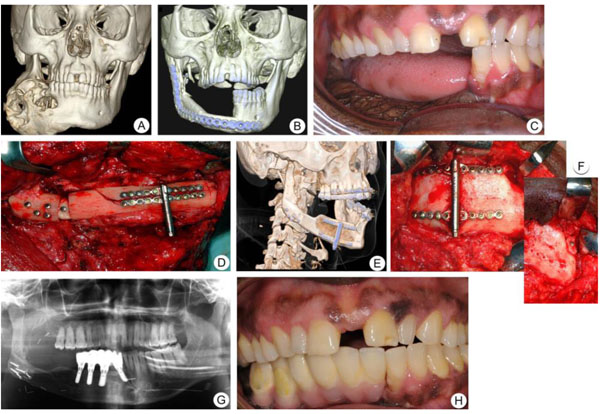
After a latency period of 7 days, the superior segment was moved upward at a rate of 1 mm/day in two fractions of 0.5 mm. This procedure was repeated for 14 days. Radiographic examination and clinical photographs at the end of the consolidation period confirmed that there was an excellent ossification between the mandible and the distracted segment (Figs. 2e & f). Six months after the consolidation period, the distractor was removed, and the mandibular height in the area of interest for implant fixation had increased to around 31 mm and width unchanged at circa 11 mm. Consequently, four implants were placed at the right side of the mandible (Fig. 2g). Prosthetic rehabilitation was performed after an osseointegration period of 4 months (Fig. 2h) and the patient was followed up for seven years. A high implant survival rate, with good patient satisfaction, was achieved with this therapy.
2.3. Case 3
In 2014, a healthy 11-year-old girl of African origin was referred to the Department of Maxillofacial Surgery, Ryhov Hospital, Jönköping, Sweden, due to a cyst-like lesion measuring 11.5 × 8.5 mm at the left side of the mandible. The intraoral radiograph showed a radiolucent lesion with a relatively well-defined margin between tooth 34 and 35 (Fig. 3a). No lining epithelium was found in the exposed cystic lesion at the time of surgery. Thus, radiological and surgical findings confirmed the diagnosis of Traumatic Bone Cyst (TBC). Moreover, six months later, a Cone-Beam Computed Tomography image (CBCT) showed a multicystic radiolucency measuring 28 × 20 mm located at the left side of the mandible in the same place as the previous lesion (Fig. 3b). A biopsy was performed and the histological diagnosis revealed ameloblastoma. In August 2015, the patient was referred to the Department of Oral and Maxillofacial Surgery, Linköping University Hospital, Sweden for further treatment and rehabilitation.
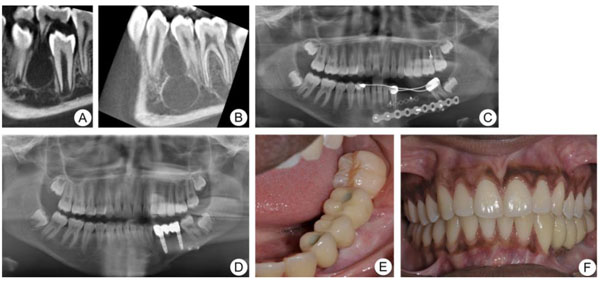
A segmentary resection with an 8-mm safety margin and immediate reconstruction with nonvascularized iliac bone graft (3 × 2.5 cm) and a titanium plate was performed (Fig. 3c). Twelve months later, the panoramic radiograph showed complete graft integration and the patient received two dental implants (Microthread® conical; OsseoSpeed, Mölndal, Sweden; 4.2 mm in diameter and 11 mm in length) to allow an implant-supported fixed complete prosthesis. At the one-year follow-up post-implantation, no marginal bone loss was seen and the implant-supported prosthesis was functioning well with no signs of gingival pockets (Figs. 3d-f).
3. DISCUSSION
Ameloblastoma is a common odontogenic tumour, and the treatment of these tumours is highly debated. Ameloblastoma of the jaw is considered to have aggressive growth with a high rate of recurrence if not removed appropriately. The possible risk of recurrence thus explains the need for a long-term follow-up period. The most common clinical manifestation is swelling and/or pain [1, 2]. However, a variable proportion of cases have been diagnosed incidentally by routine radiographic examination [8]. Several strategies have been used for the treatment of ameloblastoma of the jaw, including surgical resection, conservative treatment, enucleation, and bone curettage [6, 9]. The importance of wide surgical resection to minimize the risk of recurrence has been discussed previously in the context of a randomized trial involving 48 patients [10]. Eleven of the 48 were initially treated with radical resection and none recurred. Twenty-two of the remaining 37 who were initially treated with conservative resection presented with recurrences. Sixteen of the 22 then had conservative secondary resections, which resulted in further recurrence in 6 patients. The authors showed that initial radical resection is therefore superior to conservative management.
Although segmental osteotomies are recommended, this treatment has a high morbidity and affects the patient’s quality of life. However, with immediate reconstruction of the jaw and prosthetic rehabilitation with dental implants there is a good chance of restoring the masticatory function and esthetics in these patients. In this case series, an 11-year-old patient with ameloblastoma of the jaw underwent surgical osteotomies and reconstruction with nonvascularized iliac graft bone. Two dental implants were inserted in the grafted bone at the left side of the mandible. There were no signs of implant displacement after 18 months of radiographic follow-up (Fig. 3e). The treatment of ameloblastoma in children is complicated because of continuing facial growth, especially in the posterior mandible [11]. It has been suggested that there is a risk of dental implants becoming embedded or displaced as the jaw grows. However, this has not been studied in grafted bone in children.
Several other surgical interventions have been reported in the treatment of ameloblastoma, including enucleation and enucleation and curettage [12]. However, it is questionable why surgeons would choose these therapies with a high recurrence rate for aggressive neoplasms.
We have presented two cases of mandibular ameloblastoma with successful management involving bone resection and immediate reconstruction using DCIA and OFFF. Previous studies have shown no significant difference between these two surgical techniques regarding pain, morbidity, and quality of life [13, 14]. However, DCIA shows greater bone resorption with a remaining volume of 88% after 48 months, as compared to OFFF with 95% remaining volume after the same follow-up period [14].
One disadvantage of using the DCIA flap is the unnecessary bulk of the “obligatory muscle cuff” and abdominal subcutaneous fat, which makes it uncomfortable for the patient during the early healing phase [15-18]. However, the natural curvature and adequate bone height offer the possibility of exact reconstruction of the jaw. Thus, implant insertion may be easier and rehabilitation may be quicker.
CONCLUSION
Ameloblastoma is a locally aggressive neoplasm. In order to reduce the risk of recurrence, wide resection has been recommended. Immediate reconstruction with an iliac crest transplant is a favorable flap option. There is no need for volume-enhancing treatment prior to implant therapy. In addition, there is the added benefit of reduced rehabilitation time.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was approved by the Regional Committee for Ethics in Linköping, Sweden (2015/415-31).
HUMAN AND ANIMAL RIGHTS
No Animals were used in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
This study was supported by the Department of Oral and Maxillofacial Surgery, Linköping University Hospital, Linköping, Sweden.


